Acids & Bases - Chemistry
Acids & Bases - Chemistry
Acids & Bases - Chemistry
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
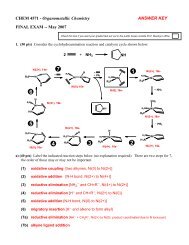
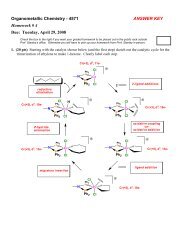
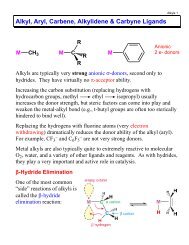
<strong>Acids</strong> & <strong>Bases</strong> 344) For almost all acids, the more electronegativeatoms present (like O or F) the higher theacidity of that acid.Reasoning: the more electronegative atoms present, themore the negative charge on the anion will be pulledtowards these atoms and away from the atom that the H + isassociated with.Example:HClO HClO 2 HClO 3 HClO 4Hypochlorous Chlorous Chloric PercloricK a = 3 x 10 −8 K a = 1 x 10 −2 K a = 5 x 10 2 K a ≈ 1 x 10 10Electrostatic charge potential (ECP) surface plots for ClO −through ClO 4−anions (no H + ). The red color (dark) indicatesmore negative charge and a stronger electrostatic attractionto the H + cation (weaker acid). Positive charge is indicatedby the blue color (darker color on center atom).