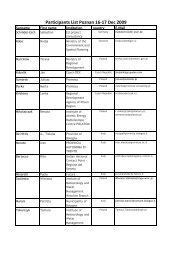
OP 3.3.1 Evaluation of clinics in every region - Central Europe
OP 3.3.1 Evaluation of clinics in every region - Central Europe
OP 3.3.1 Evaluation of clinics in every region - Central Europe
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
3.1.3. Analyse <strong>of</strong> exist<strong>in</strong>g approaches –projects + <strong>3.3.1</strong>. evaluation <strong>of</strong> <strong>cl<strong>in</strong>ics</strong><br />
Institute for protection <strong>of</strong> health<br />
social medic<strong>in</strong>e with health statistics and health promotion, hygiene and<br />
health ecology with laboratory diagnostics <strong>of</strong> foodstuffs, water and other<br />
environmental parameters, and monitor<strong>in</strong>g <strong>of</strong> <strong>in</strong>fectious diseases with<br />
laboratory diagnostics <strong>of</strong> <strong>in</strong>fectious agents <strong>in</strong> humans.<br />
3.1.3. Analyse <strong>of</strong> exist<strong>in</strong>g approaches –projects + <strong>3.3.1</strong>. evaluation <strong>of</strong> <strong>cl<strong>in</strong>ics</strong><br />
Technology platform; I Techmed<br />
Ma<strong>in</strong> areas <strong>of</strong> work<br />
physical medic<strong>in</strong>e and medical rehabilitation<br />
• IPL technologies<br />
• dermato-venerology<br />
•ophthalmology<br />
• dental programmes, development <strong>of</strong> implants (materials, technologies),<br />
<strong>in</strong>frastructural research<br />
• preventive medic<strong>in</strong>e<br />
• health tourism programmes<br />
• wellness programmes<br />
• development <strong>of</strong> recomb<strong>in</strong>ant prote<strong>in</strong>s<br />
• knowledge <strong>of</strong> physical-chemical and biological properties <strong>of</strong> pharmaceutical<br />
compounds<br />
• development and assessment <strong>of</strong> delivery systems<br />
• development <strong>of</strong> biotechnological products<br />
• research <strong>of</strong> new substances<br />
• synthesis <strong>of</strong> generic drugs<br />
• telemedic<strong>in</strong>e and eHealth<br />
Ma<strong>in</strong> goals;<br />
establishment <strong>of</strong> a technological platform for <strong>in</strong>novative and support<strong>in</strong>g<br />
technologies <strong>in</strong> medic<strong>in</strong>e on a national and <strong>Europe</strong>an level.<br />
09.08.2010 – Lead Partner 14 / 23