Non-DLVO, steric and fluctuation forces
Non-DLVO, steric and fluctuation forces
Non-DLVO, steric and fluctuation forces
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
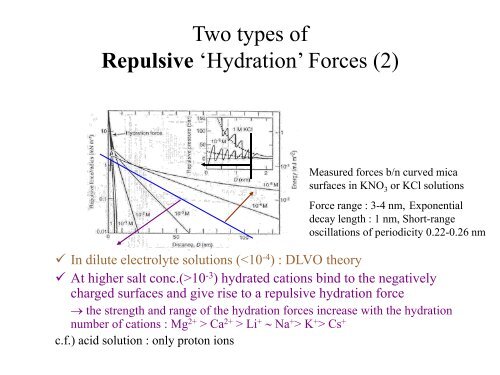
Two types ofRepulsive ‘Hydration’ Forces (2)Measured <strong>forces</strong> b/n curved micasurfaces in KNO 3 or KCl solutionsForce range : 3-4 nm, Exponentialdecay length : 1 nm, Short-rangeoscillations of periodicity 0.22-0.26 nm In dilute electrolyte solutions (10 -3 ) hydrated cations bind to the negativelycharged surfaces <strong>and</strong> give rise to a repulsive hydration force→ the strength <strong>and</strong> range of the hydration <strong>forces</strong> increase with the hydrationnumber of cations : Mg 2+ > Ca 2+ > Li + ∼ Na + > K + > Cs +c.f.) acid solution : only proton ions