Genetic Testing: Who Should Do the Testing and What Is theRole of Genetic Testing in the Setting of <strong>Celiac</strong> <strong>Disease</strong>?George S. Eisenbarth, M.D.At present, there is public health screening for a series of disorders that can be identifiedshortly after birth. These disorders are all relatively infrequent, but include diseases such ashypothyroidism (1/4,000 newborns) and phenylketonuria (1/13,500 to 1/19,000), and for someU.S. States and France, even diseases such as cystic fibrosis are assessed with newbornscreening. The major driving force for screening is the ability to identify a treatable disorder withsignificant incidence such that there is a public health risk. Essentially all of the disorders areidentifiable as a neonatal disorder requiring immediate therapy (e.g., neonatal hypothyroidism)or are Mendelian disorders with high penetrance (e.g., cystic fibrosis). The threshold fordetermining the cost-effectiveness of screening for a given disease is very much influenced bythe infrastructure put into place for newborn screening due to the almost universal screening forphenylketonuria and hypothyroidism. As we consider celiac disease, we are considering a verycommon disorder of children and adults, a disease that at present usually does not manifest(expression of transglutaminase autoantibodies) until after 2 years of age, and most important, acomplex genetic disorder. For complex genetic disorders that do not manifest in neonates and inparticular immune-mediated diseases, which is determined by HLA alleles, we lack a publichealth screening infrastructure. It is likely that if the paradigm of screening for celiac disease isdeveloped, it will provide an infrastructure that would affect and lower the threshold for publichealth intervention for a series of disorders including type 1A diabetes and Addison’s disease.In many ways, celiac disease is an ideal HLA-associated disorder for public healthscreening consideration. The disease is common (approximately 1 percent of children inColorado, 6 percent of children with type 1A diabetes, 16 percent of children who are DQ2homozygous with type 1A diabetes). Specific HLA class II alleles (DQ2 or DQ8) are present inapproximately 95 percent of affected individuals. The disease is asymptomatic and thus notdiagnosed in the majority of children. The transglutaminase autoantibody assay is inexpensive,sensitive, and specific, and small-bowel biopsy confirms diagnosis. There is an effectivetherapy (gluten-free diet). Major caveats include (1) whether the bulk of asymptomaticindividuals with celiac disease would have morbidity/mortality, and whether early diagnosisand treatment are useful in preventing such morbidity/mortality; (2) only a subset of thoseidentified with HLA-determined genetic risk will develop transglutaminase autoantibodies andbiopsy-confirmed celiac disease (0.3 percent with no DR3/DQ2 allele develop persistenttransglutaminase autoantibody by age 7 versus approximately 3 percent for those withDR3/DQ2); and (3) public health infrastructure is not in place for screening diseases thatmanifest by the expression of autoantibodies followed by diagnosed pathology in infancy,childhood, and adulthood.37
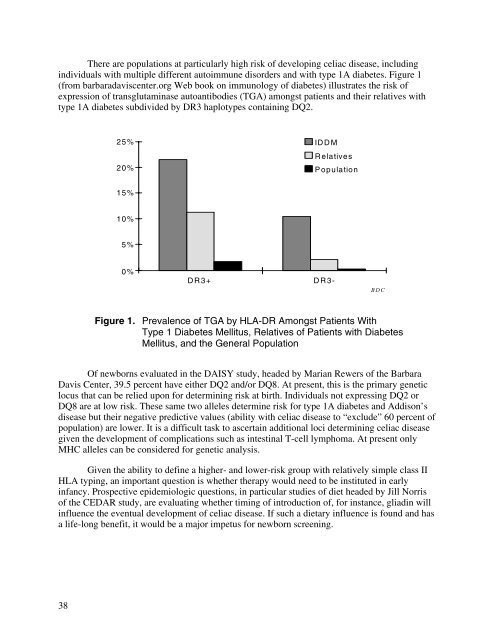
There are populations at particularly high risk of developing celiac disease, includingindividuals with multiple different autoimmune disorders and with type 1A diabetes. Figure 1(from barbaradaviscenter.org Web book on immunology of diabetes) illustrates the risk ofexpression of transglutaminase autoantibodies (TGA) amongst patients and their relatives withtype 1A diabetes subdivided by DR3 haplotypes containing DQ2.25% ID D M20%RelativesP opulation15%10%5%0%DR3+DR3BDCFigure 1. Prevalence of TGA by HLA-DR Amongst Patients WithType 1 Diabetes Mellitus, Relatives of Patients with DiabetesMellitus, and the General PopulationOf newborns evaluated in the DAISY study, headed by Marian Rewers of the BarbaraDavis Center, 39.5 percent have either DQ2 and/or DQ8. At present, this is the primary geneticlocus that can be relied upon for determining risk at birth. Individuals not expressing DQ2 orDQ8 are at low risk. These same two alleles determine risk for type 1A diabetes and Addison’sdisease but their negative predictive values (ability with celiac disease to “exclude” 60 percent ofpopulation) are lower. It is a difficult task to ascertain additional loci determining celiac diseasegiven the development of complications such as intestinal T-cell lymphoma. At present onlyMHC alleles can be considered for genetic analysis.Given the ability to define a higher- and lower-risk group with relatively simple class IIHLA typing, an important question is whether therapy would need to be instituted in earlyinfancy. Prospective epidemiologic questions, in particular studies of diet headed by Jill Norrisof the CEDAR study, are evaluating whether timing of introduction of, for instance, gliadin willinfluence the eventual development of celiac disease. If such a dietary influence is found and hasa life-long benefit, it would be a major impetus for newborn screening.38
- Page 1 and 2: NIH Consensus Development Conferenc
- Page 3 and 4: III. What Are the Manifestations an
- Page 5 and 6: • What is the management of celia
- Page 7 and 8: Monday, June 28, 2004 (continued)I.
- Page 9 and 10: Monday, June 28, 2004 (continued)II
- Page 11 and 12: Wednesday, June 30, 2004 (continued
- Page 13 and 14: Lisa H. RichardsonConsumer Represen
- Page 15 and 16: Ciaran P. Kelly, M.D.Director, Celi
- Page 17 and 18: Van S. Hubbard, M.D., Ph.D.Director
- Page 19 and 20: AbstractsThe following are abstract
- Page 21 and 22: susceptibility (e.g., DR17 homozygo
- Page 23 and 24: The Pathology of Celiac DiseaseDavi
- Page 25 and 26: In this regard, the transport pathw
- Page 27 and 28: for the IgG-based test, while speci
- Page 29 and 30: 15. de Lecea A, Ribes-Koninckx C, P
- Page 31 and 32: Clinical Algorithm in Celiac Diseas
- Page 33: Considera diagnosisof celiac diseas
- Page 37 and 38: Serological Testing for Celiac Dise
- Page 39 and 40: Estimates of the sensitivity of the
- Page 41 and 42: the risk and severity of CD may als
- Page 43 and 44: What Are the Prevalence and Inciden
- Page 45 and 46: ascribed to excess menstrual loss.
- Page 47 and 48: ReferencesFamily History of Celiac
- Page 49 and 50: Carroccio A, Iannitto E, Cavataio F
- Page 51 and 52: identified by surveys or through so
- Page 53 and 54: Clinical Presentation of Celiac Dis
- Page 55 and 56: een widely reported. The question r
- Page 57 and 58: The Many Faces of Celiac Disease: C
- Page 59 and 60: References1. Green PH, Jabri B. Coe
- Page 61 and 62: Association of Celiac Disease and G
- Page 63 and 64: Does the Gluten-Free Diet Protect F
- Page 65 and 66: Skin Manifestations of Celiac Disea
- Page 67 and 68: allow a better understanding of the
- Page 69 and 70: 4. Henriksson KG, Hallert C, Walan
- Page 71 and 72: characterized, clinically-identifie
- Page 73 and 74: 10. Hoffenberg EJ, Emery LM, Barrig
- Page 75 and 76: ataxia, epilepsy with posterior cer
- Page 77 and 78: Consequences of Testing for Celiac
- Page 79 and 80: Osteoporosis/FracturesThere were 11
- Page 81 and 82: Dietary Guidelines for Celiac Disea
- Page 85 and 86:
21. Thompson T. Thiamin, riboflavin
- Page 88 and 89:
In order to effectively counsel ind
- Page 90 and 91:
9. Hallert C, Granno C, Hulten S, M
- Page 92 and 93:
adhered to the GFD after more than
- Page 94 and 95:
Patient education, close supervisio
- Page 96:
26. Mustalahti K, Lohiniemi S, Laip