using the ideal gas equation date
using the ideal gas equation date
using the ideal gas equation date
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
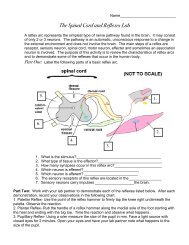
CHEMISTRY/EXPERIMENTUSING THE IDEAL GAS EQUATIONNAME ___________________________________DATE ______________________PERIOD _____In this experiment a sample of butane <strong>gas</strong> will be collected from a commercial lighter by <strong>the</strong> waterdisplacement method. After correcting <strong>the</strong> data for a dry sample of <strong>the</strong> <strong>gas</strong>, <strong>the</strong> molar mass of butane canbe calculated by rearranging <strong>the</strong> <strong>ideal</strong> <strong>gas</strong> <strong>equation</strong> from PV = nRT.PROCEDURE:1. Record <strong>the</strong> room temperature, in degrees Celsius.2. Obtain a bucket with room temperature water.FLAME FROM LIGHTER = ZERO ON LAB3. Place a lighter under water in <strong>the</strong> bucket. Remove <strong>the</strong> lighter, shake off <strong>the</strong> water, and dry <strong>the</strong> outsidewith a tissue. Determine <strong>the</strong> mass of <strong>the</strong> lighter to <strong>the</strong> nearest 0.01 gram. Record.4. Completely fill a 100 mL graduated cylinder with water and invert in <strong>the</strong> bucket. Be careful not to let anyair into <strong>the</strong> cylinder.5. Release <strong>the</strong> butane <strong>gas</strong> from <strong>the</strong> lighter by pressing <strong>the</strong> small lever near <strong>the</strong> flint wheel. Release <strong>the</strong> <strong>gas</strong>under water being careful that all of <strong>the</strong> butane is collected in <strong>the</strong> graduated cylinder by waterdisplacement. Release enough <strong>gas</strong> to fill <strong>the</strong> graduated cylinder between 80-90 mL.6. Allow <strong>the</strong> butane to reach room temperature (about 5 minutes). Adjust <strong>the</strong> level of <strong>the</strong> water inside andoutside <strong>the</strong> graduate until <strong>the</strong>y are <strong>the</strong> same by raising or lowering <strong>the</strong> cylinder in <strong>the</strong> bucket. Thepressure from <strong>the</strong> <strong>gas</strong> is now equalized with atmospheric pressure.7. Read <strong>the</strong> volume of <strong>the</strong> <strong>gas</strong> collected <strong>using</strong> <strong>the</strong> cylinder's graduations (to <strong>the</strong> nearest 0.5 mL). Record.8. Remove <strong>the</strong> lighter from <strong>the</strong> bucket. Shake off any excess water, and dry with a tissue.Measure and record <strong>the</strong> mass of <strong>the</strong> lighter.PURPOSE: The purpose of this experiment is to determine <strong>the</strong> molar mass of butane <strong>gas</strong> <strong>using</strong> <strong>the</strong> <strong>ideal</strong> <strong>gas</strong><strong>equation</strong> and to compare <strong>the</strong> experimental value with <strong>the</strong> <strong>the</strong>oretical value from <strong>the</strong> periodic table.DATA:Temp(room) °C KPressure(room) 758 mmHg kPaMass lighter(before)Mass lighter(after)gramsgramsVolume butane mL LVapor pressure water (P H2O ) 2.2 kPaANALYSIS/CALCULATIONS: (show calculations with complete units)1. Convert <strong>the</strong> room temperature from °C to K and add to <strong>the</strong> table above.2. Convert atmospheric pressure from mm of Hg to kPa and add to <strong>the</strong> table above.
3. Convert volume in mL to L and add to <strong>the</strong> table above.4. Determine <strong>the</strong> mass (in grams) of butane released from <strong>the</strong> lighter.5. Determine <strong>the</strong> partial pressure of dry butane <strong>gas</strong>. (P butane = P room - P H2 O ).6. Use <strong>the</strong> <strong>ideal</strong> <strong>gas</strong> <strong>equation</strong> to determine <strong>the</strong> moles of butane released.PV =nRT where R = 8.31 L kPamol K(Solve for n and use partial pressure of butane from #5 as pressure.)7. Use your experimental data (Do not use <strong>the</strong> periodic table.) to determine <strong>the</strong> molar mass of butane.(Remember molar mass = g/mol.)CONCLUSION/QUESTIONS:1. Butane has <strong>the</strong> formula C4H10. Determine <strong>the</strong> <strong>the</strong>oretical molar mass of butane.(Now use <strong>the</strong> values from <strong>the</strong> periodic table.)2. Calculate <strong>the</strong> % Error between your experimental value and <strong>the</strong> actual (<strong>the</strong>oretical) molar mass ofbutane.3. Can <strong>the</strong> same experimental techniques be used to determine <strong>the</strong> molar mass of all <strong>gas</strong>es? Explain.(Hint: Butane is insoluble in water. What if <strong>the</strong> <strong>gas</strong> were soluble in water?)