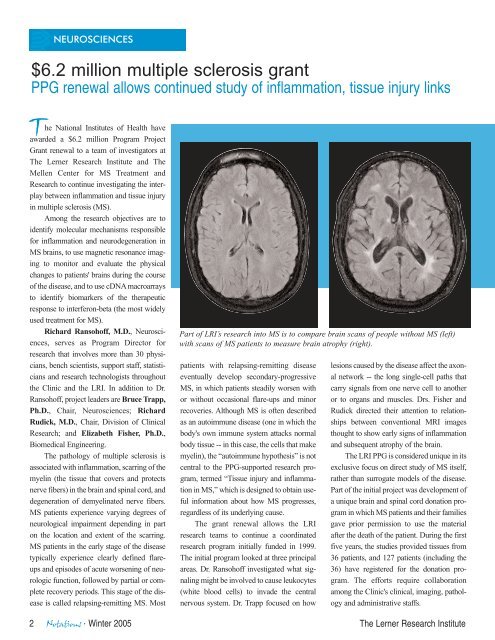
NEUROSCIENCES$6.2 million multiple sclerosis grantPPG renewal allows continued study of inflammation, tissue injury links<strong>The</strong> National <strong>Institute</strong>s of Health haveawarded a $6.2 million Program ProjectGrant renewal to a team of investigators at<strong>The</strong> <strong>Lerner</strong> <strong>Research</strong> <strong>Institute</strong> and <strong>The</strong>Mellen Center for MS Treatment and<strong>Research</strong> to continue investigating the interplaybetween inflammation and tissue injuryin multiple sclerosis (MS).Among the research objectives are toidentify molecular mechanisms responsiblefor inflammation and neurodegeneration inMS brains, to use magnetic resonance imagingto monitor and evaluate the physicalchanges to patients' brains during the courseof the disease, and to use cDNA macroarraysto identify biomarkers of the therapeuticresponse to interferon-beta (the most widelyused treatment for MS).Richard Ransohoff, M.D., Neurosciences,serves as Program Director forresearch that involves more than 30 physicians,bench scientists, support staff, statisticiansand research technologists throughoutthe <strong>Clinic</strong> and the LRI. In addition to Dr.Ransohoff, project leaders are Bruce Trapp,Ph.D., Chair, Neurosciences; RichardRudick, M.D., Chair, Division of <strong>Clinic</strong>al<strong>Research</strong>; and Elizabeth Fisher, Ph.D.,Biomedical Engineering.<strong>The</strong> pathology of multiple sclerosis isassociated with inflammation, scarring of themyelin (the tissue that covers and protectsnerve fibers) in the brain and spinal cord, anddegeneration of demyelinated nerve fibers.MS patients experience varying degrees ofneurological impairment depending in parton the location and extent of the scarring.MS patients in the early stage of the diseasetypically experience clearly defined flareupsand episodes of acute worsening of neurologicfunction, followed by partial or completerecovery periods. This stage of the diseaseis called relapsing-remitting MS. MostPart of LRI’s research into MS is to compare brain scans of people without MS (left)with scans of MS patients to measure brain atrophy (right).patients with relapsing-remitting diseaseeventually develop secondary-progressiveMS, in which patients steadily worsen withor without occasional flare-ups and minorrecoveries. Although MS is often describedas an autoimmune disease (one in which thebody's own immune system attacks normalbody tissue -- in this case, the cells that makemyelin), the “autoimmune hypothesis” is notcentral to the PPG-supported research program,termed “Tissue injury and inflammationin MS,” which is designed to obtain usefulinformation about how MS progresses,regardless of its underlying cause.<strong>The</strong> grant renewal allows the LRIresearch teams to continue a coordinatedresearch program initially funded in 1999.<strong>The</strong> initial program looked at three principalareas. Dr. Ransohoff investigated what signalingmight be involved to cause leukocytes(white blood cells) to invade the centralnervous system. Dr. Trapp focused on howlesions caused by the disease affect the axonalnetwork -- the long single-cell paths thatcarry signals from one nerve cell to anotheror to organs and muscles. Drs. Fisher andRudick directed their attention to relationshipsbetween conventional MRI imagesthought to show early signs of inflammationand subsequent atrophy of the brain.<strong>The</strong> LRI PPG is considered unique in itsexclusive focus on direct study of MS itself,rather than surrogate models of the disease.Part of the initial project was development ofa unique brain and spinal cord donation programin which MS patients and their familiesgave prior permission to use the materialafter the death of the patient. During the firstfive years, the studies provided tissues from36 patients, and 127 patients (including the36) have registered for the donation program.<strong>The</strong> efforts require collaborationamong the <strong>Clinic</strong>'s clinical, imaging, pathologyand administrative staffs.2 <strong>Notations</strong> · Winter 2005 <strong>The</strong> <strong>Lerner</strong> <strong>Research</strong> <strong>Institute</strong>
“I'm proud we study the human disease,”said Dr. Ransohoff. “It's the hardest thing todo (compared with studying models), but inessence it's the only thing that matters.”<strong>The</strong> grant renewal supportsfour primary projects:Chemokines and chemokine receptorsDr. Ransohoff's team has the overall objectiveto study chemokines, peptides that governthe migration and activation profiles ofleukocytes -- the major component of theinflammation that subsequently damages thetissues. <strong>The</strong> goal is to identify more rationaland effective treatment of inflammation,especially during the early stages of the diseasewhen inflammation seems to be moremanageable.Axonal pathologyDr. Trapp's previous project had two objectives:to determine the extent and nature ofthe disease and degeneration of the axonalpathways, and to study oligodendrocyte progenitorcells -- the cells of the central nervoussystem that are essential to the myelinsheath -- in the brains and lesions of MSpatients. <strong>The</strong> renewal allows him to investigatein greater detail the molecular mechanismsthat underlie neurodegeneration in thebrains and spinal cords of MS patients. Hiscontinued research is based on the premisethat reduced axonal energy metabolism inchronically demyelinated axons affects ionicfluxes across the axolemma, culminatingwith increased intraxonal Ca ++ and axonaldegeneration.“We are now at the point where we areconsidering adding neuroprotective therapiesto our arsenal of treatments for MS,” Dr.Trapp said. “If we can delay neurodegeneration,we can keep the MS patient out of thewheel chair.”Brain atrophy during the course of MS..Dr. Fisher will use advanced neuroimagingtechniques to study brain atrophy in patientswith relapsing-remitting and secondary progressiveMS. During the first phase of theresearch, a cohort of patients was identifiedand followed longitudinally over the courseABIOMEDICAL ENGINEERING14-year study of multiple sclerosispatients is providing a unique glimpseinto the significance of brain atrophy.Elizabeth Fisher, Ph.D., BiomedicalEngineering, said that for many years thestudy of brain tissue loss due to MS diseaseprocesses was not a high priority. Itwas only recently that the research communityhas started to examine brain atrophyin MS patients.“People knew brain atrophy happens,but many thought of the tissue loss assomething that only happens very late inthe disease in a subset of patients, perhapsas a consequence of repeated focal tissuedamage,” she said. “<strong>The</strong>re were also technologicallimitations at the time to preciselymeasuring the very slow process ofbrain atrophy.”Through <strong>The</strong> <strong>Lerner</strong> <strong>Research</strong><strong>Institute</strong>'s long-term dedication to MSresearch and landmark software developedby Dr. Fisher that provides accurate measurementsof minute changes in brain tissueeven early on in the disease, researchers havetracked brain atrophy -- an effort that willcontinue for another five years thanks to therenewal of a $6.2 million NIH grant.“Five years is a long time for an MSstudy,” Dr. Fisher said. “So a study thatwill eventually be 19 years is truly unique.Many people live with this disease for along time. It's a long-lasting and chronicillness, and this provides an opportunityto study how the disease changes andof four years. Analysis of MRIs from thesepatients has revealed that the pathologicprocesses that lead to overall brain tissue losscontinued on Page 13Brain atrophyGrant offers unique 19-year look at tissue lossaffects brain tissue over a long period oftime.”<strong>The</strong> precision of the imaging is essentialto the importance of the study.“On average, MS patients lose 0.5%to 1% of their brain tissue each year, soyou have to be able to detect very smallamounts of loss with a great deal of accuracyif you're going to monitor the benefitsof medications or other therapies,” Dr.Fisher said.About half of the 60 patients in thecurrent study were originally part of atwo-year trial started in 1990 to examinethe effects of a medication that was eventuallyapproved for use in treating MSpatients. With developments in technologyand Dr. Fisher's software, LRIresearchers were able to go back and reexaminethe images taken in 1990 -- andlocate and re-test the patients who werepart of the original clinical study.When the latest research is completedin five years, the LRI will have 19 yearsworth of images and quantitative informationon brain atrophy, in addition to clinicalfollow-up data on disability.“It's definitely the longest time to follow(brain atrophy) with a standardizedimaging protocol,” Dr. Fisher said.“Understanding atrophy is important.We're trying to determine what causes itand why it progresses faster in somepatients than in others. It also can be usedto help validate new treatments.”<strong>The</strong> <strong>Lerner</strong> <strong>Research</strong> <strong>Institute</strong> <strong>Notations</strong> · Winter 2005 3