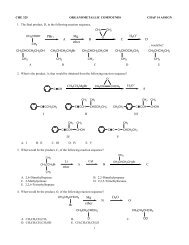
30. Which of the following species have tetrahedral bond angles?NH 3 BF 3+CH 3−CH 3 H 2 O H 3 O +A B C D E FA. A, D and E B. A, E C. A, D, E, F D. only D31. Which of the following species is planar?CH 3+BF 3 H 3 O + +NH 4 H 2 C=CH 2 AlCl 3A B C D E FA. all but C and D B. A and B C. all but C D. B, E, F32. How many constitutional isomers are possible for C 4 H 10 O?A. three B. four C. five D. seven33. In the reaction below, the strongest base isA H 2 O B. NH 2‒C. HO ‒ D. NH 3H 2 O +NH 2‒ HO ‒ + NH 334. A thorough study of organic chemistry requires a basic understanding of acid and base strengths. Which of the followingmolecules most likely has a pKa = 15.5?A. H 2 SO 4 B. HCl C. Methanol D. Methane E. Acetic Acid35. Match the hybridization of carbon with the correct electron domain shapeA. sp 3 X. trigonalB. sp 2 Y. linearC. sp Z. tetrahedralA. AX, BY, CZ B. AZ, BX, CY C. AY, BZ, CX D. AX, BZ, CY36. Which is the shortest of the carbon-carbon single bonds indicated by arrows in the following compounds?CH 2 H 2 CH 3 C CH 3 H 3 C C CH H 3 C CH HC C C CH HC C CHA B C D E37. Taxol (see structure below) is a potent chemotherapeutic agent (isolated from the Pacific Yew tree) which is especiallyeffective against ovarian cancer. Which of the following functional groups is not contained in taxol?A. KetoneB. EsterC. AmideD. AmineE. Alcohol6
38. For the simple hydrides, MH n , acidities increase in the order:A. CH 4 , NH 3 , H 2 O , H 2 S , HBr B. HBr , H 2 S , H 2 O , NH 3 , CH 4C. HBr , H 2 O , NH 3 , H 2 S , CH 4 D. NH 3 , H 2 S , CH 4 , H 2 O , HBrE. H 2 S , H 2 O , HBr , NH 3 , CH 439. Which acid-base reaction would not take place as written?A. CH 3 Li + CH 3 CH 2 CH 2 CH 2 NH 2 CH 4 + CH 3 CH 2 CH 2 CH 2 NHLiB. CH 3 CCH + NaOCH 3 CH 3 CCNa + CH 3 OHC. HCCNa + H 2 O HCCH + NaOHD. CH 3 OH + NaNH 2 CH 3 ONa + NH 3E. CH 3 CO 2 H + CH 3 ONa CH 3 CO 2 Na + CH 3 OH40. A group of acids arranged in order of decreasing acidity is:HNO 3 > CH 3 COOH > C 6 H 5 OH > H 2 O > HCCHWhat is the arrangement of the conjugate bases of these compounds in decreasing order of basicity?A. NO 3 > CH 3 COO > C 6 H 5 O > OH > HCC B. CH 3 COO > C 6 H 5 O > NO 3 > OH > HCC C. C 6 H 5 O > NO 3 > HCC > OH > CH 3 COO D. HCC > OH > C 6 H 5 O > CH 3 COO > NO 3E. No prediction of relative base strength is possible.NAME__________________________________________DATE___________________ANSWER SHEET<strong>CHE</strong> <strong>275</strong> – <strong>CHAP</strong> 1 ASSIGN1. _________________ 11. _________________ 21. _________________ 31. _________________2. _________________ 12. _________________ 22. _________________ 32. _________________3. _________________ 13. _________________ 23. _________________ 33. _________________4. _________________ 14. _________________ 24. _________________ 34. _________________5. _________________ 15. _________________ 25. _________________ 35. _________________6. _________________ 16. _________________ 26. _________________ 36. _________________7. _________________ 17. _________________ 27. _________________ 37. _________________8. _________________ 18. _________________ 28. _________________ 38. _________________9. _________________ 19. _________________ 29. _________________ 39. _________________10. _________________ 20. _________________ 30. _________________ 40. _________________SS I 20137