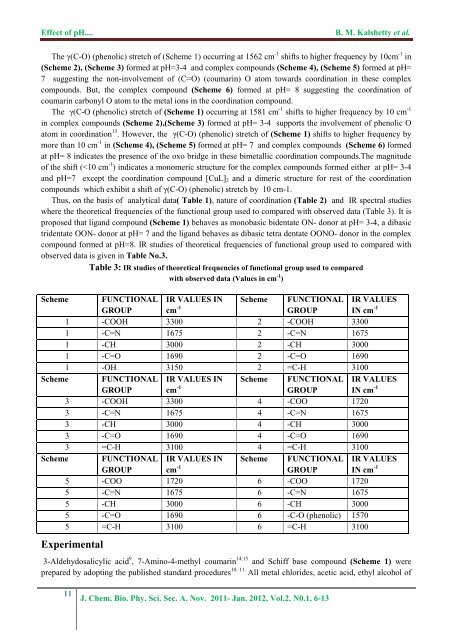
<str<strong>on</strong>g>Effect</str<strong>on</strong>g> <str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>pH</str<strong>on</strong>g>....B. M. Kalshetty et al.The γ(C-O) (phenolic) stretch <str<strong>on</strong>g>of</str<strong>on</strong>g> (Scheme 1) occurring at 1562 cm -1 shifts to higher frequency by 10cm -1 in(Scheme 2), (Scheme 3) formed at <str<strong>on</strong>g>pH</str<strong>on</strong>g>=3-4 and <strong>complex</strong> <strong>compounds</strong> (Scheme 4), (Scheme 5) formed at <str<strong>on</strong>g>pH</str<strong>on</strong>g>=7 suggesting <strong>the</strong> n<strong>on</strong>-involvement <str<strong>on</strong>g>of</str<strong>on</strong>g> (C=O) (coumarin) O atom towards coordinati<strong>on</strong> in <strong>the</strong>se <strong>complex</strong><strong>compounds</strong>. But, <strong>the</strong> <strong>complex</strong> compound (Scheme 6) formed at <str<strong>on</strong>g>pH</str<strong>on</strong>g>= 8 suggesting <strong>the</strong> coordinati<strong>on</strong> <str<strong>on</strong>g>of</str<strong>on</strong>g>coumarin carb<strong>on</strong>yl O atom to <strong>the</strong> metal i<strong>on</strong>s in <strong>the</strong> coordinati<strong>on</strong> compound.The γ(C-O (phenolic) stretch <str<strong>on</strong>g>of</str<strong>on</strong>g> (Scheme 1) occurring at 1581 cm -1 shifts to higher frequency by 10 cm -1in <strong>complex</strong> <strong>compounds</strong> (Scheme 2),(Scheme 3) formed at <str<strong>on</strong>g>pH</str<strong>on</strong>g>= 3-4 supports <strong>the</strong> involvement <str<strong>on</strong>g>of</str<strong>on</strong>g> phenolic Oatom in coordinati<strong>on</strong> 13 . However, <strong>the</strong> γ(C-O) (phenolic) stretch <str<strong>on</strong>g>of</str<strong>on</strong>g> (Scheme 1) shifts to higher frequency bymore than 10 cm -1 in (Scheme 4), (Scheme 5) formed at <str<strong>on</strong>g>pH</str<strong>on</strong>g>= 7 and <strong>complex</strong> <strong>compounds</strong> (Scheme 6) formedat <str<strong>on</strong>g>pH</str<strong>on</strong>g>= 8 indicates <strong>the</strong> presence <str<strong>on</strong>g>of</str<strong>on</strong>g> <strong>the</strong> oxo bridge in <strong>the</strong>se bimetallic coordinati<strong>on</strong> <strong>compounds</strong>.The magnitude<str<strong>on</strong>g>of</str<strong>on</strong>g> <strong>the</strong> shift (
<str<strong>on</strong>g>Effect</str<strong>on</strong>g> <str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>pH</str<strong>on</strong>g>....B. M. Kalshetty et al.analytical R grade were used and <strong>the</strong> sodium hydroxide soluti<strong>on</strong>s were prepared in double distilled water andit is standardized by <strong>the</strong> usual method.(i). Syn<strong>the</strong>sis <str<strong>on</strong>g>of</str<strong>on</strong>g> Ligand compound LH 2 (Scheme 1) and metal <strong>complex</strong>es: The ligand (LH 2 ) wasprepared by <strong>the</strong> method reported by Nag et al 11 . The metal <strong>complex</strong>es [M(LH 2 )] where M= Cu(II),Zn(II),Cd(II) ,[M(LH) 2 (H 2 O) 2 ] where M= Co(II),Ni(II) , [CuL] 2, [ML(EtOH)] where M= Zn(II), Cd(II) and[ML(EtOH)] 2 where M= Cu(II), Zn(II), Ni(II), Co(II), Cd(II) where prepared by following <strong>the</strong> methodreported by Kumar et al 16 .(ii).Syn<strong>the</strong>sis <str<strong>on</strong>g>of</str<strong>on</strong>g> [M(LH) 2 ] M= Cu(II), Cd(II) (Scheme 2) and [M(LH) 2 (H 2 O) 2 ] M= Co(II),Ni(II) (Scheme 3): Ligand LH 2 (2.10g) was dissolved in aqueous NaOH soluti<strong>on</strong> (0.5 g in 10 ml), yellowcoloured soluti<strong>on</strong> was obtained. After <strong>the</strong>n 30-50 ml <str<strong>on</strong>g>of</str<strong>on</strong>g> an aqueous soluti<strong>on</strong> <str<strong>on</strong>g>of</str<strong>on</strong>g> metal chloride was added. Themixture was stirred magnetically for 10 minutes. Glacial acetic acid or hydrochloric acid (1:1) was <strong>the</strong>n addeddrop wise to above soluti<strong>on</strong> till <str<strong>on</strong>g>pH</str<strong>on</strong>g> <str<strong>on</strong>g>of</str<strong>on</strong>g> soluti<strong>on</strong> becomes 3-4. The resulting turbid mixture was refluxed for twohours. The coloured solids formed <strong>on</strong> cooling, filtered, washed and recrystalized from water followed bywashing <strong>with</strong> e<strong>the</strong>r. M.P. 250 o C. Yield =40%.(iii).Syn<strong>the</strong>sis <str<strong>on</strong>g>of</str<strong>on</strong>g> [CuL] 2 (Scheme 4):A ethanol soluti<strong>on</strong> (30 ml) <str<strong>on</strong>g>of</str<strong>on</strong>g> LH 2 (1.05g) was added to ethyl alcoholsoluti<strong>on</strong> (35ml) <str<strong>on</strong>g>of</str<strong>on</strong>g> appropriate metal chloride and <strong>the</strong> mixture was heated under reflux for 3 hours <strong>on</strong> a waterbath and <strong>the</strong>n cooled to room temperature. The solid product formed were filtered, washed <strong>with</strong> andrecrystallized from ethyl alcohol followed by washing <strong>with</strong> e<strong>the</strong>r. M.P. 248 o C, yield= 60%.(iv).Syn<strong>the</strong>sis <str<strong>on</strong>g>of</str<strong>on</strong>g> [ML(EtOH)] 2 M= Cu(II), Zn(II), Ni(II),Co(II),Cd(II) (Scheme 5): A ethylalcohol soluti<strong>on</strong> (30ml) <str<strong>on</strong>g>of</str<strong>on</strong>g> LH 2 (1.05g) was added to a EtOH soluti<strong>on</strong> (35ml) <str<strong>on</strong>g>of</str<strong>on</strong>g> appropriate metal chloridesand <strong>the</strong> mixture was stirred magnetically for 10 minutes. A 10% ethyl alcohol soluti<strong>on</strong> <str<strong>on</strong>g>of</str<strong>on</strong>g> EtONa was <strong>the</strong>nadded drop wise to <strong>the</strong> above soluti<strong>on</strong> till <strong>the</strong> <str<strong>on</strong>g>pH</str<strong>on</strong>g> <str<strong>on</strong>g>of</str<strong>on</strong>g> <strong>the</strong> soluti<strong>on</strong> becomes 8. The resulting mixture wasrefluxed for 3 hours <strong>on</strong> a water bath, coloured solids formed <strong>on</strong> cooling, filtered, washed <strong>with</strong> EtOH followedby washing <strong>with</strong> e<strong>the</strong>r. M.P. 86 o C, yield=70%.CONCLUSIONSThe different types <str<strong>on</strong>g>of</str<strong>on</strong>g> <strong>complex</strong> <strong>compounds</strong> <str<strong>on</strong>g>of</str<strong>on</strong>g> same ligand <strong>with</strong> metal i<strong>on</strong>s have been obtained by varying<str<strong>on</strong>g>pH</str<strong>on</strong>g> and solvent during <strong>the</strong>ir syn<strong>the</strong>sis. The basicity and denticity <str<strong>on</strong>g>of</str<strong>on</strong>g> <strong>the</strong> <strong>Schiff</strong> base (Scheme 1) derived from3-Aldehydosalicylic acid and 7-Amino-4-methyl coumarin changes c<strong>on</strong>siderably <strong>with</strong> <strong>the</strong> role <str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>pH</str<strong>on</strong>g> from 3-8.The basicity and denticity <str<strong>on</strong>g>of</str<strong>on</strong>g> ligand increase <strong>with</strong> <strong>the</strong> increase in <str<strong>on</strong>g>pH</str<strong>on</strong>g>. The ligand at <str<strong>on</strong>g>pH</str<strong>on</strong>g> 3-4 acts as m<strong>on</strong>obasicbidentate ON-d<strong>on</strong>or ligand forms m<strong>on</strong>ometallic <strong>complex</strong> <strong>compounds</strong> [M(LH) 2 ] where M= Cu(II),Zn(II),Cd(II) (Scheme 2) and [M(LH) 2 (H 2 O) 2 ] where M= Co(II), Ni(II) (Scheme 3). At <strong>the</strong> <str<strong>on</strong>g>pH</str<strong>on</strong>g>= 7, <strong>the</strong> ligand acts asa dibasic tridentate OON-d<strong>on</strong>or ligand forming <strong>the</strong> m<strong>on</strong>ometallic <strong>complex</strong> compound <str<strong>on</strong>g>of</str<strong>on</strong>g> <strong>the</strong> type [ML(EtOH)] where M= Zn (II), Cd (II), (Scheme 5) and at <strong>the</strong> same <str<strong>on</strong>g>pH</str<strong>on</strong>g> = 7 <strong>the</strong> ligand formed dimetalic <strong>complex</strong>compound <str<strong>on</strong>g>of</str<strong>on</strong>g> <strong>the</strong> type [(CuL) 2 ] <strong>with</strong> copper metal i<strong>on</strong> (Scheme 4). While at <str<strong>on</strong>g>pH</str<strong>on</strong>g>= 8 <strong>the</strong> ligand acts as a dibasictetradentate OONO-d<strong>on</strong>or ligand in EtOH and forms dimetalic compound <str<strong>on</strong>g>of</str<strong>on</strong>g> <strong>the</strong> type[ML(EtOH)]2 where M=Cu(II),Zn(II),Ni(II), Co(II), Cd(II) (Scheme 6).The basicity and denticity c<strong>on</strong>firmed by <strong>the</strong> various methods , <strong>the</strong> IR spectra <str<strong>on</strong>g>of</str<strong>on</strong>g> <strong>the</strong> <strong>Schiff</strong> base (Scheme 1)and o<strong>the</strong>r metal <strong>complex</strong>es (Scheme 2 ) reveals that <strong>the</strong> n<strong>on</strong>-involvement <str<strong>on</strong>g>of</str<strong>on</strong>g> carboxylic atom in coordinati<strong>on</strong>at <str<strong>on</strong>g>pH</str<strong>on</strong>g>=3-4. However, <strong>the</strong> coordinati<strong>on</strong> formed at <str<strong>on</strong>g>pH</str<strong>on</strong>g>=7 and <str<strong>on</strong>g>pH</str<strong>on</strong>g>=8 <strong>the</strong> carboxylic O atom involved in <strong>the</strong>coordinati<strong>on</strong> (Schemes 3 to 6) by exhibiting <strong>the</strong> γ as (COO) and γ s (COO) stretches <str<strong>on</strong>g>of</str<strong>on</strong>g> carboxylate group in <strong>the</strong>ranges: 1560-1650 cm -1 (i.e.,1600cm -1 ) and 1340-1430 cm -1 (i.e.,1400cm -1 ) respectively. The energydifference between γ as (COO) and γ s (COO) is more than 200 cm -1 indicates <strong>the</strong> m<strong>on</strong>odentate nature <str<strong>on</strong>g>of</str<strong>on</strong>g> <strong>the</strong>carboxylate moiety 12 .12J. Chem. Bio. Phy. Sci. Sec. A. Nov. 2011- Jan. 2012, Vol.2, N0.1, 6-13