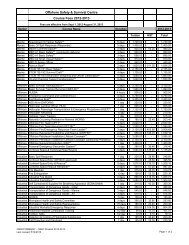
Chemistry 1011
Chemistry 1011
Chemistry 1011
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
Predicting Acid-Base Propertiesof Salts• What will be the effect of the cation on the pH of water?• Will the cation act as a weak acid or will it be a“spectator ion”– If this is a salt of a weak base, the cation will be a weak acid– If this is a salt of a strong base, the cation will be a “spectatorion”– If the cation is a hydrated metal ion, the solution will be aweak acid– If the cation is a group 1 or 2 metal ion, the cation will be a“spectator ion”<strong>Chemistry</strong> <strong>1011</strong> Slot 5 12