impurities in new drug products
impurities in new drug products
impurities in new drug products
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
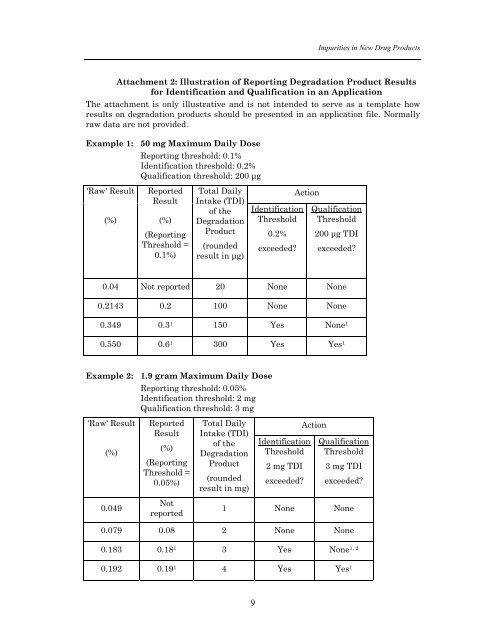
Impurities <strong>in</strong> New Drug ProductsAttachment 2: Illustration of Report<strong>in</strong>g Degradation Product Resultsfor Identification and Qualification <strong>in</strong> an ApplicationThe attachment is only illustrative and is not <strong>in</strong>tended to serve as a template howresults on degradation <strong>products</strong> should be presented <strong>in</strong> an application file. Normallyraw data are not provided.Example 1: 50 mg Maximum Daily DoseReport<strong>in</strong>g threshold: 0.1%Identification threshold: 0.2%Qualification threshold: 200 µg'Raw' Result(%)ReportedResult(%)(Report<strong>in</strong>gThreshold =0.1%)Total DailyIntake (TDI)of theDegradationProduct(roundedresult <strong>in</strong> µg)IdentificationThreshold0.2%exceeded?ActionQualificationThreshold200 µg TDIexceeded?0.04 Not reported 20 None None0.2143 0.2 100 None None0.349 0.3 1 150 Yes None 10.550 0.6 1 300 Yes Yes 1Example 2: 1.9 gram Maximum Daily DoseReport<strong>in</strong>g threshold: 0.05%Identification threshold: 2 mgQualification threshold: 3 mg'Raw' Result(%)ReportedResult(%)(Report<strong>in</strong>gThreshold =0.05%)Total DailyIntake (TDI)of theDegradationProduct(roundedresult <strong>in</strong> mg)IdentificationThreshold2 mg TDIexceeded?ActionQualificationThreshold3 mg TDIexceeded?0.049Notreported1 None None0.079 0.08 2 None None0.183 0.18 1 3 Yes None 1, 20.192 0.19 1 4 Yes Yes 19