Investigational
novartisnovartisnovartis
novartisnovartisnovartis
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
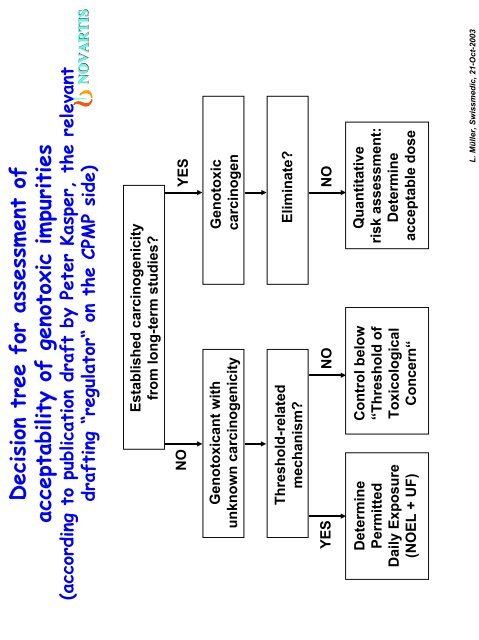
Decision tree for assessment of<br />
acceptability of genotoxic impurities<br />
(according to publication draft by Peter Kasper, the relevant<br />
drafting “regulator“ on the CPMP side)<br />
NOVARTIS<br />
Established carcinogenicity<br />
from long-term studies?<br />
NO<br />
YES<br />
Genotoxicant with<br />
unknown carcinogenicity<br />
Genotoxic<br />
carcinogen<br />
Threshold-related<br />
mechanism?<br />
Eliminate?<br />
YES<br />
NO<br />
NO<br />
Determine<br />
Permitted<br />
Daily Exposure<br />
(NOEL + UF)<br />
Control below<br />
“Threshold of<br />
Toxicological<br />
Concern“<br />
Quantitative<br />
risk assessment:<br />
Determine<br />
acceptable dose<br />
L. Müller, Swissmedic, 21-Oct-2003