MGIT TM Procedure Manual - Foundation for Innovative New ...
MGIT TM Procedure Manual - Foundation for Innovative New ...
MGIT TM Procedure Manual - Foundation for Innovative New ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
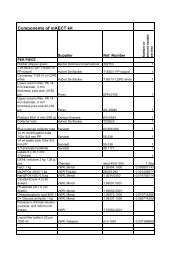
Drug<br />
Concentration of drug<br />
after reconstitution *<br />
Volume added to<br />
<strong>MGIT</strong> tube<br />
Section III: Drug Susceptibility Testing<br />
STR 83 µl/ml 100 ml 1.0 µl/ml<br />
INH 8.3 µl/ml 100 ml 0.1 µl/ml<br />
RIF 83 µl/ml 100 ml 1.0 µl/ml<br />
EMB 415 µl/ml 100 ml 5.0 µl/ml<br />
Final concentration in<br />
<strong>MGIT</strong> tube<br />
* The drugs should be reconstituted using 4 ml sterile deionized or distilled water to<br />
achieve the indicated concentrations.<br />
Calculations of the dilution factor <strong>for</strong> <strong>MGIT</strong> medium: 7.0 ml of medium + 0.8 ml of SIRE<br />
Supplement + 0.5 ml of inoculum = 8.3 ml. Addition of 0.1 ml of the drug solution in 8.3 ml<br />
of the medium = 1:83 dilution.<br />
• Aseptically add 0.5 ml of the well-mixed culture suspension (inoculum) into each of<br />
the drug containing tubes using a pipette. Do not add to the control.<br />
• For the control, first dilute the test culture suspension 1:100 by adding 0.1 ml of the test<br />
culture suspension to 10.0 ml of sterile saline. Mix well by inverting the tube 5-6<br />
times. Use this diluted suspension to add 0.5 ml into the growth control tube.<br />
• Tighten the caps and mix the inoculated broth well by gently inverting the tube<br />
several times.<br />
• Susceptibility test “Set Carriers” are provided in different numbers of drug<br />
combinations. For a routine SIRE test with critical concentration, a Set Carrier of<br />
five tubes is used (refer to BACTEC <strong>MGIT</strong> 960 User’s <strong>Manual</strong> <strong>for</strong> details). Place<br />
labeled tubes in the correct sequence in the set carrier (GC, STR, INH, RIF, EMB).<br />
• Enter the susceptibility set carrier into the BACTEC <strong>MGIT</strong> 960 instrument using the<br />
susceptibility test set entry feature. (Refer to the BACTEC <strong>MGIT</strong> 960 User’s <strong>Manual</strong>,<br />
AST Instructions.) Ensure that the order of the tubes in the AST Set Carrier con<strong>for</strong>ms<br />
to Set Carrier definitions. For example, GC, STR, INH, RIF, EMB <strong>for</strong> the SIRE<br />
standard testing.<br />
• If you need to check purity of the inoculum, streak the test culture suspension<br />
onto a blood agar plate. If blood agar is not available, use chocolate agar or BHI<br />
agar. Incubate at 35 ºC + 1ºC <strong>for</strong> 48 hours and check if there is any growth. If<br />
growth appears, do not set up the susceptibility test. It may be important to establish<br />
the purity of culture be<strong>for</strong>e setting up susceptibility test, particularly if contamination is<br />
suspected.<br />
<strong>MGIT</strong> <strong>TM</strong> <strong>Procedure</strong> <strong>Manual</strong> 45






![Download in English [pdf 2Mb] - Foundation for Innovative New ...](https://img.yumpu.com/49580359/1/184x260/download-in-english-pdf-2mb-foundation-for-innovative-new-.jpg?quality=85)






![New laboratory diagnostic tools for tuberculosis control [.pdf]](https://img.yumpu.com/43339906/1/190x135/new-laboratory-diagnostic-tools-for-tuberculosis-control-pdf.jpg?quality=85)


