Peter Campbell - University of Wollongong
Peter Campbell - University of Wollongong
Peter Campbell - University of Wollongong
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
Contents<br />
Convenor’s Welcome 1<br />
General Information 2<br />
Conference Program 3<br />
Conference Venue and Social Program 13<br />
Pre-Conference Computational<br />
Chemistry Workshop 14<br />
Plenary Speakers 15<br />
Keynote Speakers 20, 43, 89<br />
Biographies and Abstracts 22<br />
Poster Authors 92<br />
Posters 104<br />
Conference Sponsors 155<br />
CONFERENCE ORGANISING<br />
COMMITTEE<br />
Dr Adam Trevitt (<strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>)<br />
A/Pr<strong>of</strong> Jamie Vandenberg (Victor Chang Institute)<br />
Pr<strong>of</strong> Jeff Reimers (<strong>University</strong> <strong>of</strong> Sydney)<br />
Dr Haibo Yu (<strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>)<br />
A/Pr<strong>of</strong> Tim Schmidt (<strong>University</strong> <strong>of</strong> Sydney)<br />
Pr<strong>of</strong> Michelle Coote (Australian National <strong>University</strong>)<br />
Dr Adam Hill (Victor Chang Institute)<br />
Dr Ron Clarke (<strong>University</strong> <strong>of</strong> Sydney)<br />
A/Pr<strong>of</strong> Stephen Blanksby (<strong>University</strong> <strong>of</strong><br />
<strong>Wollongong</strong>)<br />
Dr Meredith Jordan (<strong>University</strong> <strong>of</strong> Sydney)<br />
CONFERENCE MANAGERS<br />
Leishman Associates<br />
113 Harrington Street<br />
Hobart TAS 7000<br />
Phone: 03 6234 7844<br />
Fax: 03 6234 5958<br />
Email: naomi@leishman-associates.com.au<br />
Web: www.leishman-associates.com.au<br />
Convenor’s<br />
welcome<br />
The Organising Committee welcomes<br />
you to the inaugural BioPhysChem 2011,<br />
a joint meeting <strong>of</strong> the RACI Physical<br />
Chemistry Division and the Australian<br />
Society for Biophysics.<br />
This meeting promises to be an exciting<br />
event, bringing together scientists from a<br />
range <strong>of</strong> disciplines to showcase<br />
research developments while providing a<br />
collegial atmosphere for scientific<br />
exchange. We have a stimulating<br />
technical program that boasts an array <strong>of</strong><br />
highly regarded plenary and keynote<br />
speakers, drawn from all across the<br />
world, supported by a relaxed social<br />
program.<br />
A warm thank you must also be<br />
extended to our sponsors and exhibitors<br />
for your support <strong>of</strong> this important event,<br />
your commitment to our Associations<br />
and the chemistry community is greatly<br />
appreciated.<br />
We welcome you and look forward to<br />
your participation in BioPhysChem 2011.<br />
Dr Adam Trevitt<br />
Conference Chair<br />
School <strong>of</strong> Chemistry,<br />
<strong>University</strong> <strong>of</strong> <strong>Wollongong</strong><br />
1
2<br />
General Information<br />
General Information<br />
REGISTRATION DESK<br />
The Registration Desk will be located in the foyer<br />
<strong>of</strong> the McKinnon Building and will be open at the<br />
following times:<br />
Saturday 3 December 10:00am – 5.00pm<br />
Sunday 4 December 8:00am – 5.30pm<br />
Monday 5 December 8:00am – 5:30pm<br />
Tuesday 6 December 8:00am – 5:00pm<br />
ACCOMMODATION<br />
If you have any queries relating to your<br />
accommodation booking, first please see the staff<br />
at your hotel. Your credit card details have been<br />
passed onto the hotel to secure your booking. If<br />
you have arrived 24 hours later than your indicated<br />
arrival day you may find that you have been charge<br />
one nights accommodation.<br />
CONFERENCE NAME BADGES<br />
All delegates and exhibitors will be provided with a<br />
name badge, please wear your name badge at all<br />
times as it will be your entry into all Sessions and<br />
all Social Functions.<br />
DISCLAIMER<br />
The BioPhysChem 2011 Conference reserves the<br />
right to amend or alter any advertised details<br />
relating to dates, program and speakers if<br />
necessary, without notice, as a result <strong>of</strong><br />
circumstances beyond their control. All attempts<br />
have been made to keep any changes to an<br />
absolute minimum.<br />
ENTRY TO SOCIAL EVENTS<br />
Entry to social events will not require a ticket,<br />
attendees and additional guests will appear on a<br />
guest list.<br />
MOBILE PHONES<br />
As a courtesy to other delegates, please ensure<br />
that all mobile phones are turned <strong>of</strong>f or in a silent<br />
mode during all sessions and social functions.<br />
SPEAKERS<br />
Speakers will be asked to bring their presentations<br />
with them on a CD or USB stick, then load their<br />
presentations onto the computer in the<br />
corresponding theatre. This must be done AT<br />
LEAST by the break prior to your presenting time<br />
– this may mean the day before your presentation.<br />
There will be dedicated speakers assistants to<br />
provide help uploading your file. All speakers are<br />
responsible for ensuring their presentation is<br />
uploaded and ready for their session.<br />
Please see the staff at the Registration Desk for<br />
further information.<br />
SPECIAL DIETS<br />
All catering venues have been advised <strong>of</strong> any<br />
special diet preferences you have indicated on<br />
your registration form. Please identify yourself to<br />
venue staff as they come to serve you and they will<br />
be pleased to provide you with all pre-ordered<br />
food. For day catering, there may be a specific<br />
area where special food is brought out, please<br />
check with catering or Conference staff.<br />
WEBSITE<br />
Updated Conference information can be found at<br />
www.biophyschem2011.net.net.au
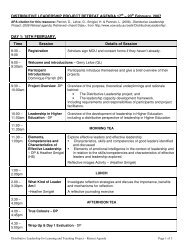
Program<br />
Friday 2 and Saturday 3 December 2011<br />
Friday 2 December 2011 – Pre Conference Workshop<br />
9:00am – 12:00pm Pre Conference Computational Chemistry Workshop<br />
Hyperion Computer Lab - Building 17, Room 105<br />
12:00pm – 12:45pm Lunch<br />
12:45pm – 5:00pm Pre Conference Computational Chemistry Workshop Continues<br />
Saturday 3 December 2011<br />
10:00am Registration Desk Opens McKinnon Building Foyer<br />
9:00am – 1:00pm Pre Conference Computational Chemistry Workshop<br />
Hyperion Computer Lab - Building 17, Room 105<br />
12:00pm – 1:15pm Lunch (workshop delegates only)<br />
1:15pm – 1:30pm Official Conference Opening Remarks Main Theatre<br />
Plenary Session Medalist Lectures Main Theatre<br />
1:30pm – 2:15pm 2010 RACI Physical Chemistry Medallist Chair: Jeffrey Reimers<br />
K.1 Adventures In Free Radical Chemistry: A Computational Approach<br />
Pr<strong>of</strong>essor Leo Radom, <strong>University</strong> <strong>of</strong> Sydney<br />
2:15pm – 3:00pm 2011 Bob Robertson Medal (ASB) Chair: Jamie Vandenberg<br />
K.2 2011 Medal Recipient<br />
3:00pm – 3:45pm 2011 RACI Physical Chemistry Medallist Chair: Adam Trevitt<br />
K.3 A Life in Physical Chemistry: From Fundamentals to Applications<br />
Pr<strong>of</strong>essor Keith King, <strong>University</strong> <strong>of</strong> Adelaide<br />
4:30pm – 6:30pm We l c o m e M i x e r M c K i n n o n B u i l d i n g F o y e r<br />
3
4<br />
Program<br />
Sunday 4 December 2011<br />
Sunday 4 December 2011<br />
8:00am Registration Desk Open McKinnon Building Foyer<br />
9:00am – 9:50am Plenary Session Main Theatre<br />
GFP: Lighting Up Life<br />
Pr<strong>of</strong>essor Martin Chalfie, <strong>University</strong> <strong>of</strong> Columbia<br />
9:50am – 10:30am Morning Refreshments McKinnon Building Foyer<br />
10:30am – 12:00pm Concurrent Session 1<br />
Theatre 2 Chair: Adam Trevitt Main Theatre Chair: Boris Martiniac<br />
10:30am – 10:45am<br />
10:30am – 10:45am<br />
1.1.1 Density Functional Theory Studies 1.2.1 Spatial and spectral super-<br />
<strong>of</strong> High-Oxidation State Palladium resolution – Optical imaging <strong>of</strong><br />
Systems<br />
nanoscopic signalling domains in 4D<br />
Pr<strong>of</strong>essor Brian Yates, <strong>University</strong> <strong>of</strong><br />
Tasmania<br />
Dr David Baddeley, <strong>University</strong> <strong>of</strong> Auckland<br />
10:45am – 11:00am<br />
1.1.2 Interpolating Molecular Potential<br />
Energy and Property Surfaces<br />
Dr Meredith Jordan, <strong>University</strong> <strong>of</strong> Sydney<br />
11:00am – 11:15am<br />
1.1.3 The Relationship Between<br />
Intrinsic Bond Energy and Intrinsic<br />
Radical Stability: Can This be Used to<br />
Test the Untestable?<br />
Pr<strong>of</strong>essor Michelle Coote, Australian<br />
National <strong>University</strong><br />
11:15am – 11:30am<br />
1.1.4 Molecular Oxygen as Energy<br />
Mediator for Photochemical<br />
Upconversion <strong>of</strong> Near-Infrared Light<br />
Dr Burkhard Fückel, The <strong>University</strong> <strong>of</strong><br />
Sydney<br />
11:30am – 11:45am<br />
1.1.5 Time Resolved Fluorescence<br />
Imaging <strong>of</strong> Conjugated Polymer Thin<br />
Films<br />
Dr Xiao-Tao Hao, The <strong>University</strong> <strong>of</strong><br />
Melbourne<br />
11:45am – 12:00pm<br />
1.1.6 Quantifying cooperative<br />
intermolecular interactions for<br />
improved carbon dioxide capture<br />
materials<br />
Dr Joseph Lane, The <strong>University</strong> <strong>of</strong> Waikato<br />
10:45am – 11:00am<br />
1.2.2 BRET based monitoring <strong>of</strong> ligand<br />
binding in the ODR-10 odorant<br />
responsive G-protein coupled receptor<br />
from Caenorhabditis elegans<br />
Dr Helen Dacres, Food Futures Flagship @<br />
CSIRO Ecosystem Sciences<br />
11:00am – 11:15am<br />
1.2.3 Photostable Fluorescent<br />
Nanodiamond Material: Labels for<br />
Biomolecules & FRET<br />
Jana Say, Macquarie <strong>University</strong><br />
11:15am – 11:30am<br />
1.2.4 BODIPY phosphatidylinositol<br />
probes incorporation into the<br />
membrane <strong>of</strong> giant unilamellar vesicles<br />
grown in carbohydrate and<br />
physiological buffer solutions<br />
Dr Pierre Moens, <strong>University</strong> <strong>of</strong> New England<br />
11:30am – 11:45am<br />
1.2.5 Monitoring the B to A<br />
conformation transition <strong>of</strong> DNA in<br />
functional cells using Fourier<br />
transform infrared spectroscopy<br />
Donna Whelan, Monash <strong>University</strong><br />
11:45am – 12:00pm<br />
1.2.6 The Structure <strong>of</strong> Mixed<br />
Lipopolysaccharide/Porin Monolayers<br />
at the Air-Liquid||Interface<br />
Dr Anton Le Brun, Australian Nuclear<br />
Science And Technology Organisation<br />
12:00pm – 1:15pm Lunch McKinnon Building Foyer
Sunday 4 December 2011 - Continued<br />
1:15pm – 3:00pm Concurrent Session 2<br />
Theatre 2 Chair: Tim Schmidt Main Theatre Chair: Haibo Yu<br />
1:15pm – 1:45pm<br />
1:15pm – 1:30pm<br />
2.1.1 Isomerization and Decomposition 2.2.1 Structure and dynamics <strong>of</strong> a<br />
Chemistry <strong>of</strong> C7Hn (n = 5,7) Radicals replisomal macromolecular assembly<br />
Dr Gabriel Da Silva, The <strong>University</strong> <strong>of</strong> Flynn Hill, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong><br />
Melbourne<br />
1:30pm – 1:45pm<br />
2.2.2 Resolving Single Molecule<br />
Fibronectin Interactions with<br />
Conducting Polymer Electrodes using<br />
Atomic Force Microscopy<br />
Dr Michael Higgins, Intelligent Polymer<br />
Research Institute<br />
1:45pm – 2:00pm<br />
2.1.2 Cross-Strand Disulfides - Poised<br />
to Act<br />
Dr Naomi Haworth, Deakin <strong>University</strong><br />
2:00pm - 2:15pm<br />
2.1.3 Computational Design <strong>of</strong><br />
Metal-Based Systems for the<br />
Functionalization <strong>of</strong> Small Molecules<br />
<strong>of</strong> Synthetic Interest<br />
Dr Germaine Cavigliasso, Australian<br />
National <strong>University</strong><br />
2:15pm - 2:30pm<br />
2.1.4 Infrared Spectroscopy: from<br />
Conformers to Clouds<br />
Dr Evan Robertson, La Trobe <strong>University</strong><br />
2:30pm – 2:45pm<br />
2.1.5 Photodetachment <strong>of</strong> Small<br />
Dianions: Adventures in Mass and<br />
Charge<br />
Associate Pr<strong>of</strong>essor Stephen Blanksby,<br />
<strong>University</strong> <strong>of</strong> <strong>Wollongong</strong><br />
Program<br />
Sunday 4 December 2011<br />
2:45pm – 3:00pm<br />
2.1.6 Laser-Based Formation and<br />
Properties <strong>of</strong> Metal Nanoparticles in<br />
Aqueous Solution<br />
Pr<strong>of</strong>essor Mark Buntine, Curtin <strong>University</strong><br />
1:45pm – 2:00pm<br />
2.2.3 Engineering new catalytic<br />
activities in enzymes through<br />
modifying the conformational<br />
landscape: experimental and<br />
theoretical insights<br />
Dr Colin Jackson, Australian National<br />
<strong>University</strong><br />
2:00pm - 2:15pm<br />
2.2.4 Towards ab initio refinement <strong>of</strong><br />
protein X-ray crystal structures:<br />
interpreting and correlating structural<br />
fluctuations<br />
Pr<strong>of</strong>essor Jeffrey Reimers, <strong>University</strong> <strong>of</strong><br />
Sydney<br />
2:15pm - 2:30pm<br />
2.2.5 Density Functional Theory<br />
Calculations <strong>of</strong> Novel Silicon<br />
Nanosheets<br />
Dr Michelle Spencer, La Trobe <strong>University</strong><br />
2:30pm – 2:45pm<br />
2.2.6 Stable Solid Supported<br />
Membranes to probe Membrane-<br />
Protein Interactions<br />
Dr Ingo Koeper, Flinders <strong>University</strong><br />
2:45pm – 3:00pm<br />
2.2.7 Directions and Results <strong>of</strong> OH<br />
Attack on Nucleic Acids: a Theoretical<br />
Study<br />
Ganna Gryn’ova, Australian National<br />
<strong>University</strong><br />
3:00pm – 3:30pm Afternoon Refreshments McKinnon Building Foyer<br />
5
6<br />
Program<br />
Sunday 4 December 2011<br />
Sunday 4 December 2011 - Continued<br />
3:30pm – 4:20pm Plenary Session Main Theatre<br />
Simulating Protein-DNA Switches Chair: Meredith Jordan<br />
Pr<strong>of</strong>essor Tim Clark, The <strong>University</strong> <strong>of</strong> Erlangen-Nuremberg<br />
4:20pm – 4:50pm Keynote Session Main Theatre<br />
K.4 Using Theory to Reconcile Experiment: The Search for Certainty in an<br />
Uncertain World Chair: Meredith Jordan<br />
Pr<strong>of</strong>essor Alan Mark, The <strong>University</strong> <strong>of</strong> Queensland<br />
4:50pm – 5:10pm K.5 Towards a Unified Picture <strong>of</strong> Color and Photisomerization Behavior in<br />
Fluorogenic Monomethine Dyes Chair: Meredith Jordan<br />
Dr Seth Olsen, The <strong>University</strong> <strong>of</strong> Queensland<br />
5:10pm – 5:30pm K.6 The roles <strong>of</strong> membrane deformations and electrostatics in charged<br />
protein-lipid||interactions. Chair: Meredith Jordan<br />
Associate Pr<strong>of</strong>essor Toby Allen, The <strong>University</strong> <strong>of</strong> California, Davis<br />
5:30pm – 7:30pm Poster Session 1 McKinnon Building Foyer
Program<br />
Monday 5 December 2011<br />
Monday 5 December 2011<br />
8:00am Registration Desk Open McKinnon Building Foyer<br />
9:00am – 9:50am Plenary Session Main Theatre<br />
Seeking the Physical Basis <strong>of</strong> Receptor Tyrosine Kinase Signaling<br />
Pr<strong>of</strong>essor Kalina Hristova, John Hopkins <strong>University</strong> Chair: Frances Separovic<br />
9:50am – 10:30am Morning Refreshments McKinnon Building Foyer<br />
10:30am – 12:00pm Concurrent Session 3<br />
Theatre 2 Chair: Irene Yarovsky Main Theatre Chair: Frances Separovic<br />
10:30am – 10:45am<br />
10:30am – 10:50am<br />
3.1.1 Organic Photovoltaic Materials at 3.2.1 Copper Stabilization <strong>of</strong> Aβ42<br />
High Spatial and Temporal Resolution Aggregation in Model Membranes<br />
Associate Pr<strong>of</strong>essor Trevor Smith,<br />
Dr Marc-Antoine Sani, <strong>University</strong> <strong>of</strong><br />
<strong>University</strong> <strong>of</strong> Melbourne<br />
Melbourne<br />
10:45am – 11:00am<br />
3.1.2 Coarse-Grained Modelling <strong>of</strong><br />
Morphology and Energy Transfer in<br />
Conjugated Polymer Nanostructures<br />
Dr David Huang, <strong>University</strong> <strong>of</strong> Adelaide<br />
11:00am – 11:15am<br />
3.1.3 Ultrafast Exciton Dynamics <strong>of</strong><br />
Conjugated Polymer Nanostructures<br />
Dr Tak W Kee, <strong>University</strong> <strong>of</strong> Adelaide<br />
11:15am – 11:30am<br />
3.1.4 Structural, electronic and<br />
transport properties <strong>of</strong> amorphous/<br />
crystalline silicon heterojunctions<br />
Dr Tim Schulze, Helmholtz-Zentrum Berlin<br />
11:30am – 11:45am<br />
3.1.5 A new understanding <strong>of</strong><br />
hybridization in terms <strong>of</strong> bond<br />
strengths and resonance energies<br />
Pr<strong>of</strong>essor Noel Hush, The <strong>University</strong> Of<br />
Sydney<br />
11:45am – 12:00pm<br />
3.1.6 Understanding electron transport<br />
in complex systems|<br />
Pr<strong>of</strong>essor Gemma Solomon, <strong>University</strong> <strong>of</strong><br />
Copenhagen<br />
10:50am – 11:10am<br />
3.2.2 The enigma <strong>of</strong> the CLIC proteins:<br />
ion channels, redox proteins, enzymes,<br />
scaffolding proteins?<br />
Paul Curmi, <strong>University</strong> <strong>of</strong> New South Wales<br />
11:10am – 11:30am<br />
3.2.3 The Advantage <strong>of</strong> Being an<br />
Oligomer: the Trimeric Betaine Carrier<br />
BetP<br />
Pr<strong>of</strong>essor Reinhard Kraemer, <strong>University</strong> Of<br />
Cologne<br />
11:30am – 11:45am<br />
3.2.4 Single-Molecule View <strong>of</strong> the<br />
Dynamics <strong>of</strong> Molecular Machines<br />
Till Boecking, <strong>University</strong> <strong>of</strong> New South<br />
Wales<br />
11:45am – 12:00pm<br />
3.2.5 The Dynamic Stator Stalk <strong>of</strong><br />
A-type ATPase<br />
Alastair Stewart, The Victor Chang Cardiac<br />
Research Institute<br />
12:00pm – 1:15pm Lunch McKinnon Building Foyer<br />
7
8<br />
Program<br />
Monday 5 December 2011<br />
Monday 5 December 2011 - Continued<br />
12:15pm – 1:00pm ASSOCIATION MEETINGS<br />
Theatre 2 RACI PChem AGM Main Theatre ASB Members Meeting<br />
1:15pm – 3:00 pm Concurrent Session 4<br />
Theatre 2 Chair: Tak Kee Main Theatre Chair: Alan Mark<br />
1:15pm – 1:45pm<br />
1:15pm – 1:30pm<br />
4.1.1 Chemistry at the Threshold: 4.2.1 Test <strong>of</strong> a Protein Docking<br />
Unexpected Products, Unusual Algorithm on K+ Channel Binding:<br />
Mechanisms, and||Generally Weird Validation and Analysis<br />
Things that Happen Near the Energetic Dr Po-Chia Chen, <strong>University</strong> <strong>of</strong> Sydney<br />
Threshold for a Reaction.<br />
Pr<strong>of</strong>essor Scott Kable, The <strong>University</strong> <strong>of</strong><br />
Sydney<br />
1:30pm – 1:45pm<br />
4.2.2 Open channel structure <strong>of</strong> MscL<br />
from restrained MD Simulations<br />
Evelyne Deplazes, <strong>University</strong> <strong>of</strong> Western<br />
Australia<br />
11:45pm – 2:00pm<br />
4.1.2 Convergent first principles<br />
quantum dynamics: v MCG and Grow<br />
Dr Terry Frankcombe, Australian National<br />
<strong>University</strong><br />
2:00pm – 2:15pm<br />
4.1.3 Quantum mechanical study <strong>of</strong> the<br />
deep well reaction H+ + D2<br />
Dr Marlies Hankel, <strong>University</strong> <strong>of</strong> Queensland<br />
2:15pm – 2:30pm<br />
4.1.4 Optical Spectroscopy <strong>of</strong><br />
Polycyclic Aromatic Nitrogen<br />
Heterocycle Cations<br />
Dr Viktoras Dryza, <strong>University</strong> <strong>of</strong> Melbourne<br />
2:30pm – 2:45pm<br />
4.1.5 Ab Initio Diabatic Potential<br />
Energy Matrix and Dynamics for<br />
OH(2S) + H2 /D2<br />
Pr<strong>of</strong>essor Michael Collins, Australian<br />
National <strong>University</strong><br />
2:45pm – 3:00pm<br />
4.1.6 G4(MP2)-6X: Accurate and<br />
Affordable Computational Chemistry<br />
Dr Bun Chan, <strong>University</strong> <strong>of</strong> Sydney<br />
1:45pm – 2:00pm<br />
4.2.3 Estimation <strong>of</strong> the pKa <strong>of</strong><br />
tri-peptides using Generalized<br />
Multiplicative ANOVA <strong>of</strong> designed data<br />
Rima Raffoul Khoury, <strong>University</strong> <strong>of</strong> New<br />
South Wales<br />
2:00pm – 2:15pm<br />
4.2.4 The Fouling <strong>of</strong> Hydrophobic<br />
Membranes by Hydrophilic Alginates:<br />
A Molecular Dynamics Study.<br />
Dr Matt Stewart, Victoria <strong>University</strong><br />
2:15pm – 2:30pm<br />
4.2.5 HIV1-TAT Peptide modified<br />
nanoparticles: insights from molecular<br />
dynamics simulations<br />
Dr Nevena Todorova, RMIT <strong>University</strong><br />
2:30pm – 2:45pm<br />
4.2.6 Ion Permeation and Selectivity in<br />
a Voltage Gated Sodium Channel<br />
Ben Corry, <strong>University</strong> <strong>of</strong> Western Australia<br />
2:45pm – 3:00pm<br />
4.2.7 The Role <strong>of</strong> Molecular Strain in<br />
Creating Ion Selectivity in Biological<br />
Molecules<br />
Michael Thomas, <strong>University</strong> <strong>of</strong> Western<br />
Australia<br />
3:00pm – 3:30pm Afternoon Refreshments McKinnon Building Foyer
Program<br />
Monday 5 December 2011<br />
Monday 5 December 2011 - Continued<br />
Keynote Session Main Theatre<br />
3:30pm – 4:00pm K.7 Molecular approaches to next generation photovoltaic energy conversion<br />
Associate Pr<strong>of</strong>essor Tim Schmidt, The <strong>University</strong> <strong>of</strong> Sydney Chair: Ron Clarke<br />
4:00pm – 4.30pm K.8 Charge photo-generation and recombination in conjugated polymer donor/<br />
fullerene acceptor bulk heterojunction solar cells<br />
Dr Attila Janos Mozer, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong> Chair: Ron Clarke<br />
4:30pm – 5:00pm K.9 Towards High-Efficiency Microalgae Bi<strong>of</strong>uel Systems<br />
Associate Pr<strong>of</strong>essor Ben Hankamer, <strong>University</strong> <strong>of</strong> Queensland Chair: Ron Clarke<br />
5:00pm – 7:00pm Poster Session 2 McKinnon Building Foyer<br />
9
10<br />
Program<br />
Tuesday 6 December 2011<br />
Tuesday 6 December 2011<br />
8.00am Registration Desk Open McKinnon Building Foyer<br />
9.00am – 9.50am Plenary Session Main Theatre<br />
Linking Protein Motions to Catalysis Chair: Michelle Coote<br />
Pr<strong>of</strong>essor Judith Klinman, <strong>University</strong> <strong>of</strong> California, Berkley<br />
9.50am – 10.30am Morning Refreshments McKinnon Building Foyer<br />
10:30am – 12:00pm Concurrent Session 5<br />
Theatre 2 Chair: Michelle Coote Main Theatre Chair: Ron Clarke<br />
10:30am – 10:45am<br />
5.1.1 A novel Molecular Dynamics<br />
approach for quantitative prediction <strong>of</strong><br />
adhesion and wettability: application<br />
to responsive surfaces<br />
Dr George Yiapanis, RMIT <strong>University</strong><br />
10:45am – 11:00am<br />
5.1.2 What can we learn from<br />
large-scale ab initio calculations <strong>of</strong><br />
ionic liquids?<br />
Dr Ekaterina Izgorodina, Monash <strong>University</strong><br />
11:00am – 11:15am<br />
5.1.3 Formation <strong>of</strong> Radical Products<br />
From Activation <strong>of</strong> Phospholipid<br />
Ozonides<br />
Shane Ellis, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong><br />
11:15am – 11:30am<br />
5.1.4 The Distal Effect <strong>of</strong> Electron-<br />
Withdrawing Groups on the Stability <strong>of</strong><br />
Peptide Enolates and its Exploitation<br />
in Synthesis<br />
Junming Ho, Australian National <strong>University</strong><br />
11:30am – 11:45am<br />
5.1.5 Molecular magnetic properties:<br />
benchmarking and applications<br />
Dr David Wilson, La Trobe <strong>University</strong><br />
11:45am – 12:00pm<br />
5.1.6 Molecular Design Rules for<br />
Frequency-Based, Universal Quantum<br />
Computers<br />
Laura McKemmish, <strong>University</strong> <strong>of</strong> Sydney<br />
10:30am – 10:45am<br />
5.2.1 Single molecule fluorescence<br />
microscopy: visualising DNA<br />
replication and repair dynamics within<br />
living E. coli cells<br />
Dr Andrew Robinson, <strong>University</strong> Of<br />
Groningen<br />
10:45am – 11:00am<br />
5.2.2 Structured Illumination<br />
Microscopy <strong>of</strong> Living Cells<br />
Dr Liisa Hirvonen, <strong>University</strong> <strong>of</strong> Melbourne<br />
11:00am – 11:15am<br />
5.2.3 Differential Dynamic Microscopy<br />
and Dynamic Light Scattering studies<br />
<strong>of</strong> Bacterial Motility<br />
Reece Nixon-Luke, RMIT <strong>University</strong><br />
11:15am – 11:30am<br />
5.2.4 Revisiting Boltzmann:<br />
disentangling solid-state NMR<br />
measurements <strong>of</strong> heterogeneous<br />
model membrane systems<br />
Dr John Gehman, Melbourne <strong>University</strong><br />
11:30am – 11:45am<br />
5.2.5 Liposomes: Stable or Kinetically<br />
Trapped?<br />
Dr Adam Mechler, La Trobe <strong>University</strong><br />
11:45am – 12:00pm<br />
5.2.6 The Effect <strong>of</strong> Crystallization on<br />
Protein Quaternary Structure<br />
Dr Don Vanselow, Nativeproteins.Blogspot.<br />
Com<br />
12:00pm – 1:15pm Lunch McKinnon Building Foyer
Program<br />
Tuesday 6 December 2011<br />
Tuesday 6 December 2011 - Continued<br />
1:15pm – 3:00pm Concurrent Session 6<br />
Theatre 2 Chair: Stephen Blanksby Main Theatre Chair: Adam Hill<br />
1:15pm – 1:45pm<br />
1:15pm – 1:30pm<br />
6.1.1 Anion Photoelectron<br />
6.2.1 Polarization effects in ion<br />
Spectroscopy <strong>of</strong> Ionic Complexes and channels and bulk from ab initio MD<br />
Clusters<br />
simulations<br />
Assistant Pr<strong>of</strong>essor Duncan Wild, The Associate Pr<strong>of</strong>essor Sedar Kuyucak,<br />
<strong>University</strong> <strong>of</strong> Western Australia<br />
<strong>University</strong> <strong>of</strong> Sydney<br />
1:30pm – 1:45pm<br />
6.2.2 A critical dual role for the pore<br />
helix in hERG K channel inactivation<br />
Dr Mathew Perry, Victor Chang Cardiac<br />
Research Institute<br />
1:45pm – 2:00pm<br />
6.1.2 Ion Mobility Mass Spectrometry<br />
Reveals New Insights into Structure<br />
and Assembly <strong>of</strong> Protein Complexes<br />
Dr Tara Pukala, <strong>University</strong> <strong>of</strong> Adelaide<br />
2:00pm – 2:15pm<br />
6.1.3 Nanomechanics <strong>of</strong> live bacterial<br />
cells<br />
Associate Pr<strong>of</strong>essor Michelle Gee,<br />
<strong>University</strong> <strong>of</strong> Melbourne<br />
2:15pm – 2:30pm<br />
6.1.4 Gas-phase structures <strong>of</strong> two<br />
isomers <strong>of</strong> deoxyguanosine radical<br />
cation: experiments and theory<br />
Dr Linda Feketova, <strong>University</strong> <strong>of</strong> Melbourne<br />
2:30pm – 2:45pm<br />
6.1.5 Attaching Molecular Hydrogen to<br />
Atomic Ions<br />
Pr<strong>of</strong>essor Evan Bieske, <strong>University</strong> <strong>of</strong><br />
Melbourne<br />
2:45pm – 3:00pm<br />
6.1.6 Which Density Functionals can be<br />
reliably applied to Main Group<br />
Thermochemistry?<br />
Dr Lars Goerigk, <strong>University</strong> <strong>of</strong> Sydney<br />
1:45pm – 2:00pm<br />
6.2.3 Bimodal Regulation <strong>of</strong> hERG<br />
Gating by the N-Terminal Tail as<br />
Revealed by Voltage Clamp<br />
Fluorometry<br />
Dr <strong>Peter</strong> Tan, Victor Chang Cardiac<br />
Research Institute<br />
2:00pm – 2:15pm<br />
6.2.4 Studies <strong>of</strong> Bacterial<br />
Mechanosensitive (MS) Channels<br />
under High Hydrostati |Pressure<br />
Evgeny Petrov, Victor Chang Cardiac<br />
Research Institute<br />
2:15pm – 2:30pm<br />
6.2.5 SPontaneous Oscillatory<br />
Contractions (SPOC): Assessing the<br />
Contractile Performance <strong>of</strong> Human<br />
Cardiomyopathies<br />
Amy Li, Sydney <strong>University</strong><br />
2:30pm – 2:45pm<br />
6.2.6 Does GLUT4 queue to get to the<br />
plasma membrane?<br />
Adelle Coster,<strong>University</strong> <strong>of</strong> New South<br />
Wales<br />
2:45pm – 3:00pm<br />
6.2.7 Dynamics <strong>of</strong> protein hydration<br />
water<br />
Dr Kathleen Wood, ANSTO, Bragg Institute<br />
3:00pm – 3:30pm Afternoon Refreshments McKinnon Building Foyer<br />
11
12<br />
Program<br />
Tuesday 6 December 2011<br />
Tuesday 6 December 2011 - Continued<br />
3:30pm – 4:20pm Plenary Session Main Theatre<br />
Technology that drives new science: 2D IR spectroscopy and its application to<br />
protein aggregation and drug binding<br />
Pr<strong>of</strong>essor Martin Zanni, <strong>University</strong> <strong>of</strong> Wisconsin, Madison Chair: Jamie Vandenberg<br />
4:20pm – 4.50pm K e y n o t e S e s s i o n M a i n T h e a t r e<br />
K.10 Chair: Jamie Vandenberg<br />
Dephosphorylation <strong>of</strong> the calcium pump – an infrared spectroscopy and density<br />
functional theory study<br />
Pr<strong>of</strong>essor Andreas Barth, <strong>University</strong> <strong>of</strong> Stockholm<br />
4:50pm – 5:10pm K.11 Chair: Jamie Vandenberg<br />
Molecular Mechanisms <strong>of</strong> K+ Selectivity <strong>of</strong> the Na/K Pump<br />
Haibo Yu, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong><br />
5:10pm – 5:30pm K.12 Chair: Jamie Vandenberg<br />
Towards rational control <strong>of</strong> the bacterial flagellar motor<br />
Dr Lawrence Lee, The Victor Chang Cardiac Research Institute<br />
7:00pm – 10:30pm Farewell BBQ Novotel <strong>Wollongong</strong><br />
Half Page Advert - Newspec
Conference Venue and Social Program<br />
Conference Venue & Location<br />
The <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong> has grown from a<br />
small college <strong>of</strong> 300 students, to an international<br />
university with over 26 000 students over the<br />
space <strong>of</strong> 50 years.<br />
The teaching, research and cultural life <strong>of</strong> the<br />
<strong>University</strong> is supported by state-<strong>of</strong>-the-art facilities,<br />
including an extensive library collection, an<br />
interactive Science Centre, and a Recreation &<br />
Aquatic Centre. In late 2002, the <strong>University</strong><br />
announced the establishment <strong>of</strong> a <strong>Wollongong</strong><br />
Innovation Campus on a 20 hectare site at<br />
Brandon Park. A joint venture with the NSW<br />
government, the private sector and local councils,<br />
this science and technology precinct will be<br />
developed over a ten year period commencing in<br />
2003.<br />
Welcome Mixer<br />
Saturday 3 December<br />
McKinnon Building Foyer<br />
4:30pm – 6:30pm<br />
Additional Tickets: $50<br />
This is your first opportunity to catch up with<br />
friends and network with new colleagues.<br />
The mixer will include a relaxed canapé and<br />
beverage service.<br />
There will be a complimentary shuttle bus<br />
operating between 6:00pm and 7:00pm to take<br />
delegates from UOW to local hotels and the city.<br />
Poster Session 1<br />
Sunday 4 December<br />
McKinnon Building Foyer<br />
5:30pm – 7:30pm<br />
Additional Tickets: $50<br />
Join us for the first <strong>of</strong> two poster sessions for<br />
BioPhysChem 2011.<br />
Authors will be available to speak about their<br />
posters during this session.<br />
Light food and beverages will be served. There will<br />
be a complimentary shuttle bus operating<br />
between 7:00pm and 8:00pm pm to take<br />
delegates from UOW to local hotels and the city.<br />
Poster Session 2<br />
Monday 5 December<br />
McKinnon Building Foyer<br />
5:00pm - 7:00pm<br />
Additional Tickets: $50<br />
Join us for the second poster session for<br />
BioPhysChem 2011.<br />
Authors will be available to speak about their<br />
posters during this session.<br />
Light food and beverages will be served. The<br />
Gong Shuttle will be running its usual service until<br />
9:00pm to take delegates from UOW to local<br />
hotels and the city.<br />
Farewell BBQ<br />
Tuesday 6 December<br />
Novotel <strong>Wollongong</strong><br />
7:00pm – 10:30pm<br />
Additional Tickets: $100<br />
Come and wind down after a full few days <strong>of</strong><br />
conferencing. Catch up with old friends and<br />
colleagues and newly made acquaintances over a<br />
drink and some scrumptious BBQ food, and be<br />
stunned with the views that the newly built outdoor<br />
deck at the Novotel <strong>Wollongong</strong> <strong>of</strong>fer.<br />
Please note: All social functions are included in a<br />
full conference registration. Day registrants and<br />
accompanying person tickets can be purchased<br />
at an extra cost. Delegates will need to make their<br />
own way to the Novotel <strong>Wollongong</strong> for the<br />
Farewell BBQ.<br />
13
14<br />
Pre-Conference<br />
Computational Chemistry Workshop<br />
This workshop will provide an introduction to<br />
computational chemistry methods including<br />
molecular mechanics, semi-empirical theories,<br />
molecular orbital methods and density functional<br />
theory (DFT).<br />
The basis sets commonly used in molecular orbital<br />
and DFT calculations will also be introduced.<br />
The reliability and accuracy <strong>of</strong> different methods<br />
and/or basis sets for different applications will be<br />
examined. The topics covered in the main part <strong>of</strong><br />
the workshop are:<br />
- Classical Mechanics and Molecular Force Fields<br />
- Semi-empirical Methods<br />
- Hartree<br />
- Fock Theory<br />
- Basis Sets<br />
- Electron Correlation<br />
- Density Functional Theory<br />
- Solvation<br />
Interspersed throughout the workshop will be<br />
“Masterclasses” <strong>of</strong> approximately 40 minutes, with<br />
guest lecturers focussing on particular<br />
computational problems. Concurrent with the<br />
masterclasses will be workshops aimed at<br />
teaching participants less familiar with<br />
computational methods how to apply these<br />
methods to standard problems.<br />
The following people are contributing to the<br />
workshop:<br />
- Pr<strong>of</strong> Tim Clarke<br />
- Pr<strong>of</strong> Michelle Coote<br />
- Pr<strong>of</strong> Leo Radom<br />
- Pr<strong>of</strong> <strong>Peter</strong> Gill<br />
- Dr Seth Olsen<br />
- Dr Haibo Yu<br />
- Pr<strong>of</strong> Brian Yates
Pr<strong>of</strong>essor Martin Chalfie<br />
<strong>University</strong> <strong>of</strong> Columbia<br />
Sunday 4 December 9.00am – 9.50am<br />
GFP: Lighting Up Life<br />
Biography<br />
Martin Chalfie is the William R. Kenan, Jr.<br />
Pr<strong>of</strong>essor <strong>of</strong> Biological Sciences and former chair<br />
<strong>of</strong> the Department <strong>of</strong> Biological Sciences at<br />
Columbia <strong>University</strong>. In 2008 he shared the Nobel<br />
Prize in Chemistry with Osamu Shimomura and<br />
Roger Y. Tsien for his introduction <strong>of</strong> Green<br />
Fluorescent Protein (GFP) as a biological marker.<br />
Dr. Chalfie was born in Chicago, Illinois. He<br />
obtained both his A.B. and Ph.D. from Harvard<br />
<strong>University</strong> and then did postdoctoral research with<br />
Sydney Brenner at the MRC Laboratory <strong>of</strong><br />
Molecular Biology, Cambridge, England. He joined<br />
the faculty <strong>of</strong> Columbia <strong>University</strong> as an Assistant<br />
Pr<strong>of</strong>essor in 1982 and has been there ever since.<br />
He uses the nematode Caenorhabditis elegans to<br />
investigate nerve cell development and function,<br />
concentrating primarily on genes used in<br />
mechanosensory neurons. His research has been<br />
directed toward answering two quite different<br />
biological questions: How do different types <strong>of</strong><br />
nerve cells acquire and maintain their unique<br />
characteristics? and How do sensory cells<br />
Plenary Speakers<br />
respond to mechanical signals? In the course <strong>of</strong><br />
his studies, he has introduced several novel<br />
biological methods in addition to his work with<br />
GFP.<br />
Dr. Chalfie is a member <strong>of</strong> the National Academy<br />
<strong>of</strong> Sciences and the Institute <strong>of</strong> Medicine and a<br />
fellow <strong>of</strong> the American Academy <strong>of</strong> Arts and<br />
Sciences, the American Association for the<br />
Advancement <strong>of</strong> Science, the Institute <strong>of</strong> Medicine,<br />
and the Royal Society <strong>of</strong> Chemistry (Hon.). He<br />
shared the 2006 Lewis S. Rosenstiel Award for<br />
Distinguished Work in Basic Medical Science from<br />
Brandeis <strong>University</strong> and the 2008 E. B. Wilson<br />
Medal from the American Society for Cell Biology<br />
with Roger Tsien.<br />
Abstract<br />
The great American baseball player Yogi Berra<br />
once said, “You can observe a lot by watching.”<br />
Unfortunately, before the early 1990s observations<br />
in the biological sciences were usually done on<br />
dead specimens that were specially prepared and<br />
permeabilized to allow entry <strong>of</strong> reagents to stain<br />
cell components. These methods allowed a<br />
glimpse <strong>of</strong> what cells were doing, but they gave a<br />
necessarily static view <strong>of</strong> life, just snapshots in<br />
time. GFP and other fluorescent proteins<br />
revolutionized the biological sciences because<br />
these proteins allowed scientists to look at the<br />
inner workings <strong>of</strong> living cells. GFP can be used to<br />
tell where genes are turned on, where proteins are<br />
located within tissues, and how cell activities<br />
change over time. Once a cell can be seen, it can<br />
be studied and manipulated. The discovery and<br />
development <strong>of</strong> GFP also provide a very nice<br />
example <strong>of</strong> how scientific progress is <strong>of</strong>ten made:<br />
through accidental discoveries, the willingness to<br />
ignore previous assumptions and take chances,<br />
and the combined efforts <strong>of</strong> many people. The<br />
story <strong>of</strong> GFP also shows the importance <strong>of</strong> basic<br />
research on non-traditional organisms.<br />
15
16<br />
Plenary Speakers<br />
Pr<strong>of</strong>essor Tim Clarke<br />
The <strong>University</strong> <strong>of</strong> Erlangen-Nuremberg<br />
Sunday 4 December 2011 – 3.30pm – 4.20pm<br />
Simulating Protein-DNA Switches<br />
Biography<br />
Tim Clark was born in southern England and<br />
studied chemistry at the <strong>University</strong> <strong>of</strong> Kent at<br />
Canterbury, where he was awarded a first class<br />
honors Bachelor <strong>of</strong> Science in 1970. He obtained<br />
his Ph.D. from the Queen’s <strong>University</strong> Belfast in<br />
1973 after working on the thermochemistry and<br />
solid phase properties <strong>of</strong> adamantane and<br />
diamantane derivatives. After two years as an<br />
Imperial Chemical Industries Postdoctoral Fellow<br />
in Belfast, he moved in 1975 to Princeton<br />
<strong>University</strong> as a NATO Postdoctoral Fellow working<br />
for Paul Schleyer. He then followed Schleyer to the<br />
Institut für Organische Chemie <strong>of</strong> the Universität<br />
Erlangen-Nürnberg in 1976. He is currently<br />
Technical Director <strong>of</strong> the Computer-Chemie-<br />
Centrum in Erlangen and Director <strong>of</strong> the Centre for<br />
Molecular design and Pr<strong>of</strong>essor <strong>of</strong> Computational<br />
Chemistry at the <strong>University</strong> <strong>of</strong> Portsmouth (UK).<br />
His research is concentrated on the development<br />
<strong>of</strong> calculational techniques for simulation and<br />
cheminformatics and their use for a variety <strong>of</strong><br />
chemical and biological applications. Because <strong>of</strong><br />
the central function <strong>of</strong> CCC in Erlangen, many <strong>of</strong><br />
the applied research topics are interdisciplinary, in<br />
particular with respect to signal transduction in<br />
biological systems and catalytic reactivity for<br />
redox-active metal complexes.<br />
The method development currently being carried<br />
out in the group follows two main directions.<br />
Firstly, development <strong>of</strong> a “next generation”<br />
semiempirical molecular orbital (MO) technique to<br />
replace the pure NDDO-methods such as MNDO,<br />
AM1 and PM3, which have changed little since<br />
1977. The new technique involves new techniques<br />
to represent the nucleus and non-valence<br />
electrons and an additional dispersion term based<br />
on a variational technique for calculating the<br />
polarizability. As part <strong>of</strong> this development, a<br />
completely new high performance semiempirical<br />
MO-program is being developed for near-linear<br />
scaling on highly parallel (1,024 cores and more)<br />
computers and clusters. It has been tested for up<br />
to 77,000 atoms.<br />
The second method-development direction is to<br />
use local properties at molecular surfaces for<br />
cheminformatics and classical simulations. The<br />
aim is to provide an alternative to the almost<br />
universal atomistic approach, which usually suffers<br />
from a lack <strong>of</strong> generality and very restricted<br />
applicability. It is hoped that anisotropic unitedatom<br />
techniques will allow us to extend the time<br />
scale <strong>of</strong> classical simulations into the range<br />
(microseconds) necessary to be able to study<br />
allosteric changes, polymer and liquid-crystal<br />
properties etc.<br />
Other important research directions involve<br />
calculations on enzyme reaction mechanisms,<br />
usually using a hybrid QM/MM approach, model<br />
CI studies on electron transfer in proteins and<br />
organic radical ions, studies <strong>of</strong> radical reaction<br />
mechanisms and <strong>of</strong> transition-metal complexes<br />
with phosphorus-containing ligands.<br />
Tim Clark is the author <strong>of</strong> over 315 articles in<br />
scientific journals and two books, was among the<br />
top 500 most cited chemists in the 1997<br />
compilation and is the founding editor <strong>of</strong> the<br />
Journal <strong>of</strong> Molecular Modeling.<br />
Abstract<br />
Perhaps the single most important component <strong>of</strong>
many biological control networks is the switching<br />
<strong>of</strong> transcription by control (C) proteins that<br />
complex specifically to promoter DNA sequences<br />
in order to activate or repress transcription.<br />
Whereas the repression mechanism is quite easily<br />
visualized (the repressor protein blocks access <strong>of</strong><br />
the RNA polymerase to the promoter sequence<br />
and therefore blocks transcription <strong>of</strong> the encoded<br />
gene), activation is more difficult to understand.<br />
Binding an activating C-protein must recruit the<br />
sigma-subunit <strong>of</strong> the RNA-polymerase to bind to<br />
the promoter region <strong>of</strong> the DNA.<br />
The lecture will address two aspects <strong>of</strong> these<br />
switching mechanisms; how do repressor proteins<br />
that are themselves switched by small molecules<br />
or peptides achieve this switching and how does<br />
an activator protein recruit the sigma subunit? The<br />
latter question is closely interlinked with that <strong>of</strong><br />
how C-proteins achieve their high selectivity for<br />
specific DNA sequences.<br />
We will discuss very extensive molecular dynamics<br />
(MD) simulations on TetR, a repressor protein that<br />
is switched by tetracycline antibiotics, and the<br />
finely tuned Esp1396I bacterial restrictionmodification<br />
(RM) system. The latter is particularly<br />
interesting because the same C-protein acts as<br />
promoter and repressor, depending on its<br />
concentration. The result is a temporal control <strong>of</strong><br />
the RM system that is essential for it to function<br />
correctly.<br />
Our results will emphasize the role <strong>of</strong> MD<br />
simulations as prospective research tools in this<br />
area, but will also point out their limitations and the<br />
need for exhaustive validation.<br />
Plenary Speakers<br />
Pr<strong>of</strong>essor Kalina Hristova<br />
John Hopkins <strong>University</strong><br />
Monday 5 December 2011 – 9.00am – 9.50am<br />
Seeking the Physical Basis <strong>of</strong><br />
Receptor Tyrosine Kinase Signaling<br />
Biography<br />
Kalina Hristova received her B.S. degree from the<br />
<strong>University</strong> <strong>of</strong> S<strong>of</strong>ia, Bulgaria, and her Ph.D. degree<br />
from Duke <strong>University</strong>, USA. She did post-doctoral<br />
work at the <strong>University</strong> <strong>of</strong> California, Irvine. She<br />
joined the faculty at Johns Hopkins <strong>University</strong> as<br />
an Assistant Pr<strong>of</strong>essor in 2001. Now she is a<br />
Pr<strong>of</strong>essor and the Marlin U. Zimmerman Faculty<br />
Scholar in the Departments <strong>of</strong> Materials Science<br />
and Engineering and Biomedical Engineering at<br />
Johns Hopkins.<br />
Kalina Hristova is a recipient <strong>of</strong> the Margaret<br />
Oakley Dayh<strong>of</strong>f award from the American<br />
Biophysical Society. The main focus <strong>of</strong> the<br />
research in her laboratory is the thermodynamic<br />
and structural principles that underlie membrane<br />
protein folding and signal transduction across<br />
biological membranes. At the meeting, she will<br />
present recent results on lateral receptor<br />
interactions in mammalian membranes. These<br />
studies have yielded new knowledge about the<br />
physical principles behind human pathologies.<br />
17
18<br />
Plenary Speakers<br />
Receptor tyrosine kinases (RTKs) conduct<br />
biochemical signals via lateral dimerization in the<br />
plasma membrane, and their transmembrane<br />
domains play an important role in the dimerization<br />
process. Single amino acid mutations in RTK<br />
transmembrane domains induce unregulated<br />
signaling and, as a consequence, pathologies.<br />
The research in our lab is focused on the<br />
molecular mechanism behind these pathologies,<br />
and it suggests that RTK signaling in health and<br />
disease can be understood based on quantitative<br />
knowledge <strong>of</strong> receptor interaction strengths.<br />
Pr<strong>of</strong>essor Judith Klinman<br />
<strong>University</strong> <strong>of</strong> California, Berkley<br />
Tuesday 6 December 2011 – 9.00am – 9.50am<br />
Linking Protein Motions to Catalysis<br />
Biography<br />
Judith Klinman received her A.B. and Ph. D.<br />
degrees in chemistry from the <strong>University</strong> <strong>of</strong><br />
Pennsylvania. She was a postdoctoral fellow at the<br />
Weizmann Institute <strong>of</strong> Science, Rehovot, Israel and<br />
spent 10 years at the Institute for Cancer Research<br />
in Philadelphia, first as a postdoctoral fellow with<br />
Irwin Rose and later as a Staff Scientist. She has<br />
been on the faculty <strong>of</strong> the <strong>University</strong> <strong>of</strong> California,<br />
Berkeley, since 1978. During her tenure at<br />
Berkeley, she has been a Chancellor’s Pr<strong>of</strong>essor,<br />
Guggenheim Fellow and Miller Fellow. She has<br />
been elected to the National Academy <strong>of</strong><br />
Sciences, the American Academy <strong>of</strong> Arts and<br />
Sciences, and the American Philosophical Society,<br />
and has received the Repligen Award and the<br />
Remsen Award from the American Chemical<br />
Society; the Merck Award from the American<br />
Advancement <strong>of</strong> Science; she is also a member <strong>of</strong><br />
the Royal Society <strong>of</strong> Chemistry. She was awarded<br />
an honorary Ph. D. from the <strong>University</strong> <strong>of</strong> Uppsala,<br />
Sweden in 2000 and an honorary degree from the<br />
<strong>University</strong> <strong>of</strong> Pennsylvania in 2006.<br />
Her research is currently focused on four areas: (i)<br />
nuclear tunneling in enzyme-catalyzed reactions<br />
and the relationship <strong>of</strong> this phenomenon to the role<br />
<strong>of</strong> protein dynamics in catalysis; (ii) the<br />
development <strong>of</strong> a general theory for enzyme<br />
catalysis that utilizes protein motions to generate<br />
active site compression; (iii) the mechanism <strong>of</strong><br />
dioxygen activation by enzymes; and (iv) the<br />
biogenesis and catalytic mechanism <strong>of</strong> quinoproteins<br />
and c<strong>of</strong>actors.<br />
In addition to her lifelong fascination with enzymes,<br />
Dr. Klinman enjoys adventure travel, spending time<br />
with friends and family (especially her seven<br />
grandchildren) and weekend retreats in Sonoma<br />
County.<br />
Abstract<br />
Understanding the physical origins <strong>of</strong> the<br />
enormous rate accelerations catalyzed by<br />
enzymes remains a major challenge in chemistry<br />
and biophysics. Early and persistent theories <strong>of</strong><br />
enzymatic rate acceleration were based on simple<br />
transition state approximations and relied on static<br />
three-dimensional representations <strong>of</strong> enzymes for<br />
interpretation. Work during the past decade has<br />
pointed increasingly toward the essential roles <strong>of</strong><br />
protein motion/flexibility, not only in facilitating<br />
substrate binding and product release steps, but<br />
also in understanding the catalysis <strong>of</strong> bond<br />
cleavage events. This talk will focus on the<br />
quantum properties <strong>of</strong> C-H activation in enzyme<br />
reactions, the resulting implication <strong>of</strong> active site<br />
compression, and the role <strong>of</strong> protein<br />
conformational sampling in achieving such<br />
compression. (Supported by grants from the NIH<br />
and NSF)
Pr<strong>of</strong>essor Martin Zanni<br />
<strong>University</strong> <strong>of</strong> Wisconsin, Madison<br />
Tuesday 6 December 2011 – 3:30pm – 4:20pm<br />
Technology that drives new science:<br />
2D IR spectroscopy and its<br />
application to protein aggregation<br />
and drug binding.<br />
Biography<br />
Martin Zanni is the Meloche-Bascom Pr<strong>of</strong>essor <strong>of</strong><br />
Chemistry at the <strong>University</strong> <strong>of</strong> Wisconsin-Madison.<br />
He was a Ph.D. student with Daniel Neumark at<br />
the <strong>University</strong> <strong>of</strong> California-Berkeley and an NIH<br />
postdoctoral researcher with Robin Hochstrasser<br />
at the <strong>University</strong> <strong>of</strong> Pennsylvania. Pr<strong>of</strong>. Zanni<br />
specializes in 2D IR spectroscopy and its<br />
application to problems in the biological and<br />
energy sciences. He has contributed to the<br />
technological underpinnings <strong>of</strong> the technique,<br />
written a textbook on the subject, and uncovered<br />
important scientific details on systems that are<br />
very difficult to study with other techniques. He<br />
has received much recognition for his work,<br />
including the Presidential Early Career Award for<br />
Scientists and Engineers, the Sackler Prize, and<br />
the National Academy <strong>of</strong> Sciences Research<br />
Initiatives Award.<br />
Plenary Speakers<br />
Abstract<br />
2D IR spectroscopy is proving to be a very useful<br />
tool for studying molecular structures and their<br />
dynamics. It is now being applied in fields ranging<br />
from biophysics to the energy sciences. In this<br />
talk, I will present our contributions to the<br />
technological development <strong>of</strong> this exciting<br />
spectroscopy as well as an application to amyloid<br />
fiber formation and drug inhibition. A few years<br />
ago, we invented a mid-IR pulse shaper that<br />
enabled us to computer generate the 2D IR pulse<br />
trains. This device makes data collection faster,<br />
more accurate, and enables new capabilities such<br />
as phase cycling. Using this method, we can now<br />
rapidly scan 2D IR spectra to monitor structural<br />
kinetics, which we have done to monitor the<br />
aggregation <strong>of</strong> amylin, which is the polypeptide<br />
associated with type 2 diabetes. We will present<br />
results in which we have time-resolved the<br />
secondary structure <strong>of</strong> individual residues,<br />
providing some <strong>of</strong> the most detailed information<br />
available on fiber formation. Moreover, I will also<br />
present our recent work on drug binding, in which<br />
we have resolved the binding site and mechanism<br />
<strong>of</strong> a peptide inhibitor that blocks fiber formation.<br />
With 2D IR spectroscopy, we obtain an<br />
unprecedented level <strong>of</strong> structural and kinetic detail<br />
on systems that are traditionally difficult to study<br />
with standard structural biology tools.<br />
19
20<br />
Biographies and Abstracts<br />
Saturday 3 December - Session 1<br />
Keynote Session - Main Theatre<br />
K.1 - 1:30pm – 1:55pm<br />
2010 RACI Physical Chemistry Medallist<br />
Lecture<br />
Adventures In Free Radical<br />
Chemistry: A Computational<br />
Approach<br />
Pr<strong>of</strong> Leo Radom<br />
School <strong>of</strong> Chemistry and ARC Centre <strong>of</strong> Excellence for Free<br />
Radical Chemistry and Biotechnology, <strong>University</strong> <strong>of</strong><br />
Sydney, Sydney, NSW 2006, Australia<br />
Biography<br />
Leo Radom is Pr<strong>of</strong>essor <strong>of</strong> Chemistry at the<br />
<strong>University</strong> <strong>of</strong> Sydney. After completing a PhD at<br />
that university in 1969, he spent an extended<br />
postdoc with John Pople at Carnegie-Mellon<br />
<strong>University</strong> before returning to Australia with a QE II<br />
Fellowship at the Australian National <strong>University</strong>.<br />
He moved from the ANU to the <strong>University</strong> <strong>of</strong><br />
Sydney in 2003. Leo has been elected to the<br />
Australian Academy <strong>of</strong> Science and to the<br />
International Academy <strong>of</strong> Quantum Molecular<br />
Science. He has been awarded the Rennie Medal<br />
and HG Smith Medal <strong>of</strong> the RACI, the Schrödinger<br />
Medal <strong>of</strong> WATOC, the Fukui Medal <strong>of</strong> APATCC, the<br />
David Craig Medal <strong>of</strong> the Australian Academy <strong>of</strong><br />
Science, and the Centenary Medal <strong>of</strong> the<br />
Australian Government. He has been a Named<br />
Lecturer or Pr<strong>of</strong>essor in Switzerland, USA, UK,<br />
Spain, Australia, Israel and Canada. Leo’s main<br />
research interests are concerned with the study <strong>of</strong><br />
the structures and stabilities <strong>of</strong> molecules and the<br />
mechanisms <strong>of</strong> reactions in which they are<br />
involved by use <strong>of</strong> ab initio quantum chemistry<br />
computations. Current areas <strong>of</strong> interest include<br />
free radical chemistry, enzyme-catalyzed reactions<br />
and zeolite chemistry. He is currently a CI in the<br />
ARC Centre <strong>of</strong> Excellence in Free Radical<br />
Chemistry and Biotechnology, and the immediate<br />
Past-President <strong>of</strong> the World Association <strong>of</strong><br />
Theoretical and Computational Chemists.<br />
Abstract<br />
Radicals are ubiquitous in chemistry and biology.<br />
Because they are reactive species, they are <strong>of</strong>ten<br />
difficult to study experimentally and therefore<br />
theory has a potentially useful role to play in their<br />
characterisation. In recent years, we have been<br />
using quantum chemistry computations to<br />
investigate the structures, stabilities and<br />
reactivities <strong>of</strong> radicals. We have also been<br />
examining ways to obtain improved theoretical<br />
descriptions <strong>of</strong> radicals. Highlights from this<br />
research will be presented.<br />
K.2 - 1:55pm – 2:20pm<br />
2011 Bob Robertson Medal (ASB)<br />
The 2011 Medal Recipient<br />
announced on the day<br />
K.3 - 2:20pm – 2:45pm<br />
A Life in Physical Chemistry: From<br />
Fundamentals to Applications<br />
Pr<strong>of</strong> Keith King1 1 School <strong>of</strong> Chemical Engineering, <strong>University</strong> <strong>of</strong> Adelaide, SA<br />
5005, keith.king@adelaide.edu.au<br />
2011 RACI Physical Chemistry<br />
Medallist<br />
Biography<br />
Keith King is currently Emeritus Pr<strong>of</strong>essor and<br />
Visiting Pr<strong>of</strong>essor <strong>of</strong> Chemical Engineering at the<br />
<strong>University</strong> <strong>of</strong> Adelaide. His areas <strong>of</strong> expertise cover<br />
chemical kinetics, energy transfer and catalysis,<br />
laser diagnostics, combustion and flames,<br />
including ignition and explosions, soot formation,<br />
and biodiesel. He is a Fellow <strong>of</strong> the Institution <strong>of</strong><br />
Chemical Engineers, a Fellow <strong>of</strong> the Royal<br />
Australian Chemical Institute, a Fellow <strong>of</strong> the Royal<br />
Society <strong>of</strong> Chemistry, and a Senior Member <strong>of</strong> the<br />
American Institute <strong>of</strong> Chemical Engineers. He was<br />
awarded the 1998 R. K. Murphy Medal by the<br />
RACI Industrial Chemistry Division for ‘Outstanding<br />
Achievements in the Practice <strong>of</strong> Chemical
Engineering and Industrial Chemistry’. He was a<br />
senior member <strong>of</strong> the team awarded the<br />
Engineering Excellence Award 2000 by Engineers<br />
Australia, SA Division for ‘Design and Development<br />
<strong>of</strong> the Fuel and Combustion System for the Sydney<br />
2000 Olympic Torch Relay’.<br />
Abstract<br />
I will present a brief account with selected<br />
highlights <strong>of</strong> my research in physical chemistry<br />
covering chemical kinetics and energy transfer,<br />
combustion, and laser diagnostics.<br />
Key Words<br />
Kinetics, energy transfer, combustion, lasers<br />
Stanton Scientific<br />
MASS S P E C TROME T E R S U PPOR T<br />
A N D V A C U U M SCIE N C E PRO D U CTS<br />
Mass Spectral data bases<br />
N.I.S.T. (USA) and Wiley Mass Spectral Libraries<br />
Mass Spectral S<strong>of</strong>tware including file conversion<br />
Aalborg Mass Flow Meters and Controllers<br />
Electron Multipliers and Channeltron Detectors<br />
Mass Spectrometer Filament repair service<br />
Representing Scientific Instrument Services New Jersey USA<br />
Granville Phillips Vacuum Gauges<br />
www.stantonscientific.com Email: bill@stantonscientific.com<br />
Ph: 02 66856902 Fax: 02 85690588<br />
21
22<br />
Biographies and Abstracts<br />
Sunday 4 December - Session 1<br />
Session 1 – Theatre 2<br />
1.1.1 10:30am – 10:45am<br />
Density Functional Theory Studies <strong>of</strong><br />
High-Oxidation State Palladium<br />
Systems<br />
Brian F Yates 1 , Alireza Ariafard2 , Allan J<br />
Canty3 1 <strong>University</strong> <strong>of</strong> Tasmania, Private Bag 75, Hobart TAS 7001, Brian.<br />
Yates@utas.edu.au<br />
2 <strong>University</strong> <strong>of</strong> Tasmania, Private Bag 75, Hobart TAS 7001,<br />
Alireza.Ariafard@utas.edu.au<br />
3 <strong>University</strong> <strong>of</strong> Tasmania, Private Bag 75, Hobart TAS 7001, Allan.<br />
Canty@utas.edu.au<br />
Biography<br />
Pr<strong>of</strong>essor Brian Yates heads up an active research<br />
program in computational chemistry with<br />
particular applications to organometallic and<br />
organic chemistry, and his research is funded by<br />
the Australian Research Council (ARC). He is a<br />
current member <strong>of</strong> the ARC College <strong>of</strong> Experts<br />
and is Chair <strong>of</strong> the Physics Chemistry and Earth<br />
Sciences panel in 2011. He sits on the board <strong>of</strong> the<br />
National Computational Infrastructure (NCI) and is<br />
chair <strong>of</strong> the committee which allocates grants <strong>of</strong><br />
supercomputer time on the national high<br />
performance computing facility. Brian has also<br />
built up a strong reputation for teaching<br />
excellence. He has been awarded competitively<br />
funded teaching development grants at the<br />
national (CAUT/CUTSD, ALTC) and state levels,<br />
and he has been rewarded with local (UTAS) and<br />
national (2006 Carrick Award, 2007 RACI<br />
Chemical Education medal) teaching excellence<br />
awards. He is currently a Pr<strong>of</strong>essor in Chemistry at<br />
the <strong>University</strong> <strong>of</strong> Tasmania and an Australian<br />
Learning and Teaching Council (ALTC) Discipline<br />
Scholar in Science.<br />
Abstract<br />
Reactions involving palladium compounds with<br />
organic reagents are important for the synthesis <strong>of</strong><br />
new materials, pharmaceuticals, and biological<br />
related molecules <strong>of</strong> value in medical research.<br />
Recently there has been much interest in the<br />
chemistry <strong>of</strong> high oxidation state palladium<br />
systems because <strong>of</strong> the enhanced ability <strong>of</strong> these<br />
systems to form new carbon-carbon bonds. In this<br />
presentation I will discuss the computational<br />
chemistry research in my group which is aimed at<br />
understanding organometallic chemistry, with a<br />
particular focus on bimetallic high oxidation state<br />
palladium systems.<br />
1.1.2 10:45am – 11:00am<br />
Interpolating Molecular Potential<br />
Energy and Property Surfaces<br />
M. J. T. Jordan, S. J. Kolmann and M. Morris<br />
Department <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney, Sydney, NSW,<br />
2006, email: m.jordan@chem.usyd.edu.au<br />
Biography<br />
Meredith Jordan is currently a senior lecturer in<br />
chemistry at the <strong>University</strong> <strong>of</strong> Sydney. She<br />
completed a PhD at Sydney with Pr<strong>of</strong>essor Bob<br />
Gilbert before undertaking a postdoctoral<br />
fellowship with Pr<strong>of</strong>essor Mick Collins at the RSC<br />
at the Australian National <strong>University</strong>, then being<br />
awarded a research fellowship at Girton College,<br />
Cambridge to work with Pr<strong>of</strong>essor David Clary<br />
(who promptly left for London!). Her research<br />
focuses on the study and description <strong>of</strong> molecular<br />
interactions.<br />
Abstract<br />
Over the last 15 years we have developed a<br />
modified Shepard interpolation scheme that can<br />
be used to iteratively construct molecular potential<br />
energy surfaces (PES). Such surfaces have been<br />
used in many applications, including classical<br />
trajectory simulations, quantum dynamics and<br />
quantum diffusion Monte Carlo simulations <strong>of</strong><br />
ground state wavefunctions. Some systems,<br />
however, remain problematic and the methodology<br />
is still not quite “black box”. Here we describe<br />
some recent applications and modifications <strong>of</strong> the<br />
scheme in pursuit <strong>of</strong> this goal.
Modified Shepard interpolation can also be used<br />
to construct molecular property surfaces, which,<br />
in many cases, are more straightforward than PES.<br />
We have used a modified Shepard interpolation to<br />
construct dipole moment surfaces (DMS) for water<br />
and hydrogen cyanide, based on analytic DMS in<br />
the literature. These analytic DMS allow us to<br />
optimise the parameters and the form <strong>of</strong> the<br />
interpolated DMS. Because the DMS is more<br />
slowly varying than the PES we obtain accurate<br />
results using both a standard (ie zeroth order)<br />
Shepard expansion <strong>of</strong> each component <strong>of</strong> the<br />
dipole moment vector and a modified Shepard<br />
interpolation based on first order Taylor<br />
expansions <strong>of</strong> the dipole moment. Given that our<br />
PES interpolation method uses second derivatives<br />
<strong>of</strong> the potential, it is straightforward to obtain the<br />
dipole moment vector as part <strong>of</strong> the “standard”<br />
PES interpolation, that is, to generate both the PES<br />
and the DMS simultaneously. The PES together<br />
with the DMS are used to predict vibrationally<br />
averaged dipole moments and rovibrational line<br />
strengths as well as the linear response <strong>of</strong> a<br />
molecule to an external electric field.<br />
1.1.3 11:00am – 11:15am<br />
The Relationship Between Intrinsic<br />
Bond Energy and Intrinsic Radical<br />
Stability: Can This be Used to Test<br />
the Untestable?<br />
Michelle L. Coote and Ching Yeh Lin<br />
Australian Research Council Centre <strong>of</strong> Excellence for Free Radical<br />
Chemistry and Biotechnology,<br />
Research School <strong>of</strong> Chemistry, Australian National <strong>University</strong>,<br />
Canberra, ACT 0200, Australia<br />
Biography<br />
Pr<strong>of</strong>essor Michelle Coote is a graduate <strong>of</strong> the<br />
<strong>University</strong> <strong>of</strong> New South Wales, where she<br />
completed a B.Sc. (Hons) in industrial chemistry<br />
(1995), followed by a Ph.D. in polymer chemistry<br />
(2000). Following postdoctoral work at the<br />
<strong>University</strong> <strong>of</strong> Durham, UK, she joined the Research<br />
School <strong>of</strong> Chemistry, Australian National <strong>University</strong><br />
in 2001, initially as a postdoctoral fellow with<br />
Pr<strong>of</strong>essor Leo Radom. She established her own<br />
research group in 2004 and has recently taken up<br />
Biographies and Abstracts<br />
Sunday 4 December - Session 1<br />
an ARC Future Fellowship. She has published<br />
extensively in the fields <strong>of</strong> polymer chemistry,<br />
radical chemistry and computational quantum<br />
chemistry, and is a member <strong>of</strong> the ARC Centre <strong>of</strong><br />
Excellence for Free-Radical Chemistry and<br />
Biotechnology. She has received many awards<br />
including the 2001 IUPAC prize for young<br />
scientists, the RACI Cornforth medal (2000),<br />
Rennie medal (2006) and David Sangster Polymer<br />
Science and Technology Achievement Award<br />
(2010), and the Le Fevre Memorial Prize <strong>of</strong> the<br />
Australian Academy <strong>of</strong> Science (2010).<br />
Abstract<br />
One <strong>of</strong> the major tasks <strong>of</strong> chemistry is structurereactivity<br />
analysis –attempting model and explain<br />
the mechanism, kinetics and thermodynamics <strong>of</strong> a<br />
chemical process in terms <strong>of</strong> contributions <strong>of</strong> the<br />
various functional groups present. This type <strong>of</strong><br />
analysis then allows one to make predictions<br />
about how to manipulate chemical reactions by<br />
changing the functional groups, and can thereby<br />
guide reagent and catalyst design. Such studies<br />
have proven extremely useful in radical chemistry<br />
as such processes <strong>of</strong>ten involve several competing<br />
reactions in which small changes to substitution<br />
patterns <strong>of</strong> one or more reagents can have a major<br />
positive or negative impact on the reaction<br />
outcome. One the key “tools” one uses when<br />
analysing radical reactions is radical stability.<br />
Unfortunately, defining and measuring intrinsic<br />
radical stability is not straightforward.1 From a<br />
quantum mechanical perspective, the stability (or<br />
‘propensity to react’) <strong>of</strong> any species is only<br />
precisely definable in the context <strong>of</strong> a balanced<br />
chemical reaction. However, this only defines the<br />
‘stability’ <strong>of</strong> a given species relative to a particular<br />
chemical system. It does not allow us to infer<br />
anything about the behaviour <strong>of</strong> that molecule in<br />
any other chemical context, thereby making this<br />
notion <strong>of</strong> ‘stability’ virtually useless for the<br />
purposes <strong>of</strong> chemical explanation and prediction.<br />
It is unsurprising, then, that chemists have sought<br />
to define a measure <strong>of</strong> stability that could be<br />
considered an intrinsic property <strong>of</strong> a given species<br />
— a measure <strong>of</strong> a molecule’s propensity to react<br />
across a range <strong>of</strong> chemical reactions that could in<br />
turn be related to the chemical structure <strong>of</strong> the<br />
molecule using, for example, qualitative molecular<br />
orbital theory arguments. However, the problem<br />
23
24<br />
Biographies and Abstracts<br />
Sunday 4 December - Session 1<br />
with this approach is that there is no obvious<br />
intrinsic property <strong>of</strong> an isolated molecule definable<br />
in quantum-mechanical terms that can provide a<br />
guide to its propensity to react across a range <strong>of</strong><br />
chemical systems.<br />
In this talk we will look at how chemists have<br />
sought to address this problem for the case <strong>of</strong><br />
radical stability. We will examine some <strong>of</strong> the<br />
leading methods for defining and measuring<br />
radical stability, including the familiar radical<br />
stabilization energy (RSE),2 along with some<br />
lesser-known alternatives based on corrected<br />
carbon-carbon bond energies,3,4 and direct<br />
measures <strong>of</strong> the extent <strong>of</strong> radical delocalisation.<br />
Using the results <strong>of</strong> high-level ab initio molecular<br />
orbital theory calculations we will compare the<br />
predictions <strong>of</strong> the various schemes with one<br />
another, and with expectations based on<br />
“chemical intuition”, with a view to establishing a<br />
reliable method for measuring relative radical<br />
stability.5 We will then examine the relationship<br />
between intrinsic radical stability and intrinsic bond<br />
energies, and show how “reliable” radical stability<br />
trends can be used to evaluate the underlying<br />
physical meaning some commonly used energy<br />
decomposition schemes.6<br />
1.1.4 11:15am – 11:30am<br />
Molecular Oxygen as Energy<br />
Mediator for Photochemical<br />
Upconversion <strong>of</strong> Near-Infrared Light<br />
Burkhard Fückel1 , Derrick A. Roberts1 , Yuen<br />
Yap Cheng1 , Raphaël G. C. R. Clady1 , Roland B.<br />
Piper2 , N. J. Ekins-Daukes 2 , Maxwell J. Crossley1 ,<br />
Timothy W. Schmidt1 1 School <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> Sydney, NSW 2006,<br />
Australia, burkhard.fueckel@chem.usyd.edu.au, timothy.<br />
schmidt@sydney.edu.au<br />
2 Department <strong>of</strong> Physics and the Grantham Institute for Climate<br />
Change, Imperial College, London, U.K.<br />
Biography<br />
Dr Burkhard Fückel is a postdoctoral researcher<br />
with A/Pr<strong>of</strong> Timothy Schmidt at The <strong>University</strong> <strong>of</strong><br />
Sydney. He has been awarded a Feodor Lynen<br />
research fellowship <strong>of</strong> the German Alexander von<br />
Humboldt foundation to work on “3rd generation<br />
photovoltaics” in Sydney. Before commencing his<br />
work in Sydney in 2010, he received his PhD from<br />
the Gutenberg <strong>University</strong> <strong>of</strong> Mainz, where he<br />
studied energy transfer process by single molecule<br />
spectroscopy and quantum chemistry under<br />
supervision <strong>of</strong> Pr<strong>of</strong> Thomas Basch<br />
Abstract<br />
Ground state molecular oxygen (3Sg), constitutes<br />
about 20% <strong>of</strong> the volume <strong>of</strong> air. Its chemical<br />
reactivity increases dramatically upon electronic<br />
excitation to the singlet (1Dg) state, leading to<br />
spontaneous reaction with a range <strong>of</strong> unsaturated<br />
organic compounds. Since the (1Dg) ← (3Sg)<br />
transition <strong>of</strong> molecular oxygen can be induced by<br />
quenching <strong>of</strong> a triplet excited state <strong>of</strong> an organic<br />
molecule, artificial organic systems, as for example<br />
organic photovoltaics, are generally protected from<br />
air. Biological systems, on the contrary, have<br />
evolved ways <strong>of</strong> handling singlet oxygen, such as<br />
sequestration by carotenoids that protect the<br />
sensitive pigments in the photosynthetic reaction<br />
center <strong>of</strong> plants.<br />
We present a strategy that employs singlet oxygen<br />
as an energy transmitter for photochemical<br />
upconversion (UC). Photochemical UC combines<br />
two low energy photons into one <strong>of</strong> higher energy<br />
by incoherent means [1] and has therefore<br />
attracted recent interest for applications in<br />
light-harvesting and light-emitting applications.[2]<br />
In the employed system, singlet oxygen is<br />
generated upon photoexcitation <strong>of</strong> the sensitizer<br />
molecules and then acts as an energy transmitter<br />
for the UC process. The excitation energy <strong>of</strong> two<br />
singlet oxygen molecules is subsequently<br />
harvested by emitter molecules, which in turn<br />
gives rise to upconverted fluorescence <strong>of</strong> the<br />
emitter species.[3] This process furthermore<br />
reduces the rate <strong>of</strong> the photo-degradation <strong>of</strong> the<br />
sensitizer molecules. We present strategies for<br />
improvement <strong>of</strong> the currently achieved efficiencies<br />
≤0.01% to produce excited singlet states in the<br />
emitter molecules, which are currently under<br />
examination.
1.1.5 11:30am – 11:45am<br />
Time Resolved Fluorescence<br />
Imaging <strong>of</strong> Conjugated Polymer Thin<br />
Films<br />
Xiao-Tao Hao1, , Trevor A Smith1, , Kenneth P.<br />
Ghiggino2 1 School <strong>of</strong> Chemistry and ARC Centre <strong>of</strong> Excellence for Coherent<br />
X-ray Science, The <strong>University</strong> <strong>of</strong> Melbourne, Victoria 3010,<br />
xhao@unimelb.edu.au; trevoras@unimelb.edu.au<br />
2 School <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> Melbourne, Victoria 3010,<br />
ghiggino@unimelb.edu.au<br />
Biography<br />
Dr. Xiaotao Hao is a research fellow working at<br />
school <strong>of</strong> chemistry in the <strong>University</strong> <strong>of</strong> Melbourne.<br />
His research interests include photophysics <strong>of</strong> light<br />
emitting conjugated polymers, time resolved<br />
fluorescence spectroscopy and microscopy. He<br />
has published over 40 papers on peer-reviewed<br />
scientific journals.<br />
Abstract<br />
The solid-state characteristics <strong>of</strong> conjugated<br />
polymers are the key factors that govern the<br />
performance <strong>of</strong> organic opto-electronic devices in<br />
many situations. Aggregates are readily formed in<br />
most conjugated polymer films resulting in good<br />
charge transport via strongly interacting π-electron<br />
systems, but these aggregates also likely induce<br />
inhomogeneous morphologies and emission<br />
quenching, and thereby affect device efficiency.1,2<br />
Determining the spatial scale <strong>of</strong> the<br />
inhomogeneities in conjugated polymer films from<br />
nanometres to many microns, and their effect on<br />
the dynamics <strong>of</strong> electron/hole transport, is<br />
necessary to develop more efficient injection<br />
processes for high performance devices.<br />
High-resolution optical microscopy methods are a<br />
useful tool to provide this spatial information.<br />
Coupling ultrafast spectroscopic methods with<br />
high spatial resolution optical techniques makes it<br />
possible to map the dynamics <strong>of</strong> the photoinduced<br />
charge transfer/transport processes as a<br />
function <strong>of</strong> location in the film.<br />
We report here the steady state and time-resolved<br />
fluorescence emission characteristics <strong>of</strong> poly[2methoxy-5-(2-ethylhexyloxy)-1,4-phenylene<br />
vinylene] (MEH-PPV) and a water-soluble PPV<br />
Biographies and Abstracts<br />
Sunday 4 December - Session 1<br />
derivative with sulphonate-containing side chains<br />
(DPS-PPV) thin films with different conformations<br />
coated using various methods.3,4 We observed<br />
inhomogeneities in the morphology and<br />
fluorescence dynamics <strong>of</strong> thin films <strong>of</strong> the light<br />
emitting conjugated polymer, on sub-micrometre<br />
spatial and picosecond temporal scales using<br />
time-resolved scanning confocal fluorescence<br />
imaging measurements.<br />
This work illustrates the power <strong>of</strong> time-resolved<br />
emission imaging to identify regions <strong>of</strong> varying<br />
degrees <strong>of</strong> aggregation, whether pre-existing<br />
aggregates or those formed during film formation,<br />
in MEH-PPV/PMMA films cast from a range <strong>of</strong><br />
solvents. Differences observed show some<br />
correlation to the solvent from which the films are<br />
cast. The spatiotemporal spectral behaviour can<br />
be associated with degrees <strong>of</strong> aggregation, from a<br />
level <strong>of</strong> “single-chain like” regions (emitting to the<br />
blue with approximately nanosecond emission<br />
decay components) to highly aggregated<br />
(short-lived, red emitting) regions, within the film<br />
morphology. The overall time-resolved behaviour<br />
was sensitive to the degree <strong>of</strong> aggregation, which<br />
in turn was dependent on the solvent from which<br />
the film was cast.<br />
In contrast, thin films <strong>of</strong> a water-soluble DPS-PPV,<br />
formed on silica substrates by a solvent-free<br />
friction transfer technique, were highly aggregated<br />
producing “rods” <strong>of</strong> polymer aligned perpendicular<br />
to the drawing direction. This “log-rolling” is in<br />
contrast with the reported behaviour <strong>of</strong> most<br />
comparable polymers, which tend to align along<br />
the drawing direction. The photophysical<br />
behaviour <strong>of</strong> the friction-transferred film is also<br />
different compared with other film formation<br />
techniques <strong>of</strong> the same polymer.<br />
25
26<br />
Biographies and Abstracts<br />
Sunday 4 December - Session 1<br />
1.1.6 11:45am – 12:00pm<br />
Quantifying cooperative<br />
intermolecular interactions for<br />
improved carbon dioxide capture<br />
materials<br />
Joseph R. Lane1 1 Department <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Waikato, Private Bag<br />
3105, Hamilton 3240, New Zealand, jlane@waikato.ac.nz<br />
Biography<br />
Jo Lane completed his undergraduate and<br />
graduate studies in the Department <strong>of</strong> Chemistry<br />
at the <strong>University</strong> <strong>of</strong> Otago. He was awarded his<br />
BSc.(Hons.) in 2005 and his PhD in 2008 under<br />
the supervision <strong>of</strong> Pr<strong>of</strong>. Henrik Kjaegaaard. Jo<br />
then worked as a postdoctoral research fellow in<br />
the same research group before being appointed<br />
to a lectureship in the Department <strong>of</strong> Chemistry at<br />
the <strong>University</strong> <strong>of</strong> Waikato in 2009<br />
Abstract<br />
Any serious effort to reduce anthropogenic carbon<br />
dioxide (CO2) emissions must contend with the<br />
geopolitical and economic reality that fossil fuels<br />
will continue to make a dominant contribution to<br />
the world’s energy supply for decades to come.<br />
This makes development <strong>of</strong> scalable, costeffective<br />
CO2 capture technologies <strong>of</strong> equal<br />
importance to innovations in renewable energy<br />
resources over the near future.<br />
Nanoporous crystalline compounds such as metal<br />
organic framework (MOF) materials have<br />
exceptionally high surface areas and demonstrate<br />
promising ability for the separation and capture <strong>of</strong><br />
CO2. However, current MOF materials do not yet<br />
show the necessary selectivity for CO2 under<br />
conditions relevant for fossil fuel combustion.<br />
To better understand the fundamental<br />
intermolecular interactions involved in CO2<br />
adsorption, we have investigated over 100 different<br />
complexes <strong>of</strong> CO2 with various combinations <strong>of</strong><br />
electron accepting (Lewis acid) and electron<br />
donating (Lewis base) molecules. We have used<br />
the recently developed explicitly correlated<br />
coupled cluster singles doubles and perturbative<br />
triples [CCSD(T)-F12] methods and the associated<br />
VXZ-F12 (where X = D,T,Q) basis sets. We observe<br />
only modest changes in the geometric parameters<br />
<strong>of</strong> CO2 upon complexation, which suggests that<br />
the geometry <strong>of</strong> CO2 adsorbed in a nanoporous<br />
material should be similar to that <strong>of</strong> CO2 in gas<br />
phase. For complexes that exhibit simultaneous<br />
CO2-Lewis acid and CO2-Lewis base<br />
intermolecular interactions, we find that the total<br />
interaction energy is greater than the sum <strong>of</strong> the<br />
interaction energies <strong>of</strong> the constituent complexes.<br />
Furthermore, the intermolecular distances <strong>of</strong> the<br />
cooperative complexes are contracted as<br />
compared to the constituent complexes. We<br />
suggest that MOF or similar nanoporous materials<br />
could be designed with adsorption sites<br />
specifically tailored for CO2 to allow cooperative<br />
intermolecular interactions, facilitating enhanced<br />
CO2 adsorption.<br />
Session 1 – Main Theatre<br />
1.2.1 10:30am – 10:45am<br />
Spatial and spectral super-resolution<br />
– Optical imaging <strong>of</strong> nanoscopic<br />
signalling domains in 4D<br />
David Baddeley1 , Isuru Jayasinghe1 , Cherrie<br />
Kong1 , David Crossman1 , Juliette Cheyne1 ,<br />
Johanna Montgomery1 , Mark Cannell1 , Christian<br />
Soeller1 ,<br />
1 Department <strong>of</strong> Physiology, <strong>University</strong> <strong>of</strong> Auckland, Private Bag<br />
92019, Auckland, New Zealand E-mail: d.baddeley@auckland.ac.<br />
nz<br />
Biography<br />
David completed a PhD at Heidelberg <strong>University</strong> in<br />
Germany studying structured illumination and 4Pi<br />
microscopy before returning to Auckland to work<br />
on single molecule localisation microscopy. Since<br />
then he has made a number <strong>of</strong> improvements to<br />
the PALM/STORM technique and applied these to<br />
the study <strong>of</strong> the proteins involved in excitation<br />
contraction coupling in cardiac myocytes<br />
Abstract<br />
Recent single molecule localisation techniques<br />
such as PALM and STORM have made far field<br />
imaging with a resolution well below the diffraction<br />
limit a reality. Optical super-resolution promises
significantly greater freedom and ease <strong>of</strong> labelling,<br />
sample preparation and imaging than electron<br />
microscopy and it is towards realising this potential<br />
that we have directed our efforts. We will present<br />
an implementation <strong>of</strong> single molecule based<br />
localisation microscopy that puts emphasis on<br />
making the approach as practical as possible and<br />
culminates in a super-resolution workflow that is<br />
no more arduous than high-quality confocal<br />
imaging.<br />
Our approach uses a single laser line to excite<br />
multiple dyes which we discriminate in a<br />
ratiometric manner <strong>of</strong>fering spectral superresolution.<br />
By using conventional dyes in the near<br />
infra-red which are commercially available as<br />
antibody conjugates, we both simplify labelling<br />
procedures and minimise the contribution <strong>of</strong><br />
sample aut<strong>of</strong>luoresence. We will also present a<br />
novel method <strong>of</strong> extracting the z position <strong>of</strong> single<br />
molecules based on a phase ramp in the objective<br />
pupil plane. This method <strong>of</strong>fers a modest<br />
improvement in axial resolution over some existing<br />
methods <strong>of</strong> 3D localisation such as astigmatism,<br />
and is very easy to implement with components<br />
which will already be present in a typical imaging<br />
lab.<br />
The distribution <strong>of</strong> proteins and their proximity on<br />
the nanometre scale critically determines cellular<br />
signalling responses, but could previously not be<br />
examined with optical techniques. We have used<br />
our super-resolution approach to investigate a<br />
number <strong>of</strong> signalling domains and will present data<br />
from our analysis <strong>of</strong> key Ca2+ signalling proteins in<br />
cardiac myocytes, and from synaptic proteins in<br />
hippocampal culture. We have already obtained a<br />
number or surprising results such as showing that<br />
the number <strong>of</strong> Ryanodine receptor Ca2+ channels<br />
(RyRs) in peripheral couplons is considerably less<br />
than initially thought, and that cluster sizes follow a<br />
near exponential distribution compatible with a<br />
stochastic assembly process. When performing<br />
two colour imaging (e.g. with RyR, NCX, and<br />
Caveolin) we consistently find much lower levels <strong>of</strong><br />
colocalisation than predicted from diffraction<br />
limited imaging.<br />
Biographies and Abstracts<br />
Sunday 4 December - Session 1<br />
1.2.2 10:45am – 11:00am<br />
BRET based monitoring <strong>of</strong> ligand<br />
binding in the ODR-10 odorant<br />
responsive G-protein coupled<br />
receptor from Caenorhabditis<br />
elegans<br />
H. Dacres, J. Wang, V. Leitch, I. Horne, A.R.<br />
Anderson, S.C. Trowell<br />
CSIRO Food Futures National Research Flagship & CSIRO<br />
Ecosystem Sciences, Australia Helen.Dacres@csiro.au<br />
Biography<br />
Dr Helen Dacres specialises in interdisciplinary<br />
approaches to chemical sensing and biosensing<br />
systems. She received a PhD and MSc in<br />
Instrumentation and Analytical Chemistry from<br />
UMIST (Manchester, UK) for developing optical<br />
chemical sensors to detect biologically and<br />
environmentally relevant gases including nitric<br />
oxide. This research made her realise the future <strong>of</strong><br />
chemical sensing - particularly for difficult<br />
problems - would lie in biosensing so she looked<br />
for a position to gain postdoctoral experience in<br />
molecular biology and biosensing. She joined<br />
CSIRO as a postdoctoral fellow in 2005 to work on<br />
the development <strong>of</strong> a number <strong>of</strong> novel biosensors.<br />
She has designed biosensors for monitoring<br />
various disease states including blood clotting<br />
disorders and to study the effect <strong>of</strong> cancer on cell<br />
death. She is currently a research scientist at<br />
CSIRO Ecosystem Sciences developing<br />
biosensors using biological odorant receptors for<br />
detecting volatile compounds with potential<br />
application areas including defence, health and<br />
biosecurity.<br />
Abstract<br />
Humans express over 750 G-protein coupled<br />
receptors (GPCRs), which represent not only the<br />
largest class <strong>of</strong> integral membrane receptors but<br />
also the largest class <strong>of</strong> targets for therapeutic<br />
drugs. GPCRs respond to a wide range <strong>of</strong><br />
ligands, including proteins peptides and small<br />
organic molecules. GPCRs also mediate<br />
chemosensation, the senses <strong>of</strong> taste and smell, in<br />
vertebrates as well as in nematode worms. We set<br />
out to develop a biosensor transduction system<br />
27
28<br />
Biographies and Abstracts<br />
Sunday 4 December - Session 1<br />
that is compatible with GPCRs generally and with<br />
odorant receptors in particular.<br />
Fluorescence resonance energy transfer (FRET)<br />
has previously been used to monitor ligand binding<br />
by secretin receptors expressed and imaged in<br />
intact COS cells [1]. However, FRET-GPCR has<br />
not previously been demonstrated in a cell-free<br />
system. We recently reported that bioluminescent<br />
resonance energy transfer (BRET) has superior<br />
limits <strong>of</strong> detection and sensitivity in a protease<br />
assay compared to FRET [2]. Here, for the first<br />
time, we have inserted BRET (bioluminescence<br />
resonance energy transfer) transduction tags in<br />
the primary sequence <strong>of</strong> a GPCR, the nematode<br />
ODR-10 receptor, which in vivo responds to the<br />
odorant 2,3-butanedione (diacetyl).<br />
In a yeast-based cell-free system, the EC50 <strong>of</strong> a<br />
BRET2-ODR-10 biosensor was in the fM range for<br />
2,3-butanedione. The response was ligandspecific<br />
and was completely abolished by a single<br />
point mutation in the receptor sequence. The<br />
percentage change in RET ratio was several fold<br />
higher than for an equivalent FRET-ODR-10<br />
construct. Novel BRET-GPCR biosensors <strong>of</strong> this<br />
type have potential application in drug discovery,<br />
clinical diagnosis, explosive detection and quality<br />
control <strong>of</strong> food and beverage production [3].<br />
1.2.3 11:00am – 11:15am<br />
Photostable Fluorescent<br />
Nanodiamond Material: Labels for<br />
Biomolecules & FRET<br />
J M Say1 , L Brown2 , J R Rabeau3 1 Centre for Quantum Science and Technology and MQ Photonics<br />
Research Centre, Department <strong>of</strong> Physics and Department <strong>of</strong><br />
Chemistry and Biomolecular Science, Macquarie <strong>University</strong>,<br />
Sydney, New South Wales, 2109, jana.say@mq.edu.au<br />
2 Department <strong>of</strong> Chemistry and Biomolecular Science, Macquarie<br />
<strong>University</strong>, Sydney, New South Wales, 2109, louise.brown@mq.<br />
edu.au<br />
3 Centre for Quantum Science and Technology and MQ Photonics<br />
Research Centre, Department <strong>of</strong> Physics, Macquarie <strong>University</strong>,<br />
Sydney, New South Wales, 2109, james.rabeau@mq.edu.au<br />
Biography<br />
Jana Say is a PhD student at Macquarie <strong>University</strong><br />
working in the Diamond Nanoscience group. She<br />
joined the team in March 2010 after finishing her<br />
Honours Degree in Quantum Mechanics at the<br />
<strong>University</strong> <strong>of</strong> British Columbia in Canada. Jana is<br />
working jointly between the Physics and Chemistry<br />
and Bimolecular Sciences Departments and her<br />
PhD is focusing on the use <strong>of</strong> luminescent<br />
nanodiamonds for biological applications. The title<br />
<strong>of</strong> her talk today is Photostable Fluorescent<br />
Nanodiamond Material: Labels for Biomolecules &<br />
FRET.<br />
Abstract<br />
Colour centres in nanodiamonds have many<br />
properties which make them attractive for<br />
biological applications including their chemical and<br />
physical stability, biocompatibility, easy surface<br />
functionalization, near unity quantum yield,<br />
electron and nuclear spin and stable<br />
photoluminescence. These qualities enable<br />
nanodiamonds to be used in a broad range <strong>of</strong><br />
applications including biosensing, protein<br />
separation, drug delivery and single particle<br />
imaging in cells. Here we propose using<br />
fluorescent nanodiamonds as an alternative<br />
nano-label to conventional fluorophores. We also<br />
explore their use as a novel donor for Förster<br />
Resonance Energy Transfer (FRET)<br />
measurements.<br />
Over 500 different optical centres have been<br />
identified in diamond. Of these, the nitrogen<br />
vacancy (NV) centre, a substitutional nitrogen atom<br />
adjacent to a carbon vacancy, is the most widely<br />
studied. The NV centre has a broad fluorescence<br />
emission band, centred at approximately 685nm.<br />
The fluorescence <strong>of</strong> an NV can therefore travel<br />
though tissue with limited absorption and is<br />
conveniently beyond the range <strong>of</strong> cellular<br />
aut<strong>of</strong>luorescence. However, several challenges in<br />
using nanodiamonds for biological applications<br />
include the size <strong>of</strong> nanodiamond particle, their<br />
surface impurities and their tendency to aggregate.<br />
To address these difficulties we have established a<br />
series <strong>of</strong> chemical treatments to produce and<br />
isolate a monodisperse population <strong>of</strong> 4nm<br />
nanodiamond particles from material fabricated by<br />
detonation synthesis. This process involves<br />
successive acid washing steps, ultrasonication<br />
and ultracentrifugation. The resulting acid treated<br />
and oxidized nanodiamonds were covalently<br />
attached to poly-L-lysine-FITC via carbodiimide<br />
chemistry as a preliminary study for establishing
the conjugation <strong>of</strong> nanodiamonds to other<br />
biomolecules <strong>of</strong> interest. Our functionalization<br />
approach has lead to our poly-L-lysine-FITC<br />
labeled nanodiamonds being used for labelling <strong>of</strong><br />
macrophage cells and also stem cells. The<br />
long-term photostability <strong>of</strong> the nanodiamonds<br />
provides the ability to image and track biological<br />
cellular processes uploaded with the<br />
nanodiamonds.<br />
Finally, due to the photostability, long lifetime and<br />
near unity quantum yield <strong>of</strong> the NV centre in<br />
nanodiamonds we have been studying their use as<br />
a donor label for FRET. We have investigated the<br />
coupling efficiency between a single NV centre in a<br />
nanodiamond and multiple IRDye-800CW dye<br />
molecules absorbed onto the surface using both<br />
fluorescence spectral intensity and lifetime<br />
measurements. The use <strong>of</strong> the stable NV centre as<br />
the donor molecule prevents the rapid<br />
photobleaching <strong>of</strong> the dye molecule and allows for<br />
distance calculations able to position the NV<br />
centre within the nanodiamond particle.<br />
1.2.4 11:15am – 11:30am<br />
BODIPY phosphatidylinositol probes<br />
incorporation into the membrane <strong>of</strong><br />
giant unilamellar vesicles grown in<br />
carbohydrate and physiological<br />
buffer solutions.<br />
Moens, P.D.J. 2 , D. M. Gau 1,2 , Salvemini, I.L. 2 ,<br />
Reid, J. 2<br />
1 Department <strong>of</strong> Bioengineering, <strong>University</strong> <strong>of</strong> Pittsburgh,<br />
Pittsburgh, PA 15213, USA, recalled@gmail.com<br />
2 School <strong>of</strong> Science & Technology, <strong>University</strong> <strong>of</strong> New England,<br />
Armidale, NSW 2351, Australia<br />
Biography<br />
David Gau, a Rotary Ambassadorial Scholar and<br />
Whitaker Fellow, hails from Pittsburgh,<br />
Pennsylvania, USA. Dave completed his<br />
undergraduate career at <strong>University</strong> <strong>of</strong> Pittsburgh<br />
completing an undergraduate thesis in<br />
bioengineering and degrees in mathematics and<br />
economics. While at PITT, Dave has been involved<br />
in many student organizations and was selected<br />
as valedictorian <strong>of</strong> the 2011 graduating class.<br />
Biographies and Abstracts<br />
Sunday 4 December - Session 1<br />
Alongside, under the guidance <strong>of</strong> Dr. Partha Roy <strong>of</strong><br />
the Bioengineering department <strong>of</strong> PITT, David has<br />
published a paper on identifying Pfn1 and VASP<br />
interaction using FRET and has another paper<br />
under review regarding effects <strong>of</strong> Pfn1-VASP<br />
interaction in breast cancer cell migration. He is<br />
now working under Dr. Pierre Moens <strong>of</strong> <strong>University</strong><br />
<strong>of</strong> New England and continuing research on Pfn1<br />
interaction with other proteins and how it relates to<br />
cancer invasion.<br />
Abstract<br />
The use <strong>of</strong> Giant Unilamellar Vesicles (GUVs)<br />
composed <strong>of</strong> fluorescently labelled lipid analogues<br />
has become an increasingly popular model to<br />
study both structural and complex biophysical<br />
properties <strong>of</strong> bilayers. However, there is a common<br />
assumption that the number <strong>of</strong> probes<br />
incorporated into the membrane <strong>of</strong> the GUVs is<br />
proportional to the mole fraction (%) <strong>of</strong> these lipid<br />
molecules in the original solvent solution. To test<br />
this assumption, a commercial confocal laser<br />
scanning microscope (Nikon C1) was used to<br />
obtain single point fluorescence correlation<br />
spectroscopy (FCS) data.<br />
We measured the diffusion coefficient and number<br />
<strong>of</strong> molecules incorporated into the membrane <strong>of</strong><br />
the GUVs for several BODIPY labelled lipid i.e.<br />
BODIPY TMR-phosphatidylinositol (4,5)<br />
bisphosphate, BODIPY TR- phosphatidylinositol<br />
(4,5) bisphosphate and BODIPY (530/550)<br />
hexadecanoyl-sn-glycero-3-phosphocholine. We<br />
investigated the effect <strong>of</strong> various mole fraction <strong>of</strong><br />
these lipids and compared the results with<br />
(1,1’-dioctadecyl-3,3,3’,3’tetramethylindocarbocyanine<br />
perchlorate [DiIC18]<br />
when grown in carbohydrate and physiological<br />
buffer solutions.<br />
We show that the number <strong>of</strong> DiIC18 molecules<br />
incorporated into the membrane <strong>of</strong> the GUVs<br />
(formed by the electr<strong>of</strong>ormation method) is in<br />
agreement with the expected number <strong>of</strong><br />
molecules calculated from the mole fraction <strong>of</strong> the<br />
organic stock solution. However, we find that the<br />
actual proportion <strong>of</strong> β-BODIPY-HPC, TR-PI(4,5)P2,<br />
and TMR-PI(4,5)P2 incorporated into the bilayer is<br />
highly variable and appear significantly less than<br />
the proportion <strong>of</strong> these lipids in the organic solvent<br />
stock solution. This apparently low incorporation is<br />
29
30<br />
Biographies and Abstracts<br />
Sunday 4 December - Session 1<br />
observed regardless <strong>of</strong> the solutions (carbohydrate<br />
or physiological buffer) used when growing the<br />
GUVs. These variations in incorporation can be<br />
explained by the formation <strong>of</strong> a blue fluorescent<br />
species which is probably due to the formation <strong>of</strong><br />
dimers <strong>of</strong> the BODIPY labelled lipids.<br />
1.2.5 11:30am – 11:45am<br />
Monitoring the B to A conformation<br />
transition <strong>of</strong> DNA in functional cells<br />
using Fourier transform infrared<br />
spectroscopy<br />
Donna R. Whelan1 , Keith R. Bambery 1 , Philip<br />
Heraud1,2 , Mark J. Tobin 3 , Don McNaughton 1 and<br />
Bayden R. Wood1 .<br />
1 Center for Biospectroscopy and School <strong>of</strong> Chemistry, Monash<br />
<strong>University</strong>, Clayton, Victoria, 3800<br />
2 Monash Immunology and Stem Cell Laboratories, Monash<br />
<strong>University</strong>, Clayton, Victoria, 3800<br />
3 Australian Synchrotron, 800 Blackburn Road, Clayton, Victoria<br />
3168, Australia<br />
Biography<br />
Donna Whelan is currently completing a PhD with<br />
the School <strong>of</strong> Chemistry at Monash <strong>University</strong><br />
concentrating on the elucidation <strong>of</strong> DNA<br />
conformation and architecture within live cells<br />
using Fourier transform infrared spectroscopy and<br />
fluorescence microscopy methods. In 2010 she<br />
successfully completely her Honours year and<br />
published her research on the B to A transition <strong>of</strong><br />
DNA in cells in ‘Nucleic Acids Research’. She has<br />
recently returned from a visit to Imperial College in<br />
London where she conducted further research on<br />
this topic under Pr<strong>of</strong>. Sergei Kazarian.<br />
Abstract<br />
The structure <strong>of</strong> DNA inside cells is a central<br />
aspect <strong>of</strong> research into the normal and diseased<br />
state. In the past, the conformation <strong>of</strong> DNA inside<br />
cells has been hypothesized as predominantly<br />
B-like with temporary transitions <strong>of</strong> small sections<br />
to alternate conformations. Using Fourier transform<br />
infrared (FTIR) spectroscopy we have been able to<br />
detect the conformation <strong>of</strong> DNA within live cells<br />
and monitor an overall B- to A-like transition during<br />
the dehydration <strong>of</strong> several cell types including<br />
avian erythrocytes, mammalian fibroblasts and<br />
bacterial strains. Upon rehydration the A-like DNA<br />
reverts to the B-like conformation observed in live<br />
cells. Changes in band pr<strong>of</strong>ile to both the<br />
symmetric phosphate stretch (1087 cm-1) and the<br />
C-O stretch (1052 cm-1), and shifts in the<br />
antisymmetric phosphate stretch (1225 – 1237<br />
cm-1) and the C-C stretch (970 – 966 cm-1)<br />
among others, were identified as diagnostic <strong>of</strong> this<br />
conformational transition. This indicates an<br />
important step forward in understanding the role<br />
<strong>of</strong> A-like DNA inside cells and the mechanism by<br />
which some dehydrated cells, including the<br />
bacteria examined, can return to a functional state.<br />
Furthermore, we demonstrate that by applying<br />
FTIR spectroscopy to hydrated samples, sharp<br />
DNA bands can be used to approximate DNA<br />
concentration. This is anticipated as enabling<br />
differentiation <strong>of</strong> cancerous from non-cancerous<br />
cells based on the increased DNA content inherent<br />
to dysplastic and neoplastic tissue.<br />
1.2.6 11:45am – 12:00pm<br />
The Structure <strong>of</strong> Mixed<br />
Lipopolysaccharide/Porin<br />
Monolayers at the Air-Liquid<br />
Interface<br />
Anton P. Le Brun1 , Luke A. Clifton2 ,<br />
Christopher L. Johnson3 , Jeremy H. Lakey3 and<br />
Stephen A. Holt1 1 Bragg Institute, Australian Nuclear Science and Technology<br />
Organisation, Locked Bag 2001, Kirrawee DC, NSW 2232,<br />
Australia.<br />
2 ISIS Neutron Facility, STFC Rutherford Appleton Laboratory,<br />
Harwell Science and Innovation Campus, Didcot, Oxfordshire,<br />
OX11 0QX, United Kingdom.<br />
3 Institute for Cell and Molecular Biosciences, The Medical School,<br />
<strong>University</strong> <strong>of</strong> Newcastle upon Tyne, Framlington Place,<br />
Newcastle upon Tyne, NE2 4HH, United Kingdom.<br />
Biography<br />
Anton Le Brun is a post-doctorial research fellow<br />
in the Bragg Institute and National Deuteration<br />
Facility, ANSTO. His research interests are in<br />
membrane structural biology with a particular<br />
focus for studying membrane proteins and lipid<br />
bilayers using neutron reflectometry. He completed<br />
his bachelors and masters degrees in
iochemistry at the <strong>University</strong> <strong>of</strong> York. Anton went<br />
onto study for a PhD in biophysics in Pr<strong>of</strong>essor<br />
Jeremy Lakeyâ€s laboratory at the <strong>University</strong> <strong>of</strong><br />
Newcastle upon Tyne. His thesis describes<br />
characterising model membranes based on the<br />
outer membrane <strong>of</strong> the bacterial Gram-negative<br />
cell envelope, work which he now continues to<br />
further at ANSTO.<br />
Abstract<br />
The Gram-negative bacterial cell envelope is a<br />
complex structure. It consists <strong>of</strong> a cytoplasmic<br />
inner membrane, the periplasm and an outer<br />
membrane. The outer membrane composes <strong>of</strong> a<br />
phospholipid inner leaflet and an outer leaflet <strong>of</strong><br />
lipopolysaccharide as well as integral membrane<br />
proteins. The challenge in studying the structures<br />
and functions at the outer membrane is in creating<br />
simple, but yet representative models <strong>of</strong> the<br />
membrane. Current models consist <strong>of</strong> solidsupported<br />
bilayers containing the lipid and protein<br />
components <strong>of</strong> the bacterial outer membrane [1].<br />
Although this makes for a good representation <strong>of</strong> a<br />
bilayer the fluidity <strong>of</strong> the lipid layers is not truly<br />
represented. By depositing mixed monolayers <strong>of</strong><br />
lipid and protein at the air-liquid interface on a<br />
Langmuir trough, a monolayer that represents the<br />
fluidity and outer surface <strong>of</strong> a membrane can be<br />
achieved. Monolayers <strong>of</strong> the anionic lipid DPPG<br />
and outer membrane protein F (OmpF) from E. coli<br />
have been successfully deposited and<br />
characterised [2]. Whilst these monolayers model<br />
the negative charge characteristics and fluidity <strong>of</strong> a<br />
bacterial outer membrane, DPPG is not a<br />
representative lipid <strong>of</strong> the outer membrane<br />
surface. This work has been extended to make a<br />
more representative outer membrane surfaces by<br />
using lipopolysaccharide (LPS) as the lipid<br />
component. Monolayers <strong>of</strong> LPS and OmpF were<br />
deposited onto an aqueous surface using a vesicle<br />
rupture method first described by Schindler and<br />
colleagues [3]. The layers were characterised<br />
using neutron reflectometry. The ability to<br />
discriminate between hydrogen (a weak scatter)<br />
and its isotope deuterium (a strong scatter) in<br />
neutron scattering makes neutron reflectometry a<br />
powerful tool for probing the layers along the axis<br />
perpendicular to the plane <strong>of</strong> the membrane.<br />
Production <strong>of</strong> deuterated versions <strong>of</strong> the<br />
molecules to be studied allows the lipid, protein<br />
Biographies and Abstracts<br />
Sunday 4 December - Session 1<br />
and solvent components <strong>of</strong> the membrane layer to<br />
be deduced. In this case we used bio-deuteration<br />
methods to produce deuterated OmpF and<br />
deuterated LPS from E. coli. The deposited<br />
material forms stable monolayers as shown by<br />
reproducible pressure-area isotherms. By using<br />
different combinations <strong>of</strong> deuterated and<br />
hydrogenated material a detailed picture <strong>of</strong> the<br />
monolayer is achieved. Finally, colicin N is a<br />
pore-forming E. coli toxin produced by E. coli cells<br />
to kill competing cells when nutrients are scarce.<br />
Colicin N uses OmpF as it receptor and<br />
translocator to cross the outer membrane. The<br />
translocation <strong>of</strong> colicin N is tracked across the<br />
LPS/OmpF monolayer using neutron reflectometry.<br />
References<br />
1. S. A. Holt et al (2009) S<strong>of</strong>t Matter 5:2576-2586.<br />
2. L. A. Clifton et al (2011) Structure submitted.<br />
3. H. Schindler et al (1978) Proc. Natl. Acad. Sci. USA<br />
75:3751-3755.<br />
31
32<br />
Biographies and Abstracts<br />
Sunday 4 December - Session 2<br />
Session 2 – Theatre 2<br />
2.1.1 1:15pm – 1:45pm<br />
Isomerization and Decomposition<br />
Chemistry <strong>of</strong> C7Hn (n = 5, 7)<br />
Radicals<br />
Gabriel da Silva<br />
Chemical and Biomolecular Engineering, The <strong>University</strong> <strong>of</strong><br />
Melbourne, Parkville 3010, Australia<br />
Biography<br />
Dr. Gabriel da Silva is a Lecturer in the Department<br />
<strong>of</strong> Chemical and Biomolecular Engineering at the<br />
<strong>University</strong> <strong>of</strong> Melbourne. After completing a Ph.D.<br />
in Chemical Engineering at the <strong>University</strong> <strong>of</strong><br />
Newcastle, Gabriel worked as a postdoctoral<br />
scholar in Chemistry and Enviornmental Science<br />
at NJIT, before moving to Melbourne in 2007.<br />
Abstract<br />
Benzyl is a cyclic C7H7 resonance-stabilized<br />
radical (RSR) that is widely encountered in flames<br />
and in numerous other reacting systems. In<br />
combustion, reaction chemistry <strong>of</strong> the benzyl<br />
radical plays a key role in the oxidation <strong>of</strong> aromatic<br />
fuels (such as toluene) and in the formation <strong>of</strong><br />
polycyclic aromatic hydrocarbon (PAH) pollutants<br />
and soot particles. Computational chemistry and<br />
statistical reaction rate theory have been used to<br />
model the mechanism and kinetics <strong>of</strong> benzyl<br />
radical decomposition, suggesting that the<br />
predominant reaction products are the C7H6<br />
species fulvenallene (+H) and the RSR<br />
cyclopentadienyl (+ HCCH). Benzyl can also<br />
isomerize to form the RSRs tropyl and<br />
vinylcyclopentadienyl; the latter is almost as stable<br />
as tropyl but much less widely known. Chemically<br />
activated bimolecular reactions on the C7H7<br />
surface, relevant to combustion and atmospheric<br />
chemistry, will also be discussed. The effect <strong>of</strong><br />
methyl substitution in benzyl (resulting in C8H9<br />
methyl-benzyl radicals) is also examined.<br />
The benzyl decomposition product fulvenallene is<br />
shown to readily decompose at flame<br />
temperatures to yield the fulvenallenyl radical<br />
(C7H5), a novel RSR that shares properties <strong>of</strong><br />
cyclopentadienyl and propargyl. Rate constant<br />
calculations for fulvenallenyl decomposition<br />
identify a large overall reaction barrier, making the<br />
closed-shell species fulvenallene approximately as<br />
stable as its open-shell parent (C7H7) and<br />
daughter (C7H5) radicals. Fulvenallenyl<br />
decomposition proceeds via a complex reaction<br />
mechanism, forming the i/n-C5H3 (+ HCCH) and<br />
C3H3 (+ C4H2) RSRs as the major products. The<br />
theoretically proposed reaction products are<br />
consistent with products identified in threhold<br />
photoionization mass spectra for C7H5 radical<br />
pyrolysis obtained using VUV synchrotron<br />
radiation. The C7H5 energy surface can also be<br />
accessed via the reverse C5H3 + C2H2 and C3H3<br />
+ C4H2 reactions, as well as the near-barrierless<br />
reactions <strong>of</strong> several C7H4 polyynes with H,<br />
reactions which are, again, <strong>of</strong> interest to<br />
combustion scientists and astrochemists. Finally,<br />
incorporating the theoretical results presented<br />
here into a detailed kinetic model for toluene<br />
pyrolysis is shown to result in significant<br />
improvements in predicted radical concentrations,<br />
as well as pointing to a role for fulvenallenyl in PAH<br />
chemistry.<br />
2.1.2 1:45pm - 2:00pm<br />
Cross-Strand Disulfides - Poised to<br />
Act<br />
Naomi L Haworth 1 , Merridee A Wouters2 1 School <strong>of</strong> Life and Environmental Sciences, Deakin <strong>University</strong>,<br />
Geelong, Victoria, 3217, naomi.haworth@deakin.edu.au<br />
2 School <strong>of</strong> Life and Environmental Sciences, Deakin <strong>University</strong>,<br />
Geelong, Victoria, 3217, m.wouters@deakin.edu.au<br />
Biography<br />
Dr Haworth completed her BSc (Hons) in<br />
Chemistry and Quantum Physics at the <strong>University</strong><br />
<strong>of</strong> Melbourne. Her PhD in Theoretical Chemistry<br />
followed. This was under Dr George Bacskay at<br />
the <strong>University</strong> <strong>of</strong> Sydney and focussed on highly<br />
accurate calculations <strong>of</strong> the thermochemistry <strong>of</strong><br />
small molecules. After a post-doc in the same field<br />
with Pr<strong>of</strong>essor Leo Radom, she changed direction<br />
to look at quantum chemical and biophysical<br />
studies <strong>of</strong> proteins. This has included a Humboldt<br />
Fellowship under Pr<strong>of</strong> Tim Clarke at Erlangen and<br />
her current position at Deakin <strong>University</strong> in<br />
Melbourne.
Abstract<br />
Cross-strand disulfides (CSDs) link cysteine (Cys)<br />
residues across adjacent strands <strong>of</strong> b-sheets. As<br />
the strands are already linked by H-bonding, CSDs<br />
appear at best to be redundant. Initially the<br />
formation <strong>of</strong> such disulfides was predicted to be<br />
forbidden, producing too much strain in the protein<br />
fold.(1) Nevertheless, CSDs do exist in nature.(2)<br />
As disulfides in strained environments have the<br />
potential to be involved in redox processes, CSDs<br />
may perform important biological roles.<br />
There are three protein environments in which a<br />
CSD can form. In antiparallel b-sheet, the two Cys<br />
can be in a non-H-bonding site (aCSDn) or in an<br />
H-bonding site (aCSDn). CSDs can also be found<br />
in parallel b-sheet (pCSD).<br />
Almost all CSDs adopt one <strong>of</strong> two disulfide<br />
conformations: the right-handed staple (RHSt) and<br />
the left-handed saddle/cis (LHC). When the Cys<br />
residues are involved in H-bonding across the<br />
b-ladder (aCSDhs) an LHC conformation is usually<br />
seen, whereas when the Cys do not form H-bonds<br />
with their b-partners (aCSDns) an RHSt<br />
conformation is almost always formed. In pCSDs,<br />
only one Cys forms H-bonds across the b-ladder<br />
while the other is non-H-bonding. This results in a<br />
mix between RHSt and LHC conformations.<br />
The CSD types and conformations interact<br />
differently with the surrounding b-sheet.<br />
Antiparallel b-sheets have positive sheet twist and<br />
shear. Parallel sheets also have positive twist but<br />
are not sheared. aCSDns with RHSt<br />
conformations generate positive twist and shear<br />
between the two Cys. The values match well with<br />
the natural sheet deformation, thus condensation<br />
<strong>of</strong> these disufides does not cause significant<br />
additional strain. In contrast, aCSDhs with LHC<br />
conformations generate positive twist but negative<br />
shear. The sheet must therefore experience<br />
significant disruption to allow the disulfide to form.<br />
This involves buckling and tilting <strong>of</strong> the b-strands<br />
as well as breaking <strong>of</strong> H-bonds in some cases.<br />
pCSDs <strong>of</strong> either conformation generate positive<br />
twist as well as large positive shear. Thus oxidation<br />
<strong>of</strong> pCSDs also results in significant sheet strain.<br />
One <strong>of</strong> the most important CSDs in nature is an<br />
aCSDh found in ribonuclease reductases <strong>of</strong> all<br />
organisms (including viruses). This highly strained<br />
Biographies and Abstracts<br />
Sunday 4 December - Session 2<br />
disulfide is responsible for the reduction <strong>of</strong><br />
ribonucleotides to deoxyribonucleotides and is<br />
therefore essential for all life.(3)<br />
1. Richardson, JS (1981) Adv. Protein Chem. 34, 167.<br />
2. Wouters, MA, George, RA, Haworth, NL (2007) Curr.<br />
Prot. Pept. Sci. 8, 484.<br />
3. Eriksson, M, et al. (1997) Structure 5, 1077.<br />
2.1.3 2:00pm – 2:15pm<br />
Computational Design <strong>of</strong> Metal-<br />
Based Systems for the<br />
Functionalization <strong>of</strong> Small Molecules<br />
<strong>of</strong> Synthetic Interest<br />
Germán E Cavigliasso 1 , Robert Stranger1 ,<br />
Brian F Yates 2<br />
1 Research School <strong>of</strong> Chemistry, Australian National <strong>University</strong>,<br />
Canberra, Australia<br />
2 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Tasmania, Hobart, Australia<br />
Biography<br />
Germán E Cavigliasso is currently a research<br />
fellow and lecturer at the Australian National<br />
<strong>University</strong>, with principal interests in computational<br />
chemistry and transition metal systems. He carried<br />
out undergraduate studies in engineering,<br />
physical, and chemical sciences in Argentina, and<br />
postgraduate and doctoral work in computational<br />
chemistry at the <strong>University</strong> <strong>of</strong> British Columbia, the<br />
<strong>University</strong> <strong>of</strong> Hull, and the <strong>University</strong> <strong>of</strong> Cambridge.<br />
He was a postdoctoral fellow at the Australian<br />
National <strong>University</strong> and <strong>University</strong> College London.<br />
Abstract<br />
Cleavage <strong>of</strong> cyanide is more difficult to achieve<br />
compared to dinitrogen and carbon monoxide,<br />
even though these species contain triple bonds <strong>of</strong><br />
greater strength. In this work, we have used<br />
computational methods to investigate<br />
thermodynamic and mechanistic aspects <strong>of</strong> the<br />
C-N bond cleavage process in [L3M-CN-M’L3]<br />
systems consisting <strong>of</strong> a central cyanide unit bound<br />
in an end-on fashion to two terminal metal<br />
tris-amide complexes. The general structural,<br />
bonding, and thermochemical trends across the<br />
transition metal series investigated indicate that<br />
the systems exhibiting the greatest degree <strong>of</strong> C-N<br />
activation, and most favourable energetics for C-N<br />
33
34<br />
Biographies and Abstracts<br />
Sunday 4 December - Session 2<br />
cleavage, also possess the most favourable<br />
electronic properties, namely, a close match<br />
between the relevant p-like orbitals on the<br />
metal-based and cyanide fragments. Therefore,<br />
metal-based systems with high-lying dp orbitals<br />
are best suited to C-N cleavage. In terms <strong>of</strong><br />
chemical periodicity, these systems can be<br />
identified as the heavier members within a group<br />
and the earlier members within a period. As a<br />
consequence, Mo complexes (which under<br />
experimental conditions have been successfully<br />
used for N-N bond scission in dinitrogen, but have<br />
failed in the case <strong>of</strong> cyanide) are not well suited to<br />
cleaving the C-N bond, whereas the Ta analogues<br />
are the most favourable systems and should, in<br />
principle, be capable <strong>of</strong> cleaving cyanide under<br />
relatively mild conditions. An important conclusion<br />
from this work is that a successful strategy for<br />
achieving cleavage <strong>of</strong> multiply-bonded, and<br />
relatively unreactive, molecular fragments, may lie<br />
in tuning the electronic structures and orbital<br />
interactions by judicious choice <strong>of</strong> metal sites and<br />
ligand groups.<br />
2.1.4 2:15pm – 2:30pm<br />
Infrared Spectroscopy: from<br />
Conformers to Clouds<br />
Evan G Robertson 1 , Isabella Antony Lobo1 ,<br />
Chris Medcraft 2 , Chris Thompson2 , Don<br />
McNaughton2 , Dominique Appadoo3 .<br />
1 Department <strong>of</strong> Chemistry, La Trobe <strong>University</strong>, Bundoora,<br />
Victoria, 3086. e.robertson@latrobe.edu.au, ialobo@student.<br />
latrobe.edu.au,<br />
2 School <strong>of</strong> Chemistry, Monash <strong>University</strong>, Victoria, 3800. chris.<br />
medcraft@monash.edu, chris.thompson@monash.edu, Donald.<br />
mcnaughton@monash.edu<br />
3 Australian Synchrotron, Blackburn Rd, Clayton, Victoria 3168,<br />
Australia. Dominique.Appadoo@synchrotron.org.au<br />
Biography<br />
Evan Robertson studied for his PhD with Don<br />
McNaughton at Monash university, before a 5 year<br />
post-doctoral stint with John Simons at the<br />
<strong>University</strong> <strong>of</strong> Oxford. He returned to Monash<br />
<strong>University</strong>, working first as an ARC fellow from<br />
2001 and then as lecturer from 2006. In 2009, he<br />
moved to La Trobe <strong>University</strong> where he has<br />
continued to pursue his research interests as a<br />
colour blind spectroscopist.<br />
Abstract<br />
IR spectroscopy, with its exquisite sensitivity to<br />
molecular structure and the intramolecular<br />
bonding environment, is ideally suited to<br />
investigating a range <strong>of</strong> chemical and physical<br />
problems. Some diverse applications arising from<br />
specialised techniques used in our research group<br />
will be highlighted:<br />
1. Conformational studies <strong>of</strong> neurotransmitters<br />
have been conducted using IR-UV ion depletion<br />
technique experiments based on nanosecond<br />
pulsed lasers. E.g. Conformer-selective IR spectra<br />
measured for amino-p-phenethylamine (APEA)<br />
allow the two most populated conformers to be<br />
unambiguously identified as those having a<br />
gauche arrangement <strong>of</strong> the side chain which<br />
facilitates an NH…p type hydrogen bond.<br />
The pharmaceutical tranylcypromine is another<br />
subject for these studies.<br />
2. Rotationally resolved, high resolution FTIR<br />
spectroscopy has been applied to molecules <strong>of</strong>
atmospheric relevance such as CFC pollutant<br />
dichlorodifluoromethane (R12) or interstellar<br />
interest such as propynal. Some samples requires<br />
cooling in order to obtain spectra suitable for<br />
rovibrational analysis, and a specialised collisional<br />
cooling cell has been employed to this end.<br />
Measurements in the far-IR region benefit<br />
considerably from synchrotron source radiation.<br />
3. Atmospheric aerosols scatter and absorb<br />
radiation, influence cloud formation and play a role<br />
in heterogeneous chemical reactions in the<br />
atmosphere, affecting the concentration and<br />
distribution <strong>of</strong> atmospheric trace gases. Also, from<br />
the perspective <strong>of</strong> climate science and modelling<br />
the far IR region is very important yet has been<br />
relatively little studied. Combining these two<br />
elements, a collisional cooling cell capable <strong>of</strong><br />
generating aerosols has been modified from the<br />
design <strong>of</strong> Bauerecker to extend its optical range<br />
into the far-infrared where the synchrotron source<br />
has the greatest advantage over thermal sources.<br />
This unique facility has been used to measure the<br />
first far IR spectra <strong>of</strong> water aerosols, accessing<br />
spectral bands with implications that extend to<br />
non-terrestrial environments.<br />
Biographies and Abstracts<br />
Sunday 4 December - Session 2<br />
2.1.5 2:30pm – 2:45pm<br />
Photodetachment <strong>of</strong> Small Dianions:<br />
Adventures in Mass and Charge<br />
Stephen J. Blanksby1 , Celli Lloyd1 ,<br />
Pramesh I. Hettiarachachi 1 , Benjamin B. Kirk1 ,<br />
Adam J. Trevitt 1<br />
1. ARC Centre <strong>of</strong> Excellence for Free Radical Chemistry and<br />
Biotechnology, School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong><br />
NSW, Australia.<br />
Biography<br />
Dr Blanksby received his PhD from the <strong>University</strong><br />
<strong>of</strong> Adelaide (1999) in the field <strong>of</strong> organic mass<br />
spectrometry before undertaking postdoctoral<br />
research at the <strong>University</strong> <strong>of</strong> Colorado (Boulder) in<br />
gas phase ion chemistry and spectroscopy. He<br />
returned to Australia in 2002 to commence his<br />
academic appointment at the <strong>University</strong> <strong>of</strong><br />
<strong>Wollongong</strong>.<br />
Abstract<br />
The advent <strong>of</strong> electrospray ionisation mass<br />
spectrometry has allowed the routine generation <strong>of</strong><br />
multiply charged anions. Coupling <strong>of</strong> electrospray<br />
ionisation sources with negative ion photoelectron<br />
spectroscopy has given deep insight into the<br />
intrinsic stabilisation <strong>of</strong> dianions; most notably<br />
characterisation <strong>of</strong> the contribution <strong>of</strong> the reverse<br />
Coulomb barrier to electron binding in such<br />
systems [1]. Another related but less well<br />
understood phenomenon is the observation <strong>of</strong><br />
zero-kinetic energy electrons arising from the<br />
photodetachment <strong>of</strong> small dicarboxylate dianions.<br />
Intriguingly, this electron population is found to be<br />
largely independent <strong>of</strong> both (i) the energy <strong>of</strong> the<br />
incident photon (provided it is greater than the<br />
electron detachment threshold) and (ii) the<br />
separation <strong>of</strong> the charged moieties (e.g., it is<br />
observed for a range <strong>of</strong> dicarboxylate dianions <strong>of</strong><br />
the form -O2C(CH2)nCO22-, where n ≥ 2). It has<br />
been suggested that following photodetachment<br />
<strong>of</strong> the first electron the nascent radical anion<br />
undergoes facile free radical decomposition<br />
resulting in thermal ejection <strong>of</strong> the second electron<br />
and stable even-electron neutrals (e.g., alkenes<br />
and carbon dioxide). One exception to this pattern<br />
has been noted. The photodetachment <strong>of</strong><br />
acetylene dicarboxylate dianion (O2CCCCO2•-)<br />
35
36<br />
Biographies and Abstracts<br />
Sunday 4 December - Session 2<br />
does not give rise to zero-kinetic energy electrons<br />
suggesting that the corresponding radical anion is<br />
stable on the timescale <strong>of</strong> the experiment. We<br />
have recently modified an electrospray ionisation<br />
linear ion-trap mass spectrometer<br />
(ThermoFinnigan LTQ) to allow irradiation <strong>of</strong><br />
mass-selected ions with both fixed frequency<br />
(Continuum, Minilite II) and tuneable (GWU<br />
Versascan) laser sources. Using this platform we<br />
have photodetached a wide range <strong>of</strong> dicarboxylate<br />
dianions and, by measuring the mass-to-charge<br />
ratio <strong>of</strong> the resulting ions, found that<br />
decarboxylation and subsequent free radical<br />
rearrangements <strong>of</strong> the resulting radical anions is<br />
facile. Consistent with predictions based on<br />
photoelectron spectroscopy, the acetylene<br />
dicarboxylate dianion is the one <strong>of</strong> a limited<br />
number <strong>of</strong> systems thus far identified where an ion<br />
corresponding to the residual radical anion is<br />
detected (m/z 112 in the Figure below). Isolation <strong>of</strong><br />
this species for varying increments <strong>of</strong> time reveals<br />
its unimolecular decomposition via loss <strong>of</strong> carbon<br />
dioxide (to form m/z 68) over several hundred<br />
milliseconds. The unique stabilisation <strong>of</strong> this radical<br />
anion relative to other aliphatic and even aromatic<br />
systems will be discussed along with results from<br />
recent investigations <strong>of</strong> photodetachment from<br />
amino acid and small peptide-based dianions<br />
where intact radical anions have also been<br />
observed.<br />
[1] WANG X. B. and WANG L. S., Photoelectron Spectroscopy <strong>of</strong><br />
Multiply Charged Anions. Annual Review <strong>of</strong> Physical Chemistry,<br />
2009. 60: p. 105-126.<br />
2.1.6 2:45pm – 3:00pm<br />
Laser-Based Formation and<br />
Properties <strong>of</strong> Metal Nanoparticles in<br />
Aqueous Solution<br />
Mark A. Buntine1,2 , Yuen-Yang Fong2 , Jason<br />
R. Gascooke 3 , Gregory F. Metha2 , Hugh Harris2 ,<br />
Ashley Mulder1 , Aidon Slaney1 , Sean Long1 , Franca<br />
Jones1 , Max Massi1 , Mark Ogden1 , Bruce Cowie4 ,<br />
Lars Thompson4 1 Department <strong>of</strong> Chemistry, Curtin <strong>University</strong>, GPO Box U1987<br />
Perth WA 6845<br />
2 School <strong>of</strong> Chemistry and Physics, The <strong>University</strong> <strong>of</strong> Adelaide,<br />
Adelaide SA 5005<br />
3 School <strong>of</strong> Chemical and Physical Sciences, The Flinders<br />
<strong>University</strong> <strong>of</strong> South Australia, GPO Box 2100 Adelaide SA 5001<br />
4 Australian Synchrotron, 800 Blackburn Road, Clayton VIC 3168<br />
Biography<br />
Mark Buntine undertook undergraduate studies at<br />
Monash <strong>University</strong> in Melbourne, Australia, and his<br />
Ph.D. with Richard Zare at Stanford <strong>University</strong>,<br />
graduating in 1992. He undertook postdoctoral<br />
studies with Mark Johnson at Yale <strong>University</strong>,<br />
before commencing an independent academic<br />
career at The <strong>University</strong> <strong>of</strong> Adelaide in 1994. He<br />
moved through the ranks to become a full<br />
pr<strong>of</strong>essor in 2007, and was Head <strong>of</strong> Chemistry<br />
from 2003. In 2009 Buntine moved to Curtin<br />
<strong>University</strong> in Perth where he is Head <strong>of</strong> the<br />
Chemistry Department and maintains an active<br />
research program focussed on the use <strong>of</strong> liquid<br />
beam methodologies to explore the molecular<br />
dynamics <strong>of</strong> liquid-vapour phase transitions via<br />
electronic spectroscopy. More recent<br />
experimental work has focussed on exploring the<br />
mechanistic aspects <strong>of</strong> the in situ laser-based<br />
production <strong>of</strong> metal nanoparticles in aqueous<br />
solution. Buntine is a former Chair <strong>of</strong> the RACI<br />
Physical Chemistry Division and President <strong>of</strong> the<br />
South Australian Branch. He is currently<br />
President-Elect <strong>of</strong> the RACI and serves on the<br />
Executive Committee <strong>of</strong> the Australian and New<br />
Zealand Society for Mass Spectrometry.<br />
Abstract<br />
We report on the production and time evolution <strong>of</strong><br />
metal nanoparticle optical properties and size<br />
distributions as a function <strong>of</strong> laser irradiation in
pure water samples and in the presence <strong>of</strong> anionic<br />
and cationic surfactants and ‘encapsulation’<br />
ligands. Our investigations provide a mechanistic<br />
insight into the laser-induced formation kinetics<br />
involved in the in situ metal nanoparticle<br />
production, as well as the electronic structure <strong>of</strong><br />
the surface atoms. Early studies explored the role<br />
<strong>of</strong> surfactant type and concentration on the<br />
longer-term stability <strong>of</strong> metal nanoparticles. More<br />
recent studies employing ligands to encapsulate<br />
the nanoparticles highlight how the electronic<br />
structure <strong>of</strong> the particle surface can be<br />
manipulated. Implications <strong>of</strong> this work in a variety<br />
<strong>of</strong> possible application areas will be discussed.<br />
Session 2 – Main Theatre<br />
2.2.1 1:15pm – 1:30pm<br />
Structure and dynamics <strong>of</strong> a<br />
replisomal macromolecular<br />
assembly<br />
Flynn R. Hill1 , Charikleia Ioannou1 , Marek M.<br />
Koza2 , Agata Rekas3 , <strong>Peter</strong> J. Holden3 , Nigel M.<br />
Kirby4 , Kathleen Wood5 , Nicholas E. Dixon1 ,<br />
Moeava Tehei 1,6 ,<br />
1 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>, <strong>Wollongong</strong>,<br />
NSW.<br />
2 Institut Laue Langevin, Grenoble, France.<br />
3 National Deuteration Facility, Australian Nuclear Science and<br />
Technology Organisation, Menai, NSW.<br />
4 Australian Synchrotron, Clayton, VIC.<br />
5 Bragg Institute, Australian Nuclear Science and Technology<br />
Organisation, Menai, NSW.<br />
6 Australian Institute <strong>of</strong> Nuclear Science and Engineering, Menai,<br />
NSW.<br />
Biography<br />
Flynn is a PhD student at the <strong>University</strong> <strong>of</strong><br />
<strong>Wollongong</strong>’s School <strong>of</strong> Chemistry working on<br />
proteins involved in bacterial DNA replication. He is<br />
co-supervised by Pr<strong>of</strong> Nick Dixon and Dr Moeava<br />
Tehei.<br />
Abstract<br />
The replication <strong>of</strong> genetic material for passing<br />
down from parent to progeny is the most essential<br />
process for the continued existence <strong>of</strong> life on<br />
Earth. Double-stranded DNA is the genetic<br />
material in the majority <strong>of</strong> lifeforms and the two<br />
Biographies and Abstracts<br />
Sunday 4 December - Session 2<br />
parental strands must be separated ahead <strong>of</strong> the<br />
catalytic elongation <strong>of</strong> the complementary<br />
daughter strands. We have investigated the DnaB<br />
helicase enzyme responsible for this strand<br />
separation in Gram-positive bacteria and the DnaI<br />
protein which complexes with DnaB to assist in<br />
loading <strong>of</strong> the helicase onto parental DNA.<br />
Differential deuterium labelling and quasielastic<br />
neutron spectroscopy (QENS) with an instrument<br />
sensitive to hydrogen motions at time scales <strong>of</strong><br />
~10 ps was used to probe the dynamics <strong>of</strong> DnaB,<br />
DnaI and the DnaB-DnaI complex. In this<br />
incoherent scattering technique, the deuterated<br />
protein is effectively invisible and the dynamical<br />
contribution <strong>of</strong> each protein to the complex can be<br />
isolated. Similarly, solvent contrast variation and<br />
deuterium labeling are planned for measurement<br />
<strong>of</strong> the shape-dependent coherent small angleneutron<br />
scattering (SANS) <strong>of</strong> these proteins in<br />
solution, to allow separation <strong>of</strong> the structural<br />
contribution <strong>of</strong> each protein within the complex. To<br />
complement these SANS experiments, we have<br />
performed synchrotron small-angle X-ray<br />
scattering (SAXS) in the presence and absence <strong>of</strong><br />
nucleotide c<strong>of</strong>actors to probe conformational<br />
transitions <strong>of</strong> the helicase ATPase cycle.<br />
The helicase shows high elasticity within the fast<br />
motion time-scale investigated, while its solution<br />
small-angle scattering suggests a structure with<br />
low conformational flexibility at the whole-domain<br />
level and a narrower central channel than is<br />
present in the crystal structure. Taken together,<br />
our results suggest that fast small-scale motions <strong>of</strong><br />
residues lining the DnaB central channel through<br />
which DNA is fed are responsible for its rapid<br />
translocation, rather than large-scale<br />
conformational transitions. These results and<br />
others concerning the mechanism <strong>of</strong> helicase<br />
loading will be presented along with an<br />
introduction to the biophysical techniques<br />
involved.<br />
37
38<br />
Biographies and Abstracts<br />
Sunday 4 December - Session 2<br />
2.2.2 1:30pm – 1:45pm<br />
Resolving Single Molecule<br />
Fibronectin Interactions with<br />
Conducting Polymer Interfaces<br />
using Atomic Force Microscopy<br />
Michael Higgins, Amy Gelmi, Gordon<br />
Wallace<br />
Intelligent Polymer Research Institute (IPRI), <strong>University</strong> <strong>of</strong><br />
<strong>Wollongong</strong>.<br />
Biography<br />
Dr Michael Higgins is an ARC Australian Research<br />
Fellow in the Intelligent Polymer Research Institute,<br />
<strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>. Dr Higgins’s main<br />
interest is on the application <strong>of</strong> Atomic Force<br />
Microscopy to biological systems, including living<br />
cells, model lipid membranes, single ligandreceptor<br />
interactions, individual protein unfolding<br />
and fundamental surface-force interactions. He<br />
has unique skills in instrument development, probe<br />
modifications and their use in imaging and probing<br />
biomolecular and cellular interaction forces.<br />
Abstract<br />
In this presentation, we focus on biomedical<br />
applications involving the emerging use <strong>of</strong><br />
conducting polymers as electrodes for controlling<br />
in vitro cell culture systems (e.g. enhanced cell<br />
proliferation and differentiation) or in vivo<br />
implantation for neural prosthesis applications.<br />
Conducting polymers represent a new generation<br />
<strong>of</strong> neural electrodes that can uniquely operate by<br />
simultaneously delivering bioregulative cues (e.g.<br />
expulsion <strong>of</strong> drugs) and electrical stimulation. For<br />
example, neurite outgrowth on these polymers can<br />
be dramatically enhanced by applying a voltage to<br />
modulate the redox state <strong>of</strong> the polymer. At<br />
present, the underlying mechanisms for the<br />
cell-material interactions are unknown and require<br />
extensive characterization in order to develop<br />
these polymers for use as implantable electrodes<br />
and drug release devices.<br />
We have recently incorporated extracellular matrix<br />
(ECM) components into a conducting polymer with<br />
the aim <strong>of</strong> improving electrode biocompatibility<br />
through the exposure <strong>of</strong> RGD peptide cell binding<br />
regions. Another important focus <strong>of</strong> our research<br />
is the adsorption <strong>of</strong> fibronectin, a major cell<br />
adhesion molecule which binds to the ECM and is<br />
critical for mediating cellular interactions with our<br />
materials. The presence <strong>of</strong> ECM and/or accretion<br />
<strong>of</strong> cell adhesion molecules at these polymer<br />
surfaces are very important for conditioning the<br />
electrode-cell interface to promote favourable<br />
interactions such as intimate contact with nerve<br />
cells while avoiding unwanted foreign body<br />
responses.<br />
To understand the interaction <strong>of</strong> fibronectin with<br />
conducting polymers, we have functionalized<br />
Atomic Force Microscopy (AFM) probes to directly<br />
measure the interaction forces between fibronectin<br />
and conducting polymers-ECM (hyaluronic acid<br />
and chondroitin sulfate) composites. We will<br />
demonstrate the use <strong>of</strong> in situ electrochemical<br />
AFM to study the fibronectin-polymer interaction in<br />
response to electrical stimulation applied through<br />
the polymer. The presentation will highlight several<br />
major findings including the ability to resolve single<br />
fibronectin protein interactions with the polymer.<br />
We will show differences in the conformation,<br />
unfolding force pr<strong>of</strong>iles and binding probabilities <strong>of</strong><br />
the protein as a function <strong>of</strong> the polymer surface<br />
chemistries. The ability to reversibly modulate the<br />
fibronectin adhesion by applying electrical<br />
stimulation to the polymer electrode will also be<br />
presented. We will finally give insight into the<br />
possible mechanisms and implications <strong>of</strong> the<br />
fibronectin-conducting polymer interactions.<br />
2.2.3 1:45pm – 2:00pm<br />
Engineering new catalytic activities<br />
in enzymes through modifying the<br />
conformational landscape:<br />
experimental and theoretical<br />
insights<br />
Colin J Jackson1 1 Research School <strong>of</strong> Chemistry, Australian National <strong>University</strong>,<br />
ACT, 0200, cjackson@rsc.anu.edu.au<br />
Biography<br />
Colin Jackson received a Bsc (Hons) in<br />
biochemistry from the <strong>University</strong> <strong>of</strong> Otago, New<br />
Zealand, before completing his PhD at the
Australian National <strong>University</strong>. From 2008 he<br />
worked at the CSIRO as a research team leader.<br />
He has recently finished a Marie Curie research<br />
fellowship at the Institut de Biologie Structurale in<br />
Grenoble, France where he has continued to use<br />
laboratory evolution and structural biology to<br />
understand the how new catalytic functions can<br />
evolve in enzymes. He now leads an independent<br />
research group at the Australian National<br />
<strong>University</strong> that uses a range <strong>of</strong> techniques to<br />
investigate molecular evolution and catalysis.<br />
Abstract<br />
Conformational plasticity is known to be an integral<br />
aspect <strong>of</strong> enzyme function, allowing enzymes to<br />
meet the different demands <strong>of</strong> substrate binding,<br />
catalysis and product release. Now that advances<br />
in computational and experimental structural<br />
biology has made characterization <strong>of</strong><br />
conformational fluctuations considerably more<br />
accurate, we seek to understand how<br />
conformational landscapes can be engineered to<br />
suit our needs as designer enzymes. The bacterial<br />
phosphotriesterase serves as an excellent model<br />
system to study conformational sampling: it<br />
catalyzes a simple one-step (SN2) hydrolysis <strong>of</strong> a<br />
phosphotriester, it has been extensively<br />
characterized and a large library <strong>of</strong> mutant<br />
enzymes is available. Our results suggest that<br />
mutations many angstroms from the active site<br />
can modulate activity by changing the relative<br />
energies <strong>of</strong> different conformations, thereby<br />
changing the populations <strong>of</strong> states associated with<br />
different steps in the catalytic cycle. Furthermore,<br />
the sequence <strong>of</strong> mutations that change the<br />
function <strong>of</strong> phosphotriesterase to an arylesterase<br />
involves an initial mutation within the active site<br />
that generates a rare, but highly efficient catalytic<br />
conformation, followed by a series <strong>of</strong> outer-shell<br />
mutations that fine-tune the free-energy landscape<br />
to stabilize this conformation.<br />
Biographies and Abstracts<br />
Sunday 4 December - Session 2<br />
2.2.4 2.00pm – 2.15pm<br />
Towards ab initio Refinement <strong>of</strong><br />
Protein X-ray Crystal Structures:<br />
Interpreting and Correlating<br />
Structural Fluctuations<br />
Jeffrey R. Reimers 3 ,Olle Falklöf1 , Charles<br />
Collyer2 ,<br />
1 School <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> Sydney, Sydney, New<br />
South Wales 2006, Australia, and Department <strong>of</strong> Chemistry,<br />
The <strong>University</strong> <strong>of</strong> Gothenburg, Sweden<br />
2 School <strong>of</strong> Molecular Bioscience, The <strong>University</strong> <strong>of</strong> Sydney,<br />
Sydney, New South Wales 2006, Australia<br />
3 School <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> Sydney, Sydney, New<br />
South Wales 2006, Australia<br />
Biography<br />
Jeffrey Reimers studied spectroscopy under Ian<br />
Ross, thermodynamics under Bob Watts, and<br />
semiclassical dynamics under Eric Heller and Kent<br />
Wilson before starting his career as a research<br />
scientist at The <strong>University</strong> <strong>of</strong> Sydney in 1985. Since<br />
then he has worked extensively with Noel Hush<br />
and Max Crossley studying molecular electronics,<br />
natural and artificial photosynthesis, and chemical<br />
theory, establishing an interdisciplinary research<br />
team studying biochemical processes, scanningtunnelling<br />
microscopy, quantum chemistry, the<br />
quantum mechanics <strong>of</strong> electron-vibration<br />
interactions, nanoparticle and surface structure,<br />
quantum computing, and consciousness<br />
research. His research has received many<br />
distinctions including the award <strong>of</strong> H.G. Smith<br />
Medal <strong>of</strong> the RACI and admission as a Fellow <strong>of</strong><br />
the Australian Academy <strong>of</strong> Science.<br />
Abstract<br />
The refinement <strong>of</strong> protein crystal structures<br />
currently involves the use <strong>of</strong> empirical restraints<br />
and force fields that are known to work well in<br />
many situations but nevertheless yield structural<br />
models with some features that are inconsistent<br />
with detailed chemical analysis and therefore<br />
warrant further improvement. Ab initio electronic<br />
structure computational methods have now<br />
advanced to the point at which they can deliver<br />
reliable results for macromolecules in realistic<br />
times using linear-scaling algorithms. The<br />
replacement <strong>of</strong> empirical force fields with ab initio<br />
39
40<br />
Biographies and Abstracts<br />
Sunday 4 December - Session 2<br />
methods in a final refinement stage could allow<br />
new structural features to be identified in complex<br />
structures, reduce errors and remove<br />
computational bias from structural models. In<br />
contrast to empirical approaches, ab initio<br />
refinements can only be performed on models that<br />
obey basic qualitative chemical rules, imposing<br />
constraints on the parameter space <strong>of</strong> existing<br />
refinements, and this in turn inhibits the inclusion<br />
<strong>of</strong> unlikely structural features. Here we focus on<br />
methods for determining an appropriate ensemble<br />
<strong>of</strong> initial structural models for an ab initio X-ray<br />
refinement, modeling as an example the highresolution<br />
single-crystal X-ray diffraction data<br />
reported for the structure <strong>of</strong> lysozyme (PDB entry<br />
“2VB1”). The AMBER force field is used in a Monte<br />
Carlo calculation to determine an ensemble <strong>of</strong> 8<br />
structures that together embody all <strong>of</strong> the partial<br />
atomic occupancies noted in the original<br />
refinement, correlating these variations into a set <strong>of</strong><br />
feasible chemical structures while simultaneously<br />
retaining consistency with the X-ray diffraction<br />
data. Subsequent analysis <strong>of</strong> these results<br />
strongly suggests that the occupancies in the<br />
empirically refined model are inconsistent with<br />
protein energetic considerations, thus depicting<br />
the 2VB1 structure as a deep-lying minimum in its<br />
optimized parameter space that actually embodies<br />
chemically unreasonable features. Indeed,<br />
density-functional-theory calculations for one<br />
specific nitrate ion with an occupancy <strong>of</strong> 62%<br />
indicate that water replaces this ion 38% <strong>of</strong> the<br />
time, a result confirmed by subsequent<br />
crystallographic R-factor analysis. It is foreseeable<br />
that any subsequent ab initio refinement <strong>of</strong> the<br />
whole structure would need to locate a globally<br />
improved structure involving significant changes to<br />
2VB1 which correct these identified local structural<br />
inconsistencies.<br />
2.2.5 2.15pm – 2.30pm<br />
Density Functional Theory<br />
Calculations <strong>of</strong> Novel Silicon<br />
Nanosheets<br />
Michelle J.S. Spencer1 , Tetsuya Morishita 2 ,<br />
Masuhiro Mikami2 , Ian K. Snook3 , Yusuke<br />
Sugiyama4 , Hideyuki Nakano4 1 Department <strong>of</strong> Chemistry, La Trobe Institute for Molecular<br />
Science, La Trobe <strong>University</strong>, Bundoora, Victoria 3086, Australia<br />
m.spencer@latrobe.edu.au<br />
2 Nanosystem Research Institute (NRI), National Institute <strong>of</strong><br />
Advanced Industrial Science and Technology (AIST), Central 2,<br />
1-1-1 Umezono, Tsukuba, Ibaraki 305-8568, Japan,<br />
t-morishita@aist.go.jp<br />
3 Applied Physics, School <strong>of</strong> Applied Sciences, RMIT <strong>University</strong>,<br />
Melbourne, Victoria 3001, Australia<br />
4 Toyota Central R&D Labs., Inc., 41-1 Yokomichi, Nagakute, Aichi<br />
480-1192, Japan<br />
Biography<br />
Dr Spencer is leader <strong>of</strong> the computational<br />
materials chemistry group at La Trobe <strong>University</strong>.<br />
Her expertise is in using density functional theory<br />
calculations, and ab initio molecular dynamics<br />
simulations to investigate the chemistry <strong>of</strong> a wide<br />
variety <strong>of</strong> materials. In particular her research<br />
focuses on surface reactions <strong>of</strong> metals, oxides and<br />
semiconductors, with an emphasis on<br />
nanomaterials which have applications as gas<br />
sensors and electronic devices.<br />
Abstract<br />
Silicon is one <strong>of</strong> the most important elements is<br />
our current society, forming the basis <strong>of</strong> most<br />
semiconducting electronic devices. As the drive<br />
intensifies to enhance our current technological<br />
capabilities and miniaturize device sizes, it is<br />
imperative to find materials and structures that<br />
meet the new demands. Nanosheets <strong>of</strong> silicon are<br />
one <strong>of</strong> the more recently discovered<br />
nanostructured morphologies (see for example<br />
[1,2]). The unique properties <strong>of</strong> these quasi 2D<br />
materials make them ideal for a variety <strong>of</strong><br />
applications, including electronic devices, batteries<br />
and sensors. In order for their full potential to be<br />
realised their properties and structural details need<br />
to be determined.<br />
We are using density functional theory calculations<br />
to provide critical information about the structure
and chemistry <strong>of</strong> clean and functionalised<br />
nanosheets (see Fig. 1). Such information is being<br />
used in conjunction with experimental synthesis<br />
[1,3] to tailor the growth and development <strong>of</strong> these<br />
materials.<br />
Fig. 1. Phenyl modified Si nanosheet [6].<br />
Our calculations have shown that the structure <strong>of</strong><br />
clean silicon nanosheets changes as a function <strong>of</strong><br />
thickness. For very thin Si(111) oriented<br />
nanosheets, we have discovered a novel surface<br />
reconstruction classified as Si(111)-2x2 that forms<br />
on the nanosheet surface layers [4]. Functionalising<br />
the nanosheet surface with different atomic or<br />
molecular species has also resulted in varying<br />
structural and electronic properties. In particular,<br />
by varying the surface termination or substitutional<br />
dopant [5], the nanosheet can be changed to a<br />
p- or an n-type semiconductor. For phenyl<br />
modified nanosheets [6] our calculations have<br />
shown that this material retains the sp3 structure<br />
after functionalisation, resulting in a wide (direct)<br />
band gap. Overall these novel materials possess<br />
the unique characteristic that the electronic<br />
properties can be easily altered by exchanging the<br />
molecules or atoms that functionalise the<br />
nanosheet.<br />
1. Y. Sugiyama, H. Okamoto, T. Mitsuoka, T. Morikawa, K.<br />
Nakanishi, T. Ohta, H. Nakano, J. Am. Chem. Soc. 132 (2010)<br />
5946.<br />
2. T. Morishita, K. Nishio, M. Mikami, Phys. Rev. B 77 (2008)<br />
081401(R).<br />
3. H. Okamoto, Y. Kumai, Y. Sugiyama, T. Mitsuoka, K. Nakanishi,<br />
T. Ohta, H. Nozaki, S. Yamaguchi, S. Shirai, H. Nakano, J. Am.<br />
Chem. Soc. 132 (2010) 2710.<br />
4. T. Morishita, M.J.S. Spencer, S.P. Russo, I.K. Snook, M. Mikami,<br />
Chem. Phys. Lett. 506 (2011) 221.<br />
5. T. Morishita, S.P. Russo, I.K. Snook, M.J.S. Spencer, K. Nishio,<br />
M. Mikami, Phys. Rev. B 82 (2010) 045419.<br />
6. M.J.S. Spencer, M. Morishita, M. Mikami, I.K. Snook, Y.<br />
Sugiyama, H. Nakano, Phys. Chem. Chem. Phys. 13 (2011)<br />
15418.<br />
Biographies and Abstracts<br />
Sunday 4 December - Session 2<br />
2.2.6 2:30pm – 2:45pm<br />
Stable Solid Supported Membranes<br />
to probe Membrane-Protein<br />
Interactions<br />
Ingo Koeper1 1 Flinders <strong>University</strong>, School <strong>of</strong> Chemical and Physical Sciences,<br />
GPO Box 2100, Adelaide, SA 5011, inog.koeper@flinders.edu.<br />
au<br />
Biography<br />
Ingo Koeper studied chemistry at the <strong>University</strong> <strong>of</strong><br />
Dortmund. In 2002 he received his PhD from the<br />
<strong>University</strong> <strong>of</strong> Paris VIIn 2002 he joined the<br />
Materials Science Department at the Max Planck<br />
Institute for Polymer Research, Mainz, Germany.<br />
After one year as a post-doc, he became a project<br />
leader, leading an independent research team<br />
working on bi<strong>of</strong>unctional surfaces.<br />
In September 2009, he followed a call to become<br />
lecturer in the School <strong>of</strong> Chemical and Physical<br />
Sciences at Flinders <strong>University</strong>, Adelaide, Australia<br />
Abstract<br />
The tethered bilayer membrane has been shown<br />
to provide an ideal platform to study membrane<br />
related processes. The architecture consists <strong>of</strong> a<br />
lipid bilayer, where the proximal leaflet is covalently<br />
coupled to a solid substrate via a spacer unit. The<br />
solid substrate allows for the use <strong>of</strong> various<br />
surface analytical techniques such as surface<br />
plasmon spectroscopy, impedance spectroscopy,<br />
neutron reflectometry, quartz crystal microbalance<br />
or atomic force microscopy.<br />
We have performed extensive structural<br />
investigations <strong>of</strong> the membrane and probed the<br />
influence <strong>of</strong> the molecular structure <strong>of</strong> the lipids<br />
used on the overall membrane structure and its<br />
functionality.<br />
Furthermore, the platform has been used to probe<br />
the function <strong>of</strong> embedded membrane proteins,<br />
e.g. ion channels as well as the interaction <strong>of</strong><br />
proteins with the lipid bilayer. Here, the tethered<br />
membrane architecture has proven to provide a<br />
very robust system, allowing extensive systematic<br />
studies.<br />
In this presentation, I will summarize finding on the<br />
41
42<br />
Biographies and Abstracts<br />
Sunday 4 December - Session 2<br />
interaction between small globular proteins and<br />
lipid membrane <strong>of</strong> different composition as well as<br />
highlight some new finding on the effect <strong>of</strong> small<br />
molecules on the membrane structure and<br />
function.<br />
2.2.7 2:45pm – 3:00pm<br />
Directions and Results <strong>of</strong> OH Attack<br />
on Nucleic Acids: a Theoretical<br />
Study<br />
Ganna Gryn’ova, Michelle Coote<br />
Biography<br />
PhD Candidate<br />
ARC Centre <strong>of</strong> Excellence for Free-Radical<br />
Chemistry and Biotechnology, Research<br />
School <strong>of</strong> Chemistry, Australian National<br />
<strong>University</strong>, Canberra, ACT 0200, Australia<br />
Phone: +61 2 6125 5411 E-mail: agrynova@rsc.<br />
anu.edu.au<br />
Personal History<br />
2004-2008 B.Sc. in chemistry at the<br />
Dnepropetrovsk National <strong>University</strong>, Ukraine<br />
2008-2009 M.Sc. in chemistry at the<br />
Dnepropetrovsk National <strong>University</strong>, Ukraine<br />
Since 2010 Ph.D. candidate in chemistry at the<br />
Australian National <strong>University</strong><br />
Research interests: computational chemistry,<br />
mechanisms <strong>of</strong> polymer/biopolymer degradation<br />
and their stabilisation<br />
Abstract<br />
Radiation damage to cellular nucleic acids is a<br />
major deleterious event resulting in a cascade <strong>of</strong><br />
harmful consequences. Sources <strong>of</strong> such damage<br />
are very diverse, however hydroxyl radical, being<br />
the most abundant and extremely reactive, is<br />
considered to be one <strong>of</strong> the main degradation<br />
agents.7<br />
Experimental studies show, that hydroxyl radical<br />
reacts with DNA/RNA bases at almost diffusion<br />
controlled rates, and approximately 5-10 times<br />
slower with the sugar moiety. Whereas it can only<br />
abstract hydrogen from (deoxy)ribose ring, various<br />
pathways are suggested for its interaction with the<br />
base moiety: addition to double bonds,<br />
abstraction <strong>of</strong> hydrogen atoms from both ring<br />
atoms and substituents, and finally sequential<br />
electron proton transfer.8 These mechanistic<br />
pathways were intensively investigated both<br />
experimentally and theoretically (Monte Carlo<br />
simulations and quantum chemical calculations),<br />
however the resulting trends in site reactivity in<br />
sugars and in bases are <strong>of</strong>ten quite different.9<br />
Moreover, to the best <strong>of</strong> our knowledge, there is<br />
no evidence in literature for the high-level<br />
computational study <strong>of</strong> the kinetics <strong>of</strong> sugar radical<br />
formation, and available theoretical studies on<br />
bases are conducted with either insufficiently<br />
accurate methodology or inconsistent models.<br />
Present study focuses on the kinetics and<br />
thermodynamics <strong>of</strong> the OH• attack on DNA/RNA.<br />
We apply two levels <strong>of</strong> modelling – free sugars and<br />
bases, as well as nucleotide 3’,5’-diphosphates,<br />
and use G3(MP2)-RAD level <strong>of</strong> theory. Our results<br />
reveal a number <strong>of</strong> interesting trends, including<br />
remarkable correlation between kinetics,<br />
thermodynamics and electronic structure <strong>of</strong><br />
attacked substrates (for example, see Fig. 1).<br />
Comparison <strong>of</strong> the obtained site reactivity trends<br />
with the experimental/MD ones gives a deeper<br />
insight into the protective mechanisms that nature<br />
applies.<br />
Fig. 1 - Calculated reaction Gibbs free energies (kJ mol-1, aqueous<br />
solution, 298.15 K) and rate constants (logarithms) for the<br />
reactions <strong>of</strong> hydroxyl radical with free pyrimidine bases.
Keynote Session<br />
- Main Theatre<br />
K.4 4:20pm – 4:50pm<br />
Using Theory to Reconcile<br />
Experiment: The Search for<br />
Certainty in an Uncertain World<br />
Alan E. Mark1 ,<br />
1 School <strong>of</strong> Chemistry and Molecular Biosciences (SCMB) and<br />
Institute for Molecular Biosciences (IMB), <strong>University</strong> <strong>of</strong><br />
Queensland, Brisbane, QLD 4072, Australia. Email: a.e.mark@<br />
uq.edu.au<br />
Biography<br />
Pr<strong>of</strong>essor Alan Mark originally studied Chemistry/<br />
Biochemistry at Sydney <strong>University</strong>. He obtained his<br />
Ph.D in physical biochemistry at the JCSMR, ANU.<br />
He went on to hold positions at RSC, ANU,<br />
<strong>University</strong> <strong>of</strong> Groningen, The Netherlands and at<br />
ETH-Zurich, Switzerland. In 1998 he was<br />
appointed Pr<strong>of</strong>. Molecular Simulation <strong>University</strong> <strong>of</strong><br />
Groningen. In 2005 he moved to the <strong>University</strong> <strong>of</strong><br />
Queeensland as a Federation Fellow. His primary<br />
interests are in understanding the dynamic and<br />
thermodynamic properties <strong>of</strong> biomolecular<br />
systems at an atomic level. He is associated with<br />
the development <strong>of</strong> the GROMOS and GROMACS<br />
simulation packages as well as the associated<br />
force fields. His group have also performed<br />
pioneering simulations <strong>of</strong> a wide range <strong>of</strong><br />
biological phenomena. He was awarded the<br />
Ruzicka prize in 1998 for his work on peptide<br />
folding, In 2005 he was awarded an honorary<br />
personal Chair at the <strong>University</strong> <strong>of</strong> Groningen.<br />
Abstract<br />
Despite continuing advances in structural biology it<br />
is still not possible to directly observe the energetic<br />
and dynamic properties <strong>of</strong> individual atoms in<br />
biomolecular systems using currently available<br />
experimental techniques. In fact, everything we<br />
know (or think we know) regarding biomolecular<br />
systems is to some degree based on a theoretical<br />
or structural model. The question is to what extent<br />
can these models be trusted? For example, while<br />
the overall structure <strong>of</strong> a protein may be resolved in<br />
near atomic detail, the position, orientation and/or<br />
conformation <strong>of</strong> a small molecular ligand (c<strong>of</strong>actor,<br />
Biographies and Abstracts<br />
Sunday 4 December - Session 2<br />
substrate, inhibitor etc.) bound to such a protein is<br />
<strong>of</strong>ten much less certain.1 Electrostatics will <strong>of</strong>ten<br />
determine the interaction between a protein and its<br />
ligand but are generally ignored in structure<br />
refinement. In other cases there is evidence <strong>of</strong><br />
systematic bias in how data is interpreted. The<br />
variation in the area per lipid in theoretical<br />
calculations <strong>of</strong> membrane systems is much less<br />
than the variation in the experimental data on<br />
which they are based.2 This is not only a problem<br />
for experimentalists but represent a fundamental<br />
challenge to theoreticians attempting to validate<br />
computational models.3 The talk will illustrate how<br />
the models we use can bias our interpretation <strong>of</strong><br />
experimental data and how atomistic simulations<br />
can be used to identify and correct common<br />
errors is in structural models <strong>of</strong> protein ligand<br />
complexes.<br />
1. Malde, A.K. and Mark, A.E. (2011) Challenges in the<br />
determination <strong>of</strong> the binding modes <strong>of</strong> non-standard ligands in<br />
X-ray crystal complexes. J. Comp. Aided Mol. Des. 25, 1-12.<br />
2. Poger, D. and Mark, A. E. (2010)<br />
On the Validation <strong>of</strong> Molecular Dynamics Simulations <strong>of</strong> Saturated<br />
and cis-Monounsaturated Phosphatidylcholine Lipid Bilayers: A<br />
Comparison with Experiment.<br />
J. Chem. Theory Comput. 6, 325-336.<br />
3. Nair, P,C., Malde, A.K. and Mark, A.E. (2011)<br />
Using theory to reconcile experiment: the structural and<br />
thermodynamic basis <strong>of</strong> ligand recognition by Phenylethanolamine<br />
N-Methyl Transferase (PNMT). J. Chem. Theory Comput. 7,<br />
1458-1468<br />
K.5 4:50pm – 5:10 pm<br />
Towards a Unified Picture <strong>of</strong> Color<br />
and Photisomerization Behavior in<br />
Fluorogenic Monomethine Dyes<br />
Seth Olsen1 The <strong>University</strong> <strong>of</strong> Queensland<br />
Biography<br />
Seth Olsen was born February 8, 1975 in New<br />
York City. His early years were spent growing up<br />
at a series <strong>of</strong> US Coast Guard bases distributed<br />
across the United States; after his father left the<br />
service his family settled in Maryland (in the center<br />
<strong>of</strong> the US Atlantic coast). He obtained his BSc<br />
with Honors in Physics at the College <strong>of</strong> William &<br />
Mary in Virginia (Williamsburg, VA, USA),<br />
completing his undergraduate thesis in solid state<br />
43
44<br />
Biographies and Abstracts<br />
Sunday 4 December - Session 2<br />
biomolecular NMR spectroscopy. He went on to<br />
obtain his PhD at the Center for Biophysics and<br />
Computational Biology at the <strong>University</strong> <strong>of</strong> Illinois,<br />
Urbana-Champaign. Seth’s career has been<br />
marked by a strong interdisciplinary focus that<br />
spans all three <strong>of</strong> the natural sciences <strong>of</strong> Biology,<br />
Chemistry and Physics. He has worked in<br />
departments <strong>of</strong> chemistry and physics, and<br />
publishes in journals devoted to biology, chemistry<br />
and physics. His research interests are focussed<br />
on the development <strong>of</strong> models to explain<br />
molecular photochemical and photobiological<br />
processes - problems that lie at the intersection <strong>of</strong><br />
all three natural sciences.<br />
Abstract<br />
Many techniques for biological fluorescence<br />
imaging rely on dyes that are non-fluorescent in<br />
fluid solution (quantum yield ϕ ~10-5), but become<br />
f<br />
highly fluorescent (ϕ ~1) upon binding to<br />
f<br />
biomolecules. Interestingly, many <strong>of</strong> these dyes<br />
also share a common charge-resonant electronic<br />
structure, wherein the ground state <strong>of</strong> the dye can<br />
be written as a superposition <strong>of</strong> Lewis structures<br />
with opposing bond alternation and formal charge<br />
localization. A well-known example is the<br />
chromophore <strong>of</strong> the green fluorescent protein[1]:<br />
Other dyes for which display both chargeresonance<br />
and binding-dependent fluorescence<br />
enhancement include di- and tri-arylmethanes and<br />
monomethine cyanines. These dyes are venerable<br />
molecules, some <strong>of</strong> them dating to the dawn <strong>of</strong><br />
industrial chemistry itself.<br />
In all these cases, the mechanism <strong>of</strong> fluorescence<br />
enhancement is attributed to suppression <strong>of</strong> a<br />
competing double bond photoisomerization<br />
reaction. In the case <strong>of</strong> resonant dyes, this raises<br />
an obvious question: if there are multiple bonding<br />
schemes superimposed, which bonds are most<br />
likely to twist?<br />
In this talk, I will describe how the pathway<br />
ambiguity can be addressed with high-level<br />
computational quantum chemistry[2]. Furthermore<br />
I will show that the electronic structure describing<br />
the photoisomerization reaction can be rationalized<br />
and described using a simple resonance-theoretic<br />
model that was proposed decades ago to<br />
describe the color <strong>of</strong> charge-resonant dyes[3]. I<br />
will argue that the commonly invoked mechanism<br />
<strong>of</strong> steric hindrance is incomplete. I will suggest<br />
methods <strong>of</strong> further investigation, all <strong>of</strong> which<br />
proceed naturally from the chemically motivated<br />
concept <strong>of</strong> resonance, illustrated above.<br />
[1] M. Chalfie, Angew. Chem. Int. Ed. 48 5603 (2009).<br />
[2] S. Olsen et al., J. Chem. Phys. 130 184302 (2009).<br />
[3] S. Olsen, J. Chem. Theory. Comput. 6 1089 (2010).<br />
[4] S. Olsen et al., J. Chem. Phys. 134 114520 (2011).<br />
K.6 5:10pm – 5:30pm<br />
The roles <strong>of</strong> membrane deformations<br />
and electrostatics in charged<br />
protein-lipid interactions<br />
Toby W. Allen 3 ,Igor Vorobyov1 , Libo Li2 ,<br />
1 Department <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> California, Davis, One<br />
Shields Avenue, CA, 95616, USA. ivorobyov@ucdavis.edu<br />
2 Department <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> California, Davis, One<br />
Shields Avenue, CA, 95616, USA. lili@ucdavis.edu<br />
3 School <strong>of</strong> Applied Science, RMIT <strong>University</strong>, GPO Box 2476V,<br />
Melbourne, VIC, 3001, Australia; Department <strong>of</strong><br />
Chemistry, <strong>University</strong> <strong>of</strong> California, Davis, One Shields Avenue, CA,<br />
95616, USA. toby.allen@rmit.edu.au<br />
Biography<br />
Pr<strong>of</strong>. Allen is a computational biophysicist carrying<br />
out research into membranes, protein-lipid<br />
interactions and ion channel function. His group is<br />
known for its contributions to understand how<br />
charged proteins interact with membranes, and<br />
the mechanisms <strong>of</strong> permeation and selectivity in<br />
ion channels. Pr<strong>of</strong>. Allen received his Ph.D. from<br />
the Australian National <strong>University</strong>, carried out<br />
postdoctoral work at the ANU and then at Cornell<br />
<strong>University</strong> in the USA, after which he became<br />
Assistant and then Associate Pr<strong>of</strong>essor at the<br />
<strong>University</strong> <strong>of</strong> California, Davis. He is the recipient<br />
<strong>of</strong> awards including a National Science Foundation<br />
Career award (USA), Philippe foundation award<br />
(France), Keck Fellowship (USA), Revson<br />
Fellowship (USA), Alberta Heritage Foundation<br />
lecturer award (Canada) and several competitive<br />
funding and supercomputing grants in the USA.<br />
This year he returned to Australia to take up a Vice
Chancellor’s Senior Research Fellowship at RMIT<br />
<strong>University</strong> in Melbourne.<br />
Abstract<br />
Biological membranes are both the gateways into<br />
cells and home to a range <strong>of</strong> proteins that play<br />
essential roles in the body. These membranes<br />
exhibit wide-ranging compositions that influence<br />
their topology, mechanical and electrostatic<br />
properties, which in turn govern protein function<br />
and transport. Using molecular dynamics<br />
simulations, we have uncovered a new theoretical<br />
description <strong>of</strong> the interactions <strong>of</strong> charged<br />
molecules with membranes, with implications for<br />
the actions <strong>of</strong> antimicrobial and viral peptides,<br />
toxins, integral and peripheral membrane proteins.<br />
These studies were motivated by a controversial<br />
model <strong>of</strong> voltage-gated ion channels that assumed<br />
lipid-exposed arginine side chain movement, as<br />
well as cell biological experiments that inferred<br />
small energetic costs for incorporating charged<br />
protein groups into membranes, opposing<br />
long-standing views. Using trans-membrane<br />
protein segment and simple analogue models, we<br />
reveal that the process <strong>of</strong> an ion or charged<br />
protein group crossing a membrane is very<br />
different to that depicted by traditional membrane<br />
models. We demonstrate that the membrane<br />
deforms to such an extent that the dehydration<br />
energy and the membrane electrostatic (dipole)<br />
potential are not determining factors, contrary to<br />
prevailing theory. As a consequence, we observe<br />
almost identical membrane translocation<br />
energetics for a range <strong>of</strong> charged protein groups,<br />
physiological ions, and even ion pairs and<br />
zwitterions. We use this insight to make<br />
predictions for uncatalysed ion permeation,<br />
membrane interactions with highly charged<br />
peptides, and the effects <strong>of</strong> different lipid<br />
components (in particular charged lipids and lipids<br />
<strong>of</strong> varying chain length). We also explore the<br />
competition <strong>of</strong> defect-driven and solubilitydiffusion<br />
mechanisms <strong>of</strong> permeation to allow for<br />
better predictions <strong>of</strong> membrane transport<br />
phenomena.<br />
Biographies and Abstracts<br />
Sunday 4 December - Session 2<br />
Notes<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
45
World class<br />
research leadership<br />
Ge<strong>of</strong>f Cohen<br />
Pr<strong>of</strong>essor Infrastructure Governance<br />
<strong>Peter</strong> <strong>Campbell</strong><br />
Pr<strong>of</strong>essor Infrastructure Systems<br />
Pascal Perez<br />
Pr<strong>of</strong>essor Infrastructure Modelling & Simulation<br />
Henry Ergas<br />
Pr<strong>of</strong>essor Infrastructure Economics<br />
Andrew McCusker<br />
Director, Rail Logistics Research<br />
smart.uow.edu.au
Session 3 – Theatre 2<br />
3.1.1 10:30am – 10:45am<br />
Organic Photovoltaic Materials at<br />
High Spatial and Temporal<br />
Resolution<br />
Trevor A. Smith1 and Xiaotao Hao2 1 Ultrafast and Microspectroscopy Laboratories, School <strong>of</strong><br />
Chemistry, and ARC Centre <strong>of</strong> Excellence for Coherent X-Ray<br />
Science, <strong>University</strong> <strong>of</strong> Melbourne, Vic. 3010, trevoras@unimelb.<br />
edu.au<br />
2 Ultrafast and Microspectroscopy Laboratories, School <strong>of</strong><br />
Chemistry, and ARC Centre <strong>of</strong> Excellence for Coherent X-Ray<br />
Science, <strong>University</strong> <strong>of</strong> Melbourne, Vic. 3010, xhao@unimelb.<br />
edu.au<br />
Biography<br />
Trevor Smith heads the Ultrafast and<br />
Microspectroscopy Labs in the School <strong>of</strong><br />
Chemistry at the <strong>University</strong> <strong>of</strong> Melbourne. He has<br />
worked previously in laboratories at Imperial<br />
College, London and Osaka & Tohoku Universities,<br />
Japan, and held a number <strong>of</strong> competitive research<br />
fellowships, including an ARC QEII Fellowship. His<br />
current research involves the development and<br />
application <strong>of</strong> techniques with femtosecond<br />
time-resolution and sub-diffraction limit optical<br />
resolution, and the combination <strong>of</strong> these two<br />
regimes. He is also a member <strong>of</strong> the ARC Centre<br />
<strong>of</strong> Excellence for Coherent X-Ray Science, using<br />
ultrafast lasers to generate coherent XUV and s<strong>of</strong>t<br />
X-Ray sources for coherent imaging applications.<br />
Abstract<br />
The efficiency <strong>of</strong> photovoltaic devices based on<br />
photo- and electro-luminescent polymeric and<br />
small molecule materials is critically dependent on<br />
the morphology <strong>of</strong> the films produced from these<br />
molecules. Structures can form from the 10s <strong>of</strong><br />
nanometre scale to several microns, dependent on<br />
the materials themselves, the solvent from which<br />
they are cast and the film casting method used.<br />
This is particularly the case in bulk heterojunction<br />
electron/hole donor/acceptor blend systems.<br />
It is therefore necessary to fully characterise such<br />
films on the sub-micron scale, and a range <strong>of</strong><br />
optical microscopy techniques can be applied to<br />
investigate these films. Emission spectroscopic<br />
information can also be gained at the sub-micron<br />
Biographies and Abstracts<br />
Monday 5 December - Session 3<br />
level and provides detail relating to the polymer<br />
conformation, aggregation and energy<br />
redistribution within these films. Many <strong>of</strong> the<br />
photochemical/photophysical processes that<br />
occur following excitation <strong>of</strong> the films occur on the<br />
femtosecond to nanosecond time scales.<br />
Time-resolved emission measurements at these<br />
spatial scales can provide an additional level <strong>of</strong><br />
information relating to aggregation <strong>of</strong> the film<br />
components and electron-hole separation<br />
dynamics.<br />
We will discuss the application <strong>of</strong> a range <strong>of</strong><br />
high-resolution optical microscopy techniques,<br />
coupled with emission spectroscopy and ultrafast<br />
time-resolved measurements to elucidate the<br />
morphological variability <strong>of</strong> the characteristics <strong>of</strong><br />
films <strong>of</strong> conjugated molecules and polymers.<br />
3.1.2 10:45am – 11:00am<br />
Coarse-Grained Modelling <strong>of</strong><br />
Morphology and Energy Transfer in<br />
Conjugated Polymer Nanostructures<br />
David M. Huang 5 , Ming Chiu1 , Kyra N.<br />
Schwarz2 , Scott N. Clafton3 , Tak W. Kee4 1 School <strong>of</strong> Chemistry & Physics, The <strong>University</strong> <strong>of</strong> Adelaide, SA,<br />
5005, ming.chiu@student.adelaide.edu.au<br />
2 School <strong>of</strong> Chemistry & Physics, The <strong>University</strong> <strong>of</strong> Adelaide, SA,<br />
5005, kyra.schwarz@student.adelaide.edu.au<br />
3 School <strong>of</strong> Chemistry & Physics, The <strong>University</strong> <strong>of</strong> Adelaide, SA,<br />
5005, scott.clafton@adelaide.edu.au<br />
4 School <strong>of</strong> Chemistry & Physics, The <strong>University</strong> <strong>of</strong> Adelaide, SA,<br />
5005, tak.kee@adelaide.edu.au<br />
5 School <strong>of</strong> Chemistry & Physics, The <strong>University</strong> <strong>of</strong> Adelaide, SA,<br />
5005, david.huang@adelaide.edu.au<br />
Biography<br />
Dr David Huang is a Lecturer in Chemistry at The<br />
<strong>University</strong> <strong>of</strong> Adelaide. He has a BSc (Hons)<br />
degree in Physical Chemistry from the <strong>University</strong> <strong>of</strong><br />
Sydney and a PhD in Theoretical Chemistry from<br />
the <strong>University</strong> <strong>of</strong> California, Berkeley, which he<br />
carried out as a Fulbright Scholar under the<br />
supervision <strong>of</strong> Pr<strong>of</strong> David Chandler. He worked as<br />
a postdoc with Pr<strong>of</strong> Lyderic Bocquet at the<br />
<strong>University</strong> <strong>of</strong> Lyon and then with Pr<strong>of</strong> Adam Moule<br />
at the <strong>University</strong> <strong>of</strong> California, Davis, before taking<br />
up his current position.<br />
47
48<br />
Biographies and Abstracts<br />
Monday 5 December - Session 3<br />
Abstract<br />
Self-assembled conjugated polymer<br />
nanostructures such as nanoparticles and<br />
nanowires show promise in applications like<br />
biological imaging and organic photovoltaics. One<br />
useful property <strong>of</strong> these nanostructures is the<br />
highly efficient energy transfer that they exhibit<br />
upon photoexcitation. Computational modelling<br />
can shed light on the molecular-scale mechanism<br />
<strong>of</strong> energy transfer, but modelling conjugated<br />
polymers in atomistic detail is not feasible for<br />
nanostructures <strong>of</strong> the size studied experimentally.<br />
We develop coarse-grained simulation models <strong>of</strong><br />
two widely used conjugated polymers, MEH-PPV<br />
and P3HT, in solution. In the parameterisation <strong>of</strong><br />
these models, collections <strong>of</strong> atoms from an<br />
atomistic model are mapped on to a smaller<br />
number <strong>of</strong> coarse-grained sites such that the<br />
coarse-grained models accurately reproduce the<br />
local fluid structure <strong>of</strong> the atomistic models and<br />
retain all relevant molecular details. We use these<br />
models to study the dynamics <strong>of</strong> self-assembly <strong>of</strong><br />
conjugated polymer nanostructures as a function<br />
<strong>of</strong> solvent quality and temperature. We investigate<br />
the dependence <strong>of</strong> energy transfer on<br />
experimental conditions and conjugated polymer<br />
morphology by simulating exciton migration<br />
dynamics upon photoexcitation in the resulting<br />
nanostructures using a Förster line-dipole<br />
approach. The simulated energy transfer<br />
dynamics is compared with time-resolved<br />
measurements <strong>of</strong> polarisation anisotropy decay in<br />
MEH-PPV and P3HT nanostructures.<br />
3.1.3 11:00am – 11:15am<br />
Ultrafast Exciton Dynamics <strong>of</strong><br />
Conjugated Polymer Nanostructures<br />
Tak W. Kee6 , Scott N. Clafton1 , Kyra N.<br />
Schwarz2 , William R. Massey3 , Ming Chiu4 , David<br />
M. Huang5 1 School <strong>of</strong> Chemistry & Physics, The <strong>University</strong> <strong>of</strong> Adelaide, SA,<br />
5005, scott.clafton@adelaide.edu.au<br />
2 School <strong>of</strong> Chemistry & Physics, The <strong>University</strong> <strong>of</strong> Adelaide, SA,<br />
5005, kyra.schwarz@student.adelaide.edu.au<br />
3 School <strong>of</strong> Chemistry & Physics, The <strong>University</strong> <strong>of</strong> Adelaide, SA,<br />
5005,<br />
william.massey@student.adelaide.edu.au<br />
4 School <strong>of</strong> Chemistry & Physics, The <strong>University</strong> <strong>of</strong> Adelaide, SA,<br />
5005, ming.chiu@student.adelaide.edu.au<br />
5 School <strong>of</strong> Chemistry & Physics, The <strong>University</strong> <strong>of</strong> Adelaide, SA,<br />
5005, david.huang@adelaide.edu.au<br />
6 School <strong>of</strong> Chemistry & Physics, The <strong>University</strong> <strong>of</strong> Adelaide, SA,<br />
5005, tak.kee@adelaide.edu.au<br />
Biography<br />
Dr Tak W. Kee is a senior lecturer at the <strong>University</strong><br />
<strong>of</strong> Adelaide. He received his PhD from the<br />
<strong>University</strong> <strong>of</strong> Texas at Austin, USA in 2003. From<br />
2003 to 2006, he was a postdoctoral fellow at the<br />
National Institute <strong>of</strong> Standards and Technology<br />
(NIST) at Gaithersburg, USA. He began his<br />
position with the <strong>University</strong> <strong>of</strong> Adelaide in 2006.<br />
His current research interests include the studies<br />
<strong>of</strong> energy and charge transfer <strong>of</strong> conjugated<br />
polymer nanostructures and investigations <strong>of</strong><br />
molecular processes in naturally occurring<br />
medicinal pigments. He uses time resolved<br />
spectroscopic techniques including fluorescence<br />
upconversion and femtosecond transient<br />
absorption spectroscopy in his work.<br />
Abstract<br />
Conjugated polymer nanostructures are attracting<br />
significant attentions owing to their applications in<br />
light emitting devices, fluorescence imaging and<br />
organic photovoltaics. In this presentation, recent<br />
results from a femtosecond fluorescence<br />
upconversion study on conjugated two polymer<br />
nanostructures, nanoparticles and nanowires, will<br />
be presented. Conjugated polymer nanoparticles<br />
<strong>of</strong>fer colloidal stability in aqueous solution, good<br />
photostability, and tunable luminescence<br />
properties. Conjugated polymer nanowires are<br />
elongated nanostructures that provide efficient<br />
energy transport over a distance <strong>of</strong> microns.<br />
Comparison <strong>of</strong> the time-resolved fluorescence<br />
results <strong>of</strong> these nanostructures to those <strong>of</strong><br />
conjugated polymer in a “good” solvent reveals the<br />
dependence <strong>of</strong> exciton dynamics on polymer<br />
conformation. In addition, results on fluorescence<br />
anisotropy are presented to show fluorescence<br />
depolarization <strong>of</strong> conjugated polymer<br />
nanoparticles and nanowires, which <strong>of</strong>fer further<br />
insight into the exciton dynamics.
3.1.4 11:15am – 11:30am<br />
Structural, electronic and transport<br />
properties <strong>of</strong> amorphous/crystalline<br />
silicon heterojunctions<br />
Tim F. Schulze1 ,*, Caspar Leendertz 1 , Lars<br />
Korte1 , Nicola Mingirulli1 , Bernd Rech1 1 Helmholtz-Zentrum Berlin, Insitute Silicon Photovoltaics,<br />
Kekuléstraße 5, D-12489 Berlin, Germany<br />
* corresponding author, e-mail: tim.schulze@helmholtz-berlin.de<br />
Biography<br />
Tim Schulze was born in Berlin in 1979. He studied<br />
Mechanical Engineering and Physics in Berlin, La<br />
Laguna (Spain) and Zurich (Switzerland). In 2007,<br />
he graduated at the Swiss Federal Institute <strong>of</strong><br />
Technology Zurich with a diploma on stronglycorrelated<br />
electron physics. Afterwards, he<br />
conducted his PhD work at the Helmholtz-Zentrum<br />
Berlin on silicon-based heterojunction solar cells.<br />
He was awarded a PhD by the Technical <strong>University</strong><br />
Berlin in 2011. As an Alexander-von-Humboldt<br />
visiting fellow he is currently working on<br />
photochemical upconversion <strong>of</strong> photons for solar<br />
cell applications in Timothy Schmidtâs group at<br />
the <strong>University</strong> <strong>of</strong> Sydney.<br />
Abstract<br />
Despite the widespread application in diodes and<br />
high-efficiency solar cells, fundamental aspects<br />
concerning the physics <strong>of</strong> amorphous/crystalline<br />
silicon (a-Si:H/c-Si) heterojunctions remain under<br />
dispute. This e.g. concerns the line-up <strong>of</strong> the<br />
electronic bands, the charge carrier transport<br />
across the heterojunction, or the outstandingly<br />
effective passivation <strong>of</strong> c-Si surface states by<br />
undoped a-Si:H.<br />
In the present study, these aspects are tackled by<br />
linking the microscopic properties <strong>of</strong> thin undoped<br />
a-Si:H layers with the resulting behaviour <strong>of</strong> the<br />
a-Si:H/c-Si heterojunction. Employing infrared<br />
spectroscopy, spectroscopic ellipsometry,<br />
photoelectron spectroscopy and secondary ion<br />
mass spectroscopy, the structural, electronic and<br />
optical properties <strong>of</strong> (i)a-Si:H are analysed.<br />
Then, these properties are linked with the resulting<br />
passivation <strong>of</strong> c-Si surface states, which limit the<br />
obtainable open-circuit-voltage in a heterojunction<br />
Biographies and Abstracts<br />
Monday 5 December - Session 3<br />
solar cell. It is found that in case <strong>of</strong> ideal<br />
processing, the heterojunction does not possess<br />
particular properties but can be described by the<br />
a-Si:H bulk properties projected onto the actual<br />
heterojunction. Based on this conclusion it is<br />
possible to comprehend the complex<br />
phenomenology <strong>of</strong> c-Si surface passivation by<br />
a-Si:H from the properties <strong>of</strong> the amorphous<br />
silicon passivation layer [1]. The principal limit <strong>of</strong><br />
c-Si surface passivation follows naturally from the<br />
metastability inherent to a-Si:H, as does the<br />
explanation <strong>of</strong> passivation degradation effects. The<br />
amorphous network has the propensity to adapt<br />
upon changes in externally controllable<br />
parameters like the Fermi energy, which was<br />
seldom taken into account so far when interpreting<br />
phenomena <strong>of</strong> the a-Si:H/c-Si heterojunction.<br />
Next, the line-up <strong>of</strong> the electronic bands at the<br />
heterojunction is elucidated in device-relevant<br />
a-Si:H/c-Si heterostructures. To this end, a novel<br />
method combining photoelectron spectroscopy<br />
and surface photovoltage measurements is<br />
developed and employed. It is found that upon<br />
widening the a-Si:H optical band gap by<br />
controlling its hydrogen content, predominantly the<br />
valence band <strong>of</strong>fset is increasing while the<br />
conduction band <strong>of</strong>fset stays constant [2]. This<br />
result is consistent with established theories on<br />
the a-Si:H electronic structure, but was not<br />
experimentally observed to date. The significance<br />
<strong>of</strong> the valence band <strong>of</strong>fset for solar cell operation<br />
and possible pathways for tailoring the electronic<br />
properties <strong>of</strong> the heterojunction are discussed.<br />
Thus, insight is gained in the dependence <strong>of</strong><br />
heterojunction band line-up, c-Si surface<br />
passivation, electronic transport and ultimately<br />
solar cell device performance on the structural and<br />
electronic properties <strong>of</strong> the thin a-Si:H layers.<br />
49
50<br />
Biographies and Abstracts<br />
Monday 5 December - Session 3<br />
3.1.5 11.30am – 11.45am<br />
A new understanding <strong>of</strong><br />
hybridization in terms <strong>of</strong> bond<br />
strengths and resonance energies<br />
Pr<strong>of</strong> Noel Hush<br />
The <strong>University</strong> <strong>of</strong> Sydney<br />
Biography<br />
Noel Hush was educated at the <strong>University</strong> <strong>of</strong><br />
Sydney from which he graduated as Bachelor and<br />
Master <strong>of</strong> Science in 1946 and 1948, respectively.<br />
After two decades working in the United Kingdom<br />
he returned to Sydney in 1971 and became the<br />
Foundation Pr<strong>of</strong>essor <strong>of</strong> Theoretical Chemistry,<br />
setting up a new department that had a pr<strong>of</strong>ound<br />
impact on the development <strong>of</strong> the fiels in Australia.<br />
He is most known for the theory <strong>of</strong> electron<br />
transfer processes, a theory developed for<br />
electrochemical processes and inorganic<br />
complexes that is now very widely applied<br />
throughout biochemistry and nanotechnology.<br />
This rapidly attracted very talented staff and<br />
developed a flourishing widely-based research<br />
programme including the introduction <strong>of</strong><br />
experimental Photoelectron Spectroscopy to<br />
Australia. He continued quantum chemical studies<br />
on molecular response functions, including Stark<br />
electronic and vibrational effects. In the area <strong>of</strong><br />
electron transfer he studied inter alia basic rate<br />
formalisms, long range bridged transfer, soliton<br />
theory, photosynthetic reaction-centre structure<br />
and function and applications <strong>of</strong> Stark<br />
spectroscopy to properties <strong>of</strong> mixed-valence<br />
systems. His interests drew him naturally towards<br />
Molecular Electronics, and to basic work on<br />
conductivity <strong>of</strong> molecular ‘wires’, switches and<br />
logic assemblies. Following formal retirement in<br />
1989 he works largely in this area, and is currently<br />
Convenor <strong>of</strong> the <strong>University</strong> <strong>of</strong> Sydney Molecular<br />
Electronics Group. There he undertakes basic<br />
theoretical work on molecular conductance,<br />
nanowire-molecule structure and interpretation <strong>of</strong><br />
scanning probe images, including those generated<br />
in the Group’s laboratory. This group is now well<br />
funded by the ARC and major international<br />
industry. He is a Fellow <strong>of</strong> the Royal Australian<br />
Chemical Institute and <strong>of</strong> the Australian Academy<br />
<strong>of</strong> Science, a Fellow <strong>of</strong> the Royal Society <strong>of</strong><br />
London, a Foreign Member <strong>of</strong> the American<br />
Academy <strong>of</strong> Arts and Sciences, and a Fellow <strong>of</strong><br />
the USA National Academy <strong>of</strong> Sciences. He has<br />
received many distinguished awards including the<br />
RACI Phys Chem Division Medal, the Centenary<br />
Medal <strong>of</strong> the Royal Society <strong>of</strong> Chemistry, the<br />
Flinders and inaugural David Craig Medals <strong>of</strong> the<br />
Australian Academy <strong>of</strong> Science, the Welch Prize,<br />
and election as Officer <strong>of</strong> the Order <strong>of</strong> Australia<br />
(AO).<br />
Abstract<br />
Ammonia has a HNH bond angle close to that for<br />
sp hybridization rather than the 120 3 o angle<br />
expected for a planar sp structure and a 90 2 o<br />
angle typical <strong>of</strong> say unhybridized orbitals in BiH . 3<br />
We introduce a simple description <strong>of</strong> this<br />
phenomenon in terms <strong>of</strong> coupled diabatic<br />
surfaces representing the two localized potentialwells<br />
<strong>of</strong> ammonia. This approach links to general<br />
theories <strong>of</strong> chemical reaction dynamics including<br />
proton transfer, aromaticity, and electron transfer.<br />
Standard approaches for interpreting electrontransfer<br />
reactions are then applied and it is found<br />
that a fundamental difference arises: electrontransfer<br />
reactions involve one electron moving<br />
between two orbitals whereas in ammonia two<br />
electrons in the HOMO orbital interact with the<br />
molecules lowest-energy valence unoccupied<br />
orbital. The intrinsic involvement <strong>of</strong> two electrons<br />
in the process causes the singly excited state to<br />
interact strongly with its corresponding doubly<br />
excited state. Hence three states are required in<br />
the fundamental description <strong>of</strong> the chemistry, not<br />
two. However, we describe a transformation<br />
which maps this full problem onto an effective<br />
two-state problem with renormalized parameters.<br />
This explains why 2-state diabatic approaches<br />
have in the past been applied with some success<br />
as well as significant failings- the parameters<br />
previously deduced had no apparent physical<br />
meaning. Our renormalized multi-state approach<br />
also applies to benzene and allows, for the first<br />
time, quantification <strong>of</strong> the interactions between its<br />
Kekulé structures. Hence we determine the<br />
chemical features that control hybridisation in<br />
ammonia using a general chemical theory that can<br />
be used to compare and contrast reactions <strong>of</strong><br />
dramatically different apparent character.
3.1.6 11:45am – 12:00pm<br />
Understanding electron transport in<br />
complex systems<br />
Gemma C. Solomon1 1 Nano-Science Center and Department <strong>of</strong> Chemistry, <strong>University</strong><br />
<strong>of</strong> Copenhagen, Universitetsparken 5, 2100 Copenhagen Ø,<br />
Denmark<br />
Biography<br />
Gemma completed her PhD in Chemistry at the<br />
<strong>University</strong> <strong>of</strong> Sydney in 2006 under the supervision<br />
<strong>of</strong> Jeff Reimers. After that she moved to Chicago<br />
where she postdoc’d at Northwestern with Mark<br />
Ratner. In 2010 she received a European Research<br />
Council Starting Grant and joined the faculty at the<br />
<strong>University</strong> <strong>of</strong> Copenhagen as an Assistant<br />
Pr<strong>of</strong>essor.<br />
Abstract<br />
As system complexity increases, in either<br />
biological or synthetic molecules, understanding <strong>of</strong><br />
structure-function relationships makes it possible<br />
to identify the essential functional units controlling<br />
physical properties from what may be a vast sea <strong>of</strong><br />
spectator components. Until recently, the range <strong>of</strong><br />
theoretical tools that have been implemented for<br />
elucidating structure-function relationships in<br />
molecular electron transport have been limited and<br />
consequently the ability to build chemical intuition<br />
for the behaviour <strong>of</strong> complex systems has been<br />
hindered.<br />
Here we present our efforts developing a local<br />
description <strong>of</strong> molecular electron transport1, which<br />
has allowed us to map the interactions in a<br />
molecule that mediate the tunnelling current, as<br />
shown in Figure 1. With this description <strong>of</strong> the local<br />
transport we can understand the behaviour <strong>of</strong> a<br />
complex, fluctuating system2 as a force is applied<br />
that induces conformational change. We can<br />
isolate the interactions in the molecule responsible<br />
for high currents in both the folded and unfolded<br />
conformations and can use this information to<br />
refine the system design.<br />
Biographies and Abstracts<br />
Monday 5 December - Session 3<br />
Figure 1 The local transmission through a complex molecule,<br />
showing the interactions in the molecule that mediate the<br />
tunnelling current.<br />
We illustrate how the combination <strong>of</strong> conductance<br />
and force spectroscopy can provide a large<br />
amount <strong>of</strong> information about the folded structure<br />
<strong>of</strong> a complex system, providing a model for future<br />
experiments integrating these two techniques for<br />
studying biological systems.<br />
1. G. C. Solomon, C. Herrmann, T. Hansen, V. Mujica and M. A.<br />
Ratner, Nature Chem. (2010), 2, 223-228<br />
2. I. Franco, C. B. George, G. C. Solomon, G. C. Schatz and M. A.<br />
Ratner, J. Am. Chem. Soc. (2011) 133, 2242-2249<br />
Session 3 – Main Theatre<br />
3.2.1 10.30am – 10.50am<br />
Copper Stabilization <strong>of</strong> Aβ42<br />
Aggregation in Model Membranes<br />
Marc-Antoine Sani1 , Daniel K. Weber1 ,<br />
John D. Gehman1 and Frances Separovic1 1 School <strong>of</strong> Chemistry, Bio21 Institute, <strong>University</strong> <strong>of</strong> Melbourne,<br />
VIC 3010<br />
Biography<br />
“Marco” graduated with a Masters <strong>of</strong> Science with<br />
distinctions from the <strong>University</strong> <strong>of</strong> Bordeaux 1. He<br />
then tested his cold resistance by moving for his<br />
PhD training to the <strong>University</strong> <strong>of</strong> Umeå in far north<br />
Sweden. His interest in the fascinating role <strong>of</strong> lipids<br />
started with his PhD thesis investigating the<br />
regulation <strong>of</strong> apoptosis via interaction between<br />
mitochondrial membranes and peptides from the<br />
Bcl protein family. After spending four years<br />
between 24H <strong>of</strong> beautiful sun and 22H <strong>of</strong> complete<br />
darkness, the now-Dr. Sani challenged himself<br />
again by travelling to the antipodes where he<br />
51
52<br />
Biographies and Abstracts<br />
Monday 5 December - Session 3<br />
honed skills in microbiology at the <strong>University</strong> <strong>of</strong><br />
Technology Sydney on gene cassette-based<br />
resistance in bacteria. From here, he felt in love<br />
with Australia and moved to Melbourne to fulfill his<br />
passion <strong>of</strong> biophysics in the laboratory <strong>of</strong> Pr<strong>of</strong>.<br />
Frances Separovic and Dr. John Gehman at the<br />
<strong>University</strong> <strong>of</strong> Melbourne. He currently works on<br />
Alzheimer diseases and antimicrobial peptides,<br />
where his goal is to link peptide/protein interaction<br />
to lipid membrane structure and dynamics.<br />
Abstract<br />
The amyloid-beta (Aß) peptide is associated with<br />
Alzheimer’s disease (AD). Evidence suggests that<br />
Aß interaction with neuronal cell membranes<br />
correlates strongly with neurodegeneration but<br />
understanding the molecular mechanism remains<br />
a challenge. The role <strong>of</strong> heavy metals <strong>of</strong>ten found<br />
in amyloid plaques from AD brain patients is<br />
another path for investigation. Our recent findings<br />
support the role <strong>of</strong> lipid membrane composition as<br />
crucial in many biological mechanisms. We<br />
observed that different lipids promote different<br />
fibril morphologies and aggregation kinetics and<br />
are now facing the following conundrum: how<br />
does copper mediate Aß42 aggregation and how<br />
do membranes interact with Aß42-Cu complexes?<br />
ThT fluorescence and circular dichroism showed<br />
that increasing copper concentration above<br />
equimolar ratio stabilized Aß42 into nonamyloidogenic<br />
beta-sheet structures with or<br />
without the presence <strong>of</strong> lipids. 31P and 2H<br />
solid-state NMR revealed that equimolar Aß42-Cu<br />
complexes had some effect on lipid membrane<br />
structure and dynamics while the hydrophobic<br />
core remained unperturbed and that Aß42<br />
peptides did not scavenge copper from the lipid<br />
headgroup. However, copper-free samples<br />
showed a net reduction in the headgroup<br />
dynamics, with the chemical shift anisotropy, T1<br />
and T2 relaxation values being strongly reduced<br />
and again lacked evidence <strong>of</strong> hydrophobic core<br />
perturbations. The data supports a periperipheral<br />
interaction <strong>of</strong> A←42 and peptide-Cu complexes<br />
with phospholipid membranes.<br />
3.2.2 10:50am – 11:10am<br />
The enigma <strong>of</strong> the CLIC proteins: ion<br />
channels, redox proteins, enzymes,<br />
scaffolding proteins?<br />
Paul M.G Curmi1,3 , Dene R. Littler 1 , Stephen<br />
J. Harrop 1 , Sophia C. Goodchild 2 , Juanita M.<br />
Phang1 , Andrew V. Mynott1 , Lele Jiang3 , Louise J.<br />
Brown2 , Samuel N. Breit3 1 School <strong>of</strong> Physics, <strong>University</strong> <strong>of</strong> New South Wales, Sydney<br />
NSW 2052, Australia<br />
2 Department <strong>of</strong> Chemistry and Biomolecular Sciences,<br />
Macquarie <strong>University</strong>, Sydney, New South Wales 2109,<br />
Australia.<br />
3 St Vincent’s Centre for Applied Medical Research, St Vincent’s<br />
Hospital and <strong>University</strong> <strong>of</strong> New South Wales, Sydney NSW 2010<br />
Australia<br />
Biography<br />
Pr<strong>of</strong>essor Paul Curmi leads the Protein Structure<br />
group in the School <strong>of</strong> Physics, <strong>University</strong> <strong>of</strong> New<br />
South Wales. His research focuses on the<br />
structure and function <strong>of</strong> proteins as determined<br />
by x-ray crystallography. Key areas <strong>of</strong> his research<br />
include: the CLIC proteins that undergo radical<br />
structural changes including membrane insertion;<br />
cryptophyte light harvesting proteins, where<br />
quantum coherence may play a role in biological<br />
function; and higher order organization <strong>of</strong> protein<br />
systems within cells including biological pattern<br />
formation<br />
Abstract<br />
Chloride intracellular channel proteins (CLICs) are<br />
distinct from most ion channels in that they have<br />
both soluble and integral membrane forms. CLICs<br />
are highly conserved in chordates, with six<br />
vertebrate paralogues. CLIC-like proteins are<br />
found in other metazoans. The crystal structures<br />
<strong>of</strong> the soluble form <strong>of</strong> CLIC proteins shows that<br />
they belong to the GST fold family. They differ<br />
from the GSTs in having a glutaredoxin(Grx)-like<br />
redox active site centred on a conserved cysteine.<br />
In this form, the CLIC proteins do not bind GSH<br />
strongly and yet portions <strong>of</strong> the GSH binding site<br />
are preserved. CLICs form channels in artificial<br />
bilayers in a process favoured by oxidising<br />
conditions and low pH. They are structurally<br />
plastic, with CLIC1 adopting two distinct soluble<br />
conformations that have been characterised at
high resolution by x-ray crystallography. This is a<br />
very dramatic structural transition with a complete<br />
refolding <strong>of</strong> the Grx-like N-domain <strong>of</strong> CLIC1.<br />
Phylogenetic and structural data indicate that<br />
CLICs are likely to have enzymatic function. The<br />
physiological role <strong>of</strong> CLICs appears to be<br />
maintenance <strong>of</strong> intracellular membranes, which is<br />
associated with tubulogenesis but may involve<br />
other substructures.<br />
3.2.3 11:10am – 11:30am<br />
The Advantage <strong>of</strong> Being an<br />
Oligomer: the Trimeric Betaine<br />
Carrier BetP<br />
Reinhard Kraemer1 ,Markus Becker1 , Camilo<br />
Perez2 , Christine Ziegler2 1 Institute <strong>of</strong> Biochemistry, <strong>University</strong> <strong>of</strong> Cologne, Zuelpicher Str.<br />
47, 50674 Cologne, Germany.<br />
2 Dep. Of Structural Biology, Max Planck Institute <strong>of</strong> Biophysics,<br />
Frankfurt, Germany. r,kraemer@unikoeln.de; markus.becker@<br />
uni-koeln.de; Camilo.Perez@mpibp-frankfurt.mpg.de; Christine.<br />
Ziegler@mpibpfrankfurt.mpg.de<br />
Biography<br />
Reinhard Kraemer studied Biochemistry at the<br />
Universities <strong>of</strong> Tuebingen and Munich, Germany.<br />
His PhD thesis (1978, Institute <strong>of</strong> Physical<br />
Biochemistry, LMU Munich) focused on<br />
membrane protein reconstitution. As a postdoc,<br />
he worked on energetics and regulation <strong>of</strong><br />
mitochondrial transporters. In 1987 he was jointly<br />
appointed associate pr<strong>of</strong>essor at the <strong>University</strong> <strong>of</strong><br />
Düsseldorf and the Institute <strong>of</strong> Biotechnology<br />
(Research Center Juelich). There he switched to<br />
the study <strong>of</strong> bacterial transport systems and<br />
discovered active mechanisms <strong>of</strong> amino acid<br />
export. After becoming chair in Biochemistry at<br />
Koeln <strong>University</strong> in 1997, his major interest is<br />
currently bacterial stress response and signal<br />
transduction, as well as microbial biotechnology.<br />
He has published more than 200 scientific<br />
publications in peer reviewed journals.<br />
Abstract<br />
Solute carriers occur in various states <strong>of</strong><br />
oligomerization, frequently in monomeric, dimeric,<br />
or trimeric state. So far, in not a single case, the<br />
functional significance <strong>of</strong> the oligomeric state <strong>of</strong> a<br />
Biographies and Abstracts<br />
Monday 5 December - Session 3<br />
particular transporter has been elucidated. We<br />
describe here structural (X-ray and electron<br />
crystallography) and functional studies (solute<br />
transport in intact cells and in reconstituted<br />
proteoliposomes) <strong>of</strong> the osmoregulated,<br />
secondary active, glycine betaine uptake system<br />
<strong>of</strong> the grampositive soil bacterium<br />
Corynebacterium glutamicum. The homotrimeric<br />
carrier BetP is a unique transport system in<br />
harboring three independent functions, (a) sensing<br />
<strong>of</strong> hyperosmotic stress (stimulus perception), (b)<br />
intramolecular signal transduction from the<br />
sensory to the catalytic domain, and (c) Na+coupled<br />
uptake <strong>of</strong> betaine (transport catalysis). On<br />
the basis <strong>of</strong> the X-ray structure, the contact site<br />
between the three identical monomers were<br />
identified. Site-specific replacement <strong>of</strong> three amino<br />
acids in the contact site led to a stable monomeric<br />
form <strong>of</strong> BetP. Monomeric BetP turned out to be<br />
still transport competent, albeit at low activity<br />
(function c), but has lost its regulatory competence<br />
(functions a and b). Consequently, transport does<br />
not require the trimeric state whereas regulation<br />
does. By constructing artificial heterotrimeric forms<br />
<strong>of</strong> BetP, we currently try to elucidate the<br />
conformational crosstalk between the individual<br />
BetP monomers on a molecular level.<br />
3.2.4 11:30am – 11:45am<br />
Single-Molecule View <strong>of</strong> the<br />
Dynamics <strong>of</strong> Molecular Machines<br />
Till Böcking1 1 Centre for Vascular Research, UNSW, Sydney 2052, till.<br />
boecking@unsw.edu.au<br />
Biography<br />
ARC Future Fellow Till Böcking is the leader <strong>of</strong> the<br />
Molecular Machines Unit in the Centre for Vascular<br />
Research. Originally trained in biochemistry at the<br />
<strong>University</strong> <strong>of</strong> Bonn in Germany, he crossed<br />
disciplines and completed a PhD in Biophysics at<br />
UNSW with Hans Coster and Kevin Barrow in<br />
2004. During his doctoral and subsequent<br />
postdoctoral research with Justin Gooding and<br />
Michael Gal, he developed self-assembly<br />
chemistries to assemble biomolecules on<br />
surfaces. This work lead to the development <strong>of</strong><br />
53
54<br />
Biographies and Abstracts<br />
Monday 5 December - Session 3<br />
biosensors and the discovery <strong>of</strong> fundamental<br />
principles that govern the performance <strong>of</strong> these<br />
devices. In 2006 he joined the group <strong>of</strong> Tomas<br />
Kirchhausen at Harvard Medical School to<br />
elucidate the mechanism <strong>of</strong> the chaperonemediated<br />
disassembly <strong>of</strong> the protein coat<br />
surrounding endocytic vesicles. Till was awarded a<br />
Cross-Disciplinary Fellowship <strong>of</strong> the Human<br />
Frontier Science Program in 2007. Since returning<br />
to Australia, Till leads independent research<br />
focused on understanding biological processes at<br />
the molecular level using approaches from the<br />
physical sciences. In particular his team uses<br />
single-molecule techniques to resolve<br />
mechanistsic questions inaccessible with<br />
traditional approaches.<br />
Abstract<br />
Single-molecule approaches have had a huge<br />
impact on our understanding <strong>of</strong> molecular<br />
processes because they provide insight into the<br />
dynamics <strong>of</strong> biomolecular interactions that are<br />
inaccessible with traditional techniques. The<br />
advantage <strong>of</strong> single molecule measurements is<br />
that they can resolve the kinetics <strong>of</strong> processes<br />
without the need for synchronization and permit<br />
the detection <strong>of</strong> short-lived intermediates in the<br />
reaction pathways that are otherwise averaged out<br />
in classical ensemble measurements. Here we<br />
integrate surface chemistry approaches and<br />
micr<strong>of</strong>luidics with fluorescence microscopy to<br />
visualise the dynamic interactions between protein<br />
machines and their substrates at the singlemolecule<br />
level. Glass coverslips are chemically<br />
modified with self-assembled monolayers<br />
designed to capture fluorescence labelled<br />
biomolecules while resisting the non-specific<br />
adsorption <strong>of</strong> other species. We then use<br />
fluorescence imaging to record at high temporal<br />
resolution the interactions <strong>of</strong> individual immobilised<br />
biomolecules with their interaction partners in real<br />
time. The utility <strong>of</strong> the approach is demonstrated<br />
by following in real time the kinetics <strong>of</strong> the<br />
assembly and disassembly <strong>of</strong> macromolecular<br />
complexes.<br />
3.2.5 11:45am – 12:00pm<br />
The Dynamic Stator Stalk <strong>of</strong> A-type<br />
ATPase<br />
Alastair G. Stewart 1,2 , Lawrence K. Lee1,2 ,<br />
Mhairi Donohoe3 and Daniela Stock1,2 1 The Victor Chang Cardiac Research Institute, Darlinghurst, NSW<br />
2010<br />
2 Faculty <strong>of</strong> Medicine, <strong>University</strong> <strong>of</strong> New South Wales, Sydney,<br />
NSW 2053<br />
3 National Neuroscience Facility, Melbourne, Vic 3053<br />
Biography<br />
After obtaining a Bachelor <strong>of</strong> Arts from the<br />
<strong>University</strong> <strong>of</strong> Cambridge in England, Alastair<br />
emigrated to Australia to train in crystallography<br />
under the supervision <strong>of</strong> Daniela Stock at the<br />
Victor Chang Cardiac Research Institute. Alastair’s<br />
PhD project has been to investigate proton<br />
translocating ATPases via X-ray crystallography.<br />
He is now in his final year, and has been able to<br />
obtain two crystal structures <strong>of</strong> a sub complex <strong>of</strong><br />
this biological rotary motor, which has lead to a<br />
new hypothesis <strong>of</strong> how these complex nano<br />
machines function.<br />
Abstract<br />
Rotary ATPases couple ATP hydrolysis/synthesis<br />
with proton translocation across biological<br />
membranes. The peripheral stalks are essential<br />
components <strong>of</strong> these rotary ATPases and function<br />
to counteract torque generated by rotation <strong>of</strong> the<br />
central stalk during proton translocation. The<br />
peripheral stalks have been proposed to form rigid<br />
scaffolds that prevent rotation <strong>of</strong> the soluble head<br />
domain relative to the ion channel anchored in the<br />
membrane. We have solved a 2.25 Å resolution<br />
crystal structure <strong>of</strong> the peripheral stalk from<br />
Thermus thermophilus H+-ATPase/synthase.<br />
Comparison with a different crystal form identifies<br />
a bending and twisting motion inherent within the<br />
peripheral stalk that can accommodate and<br />
complement the wobbling motion <strong>of</strong> rotary<br />
ATPases as they progress through different states<br />
<strong>of</strong> their catalytic cycle. Transitions observed<br />
between different conformations <strong>of</strong> the peripheral<br />
stalk in crystal structures and in low-resolution<br />
electron micrographs can be simulated by normal<br />
mode analysis. This motion changes the
orientation <strong>of</strong> the head <strong>of</strong> the peripheral stalk<br />
relative its coiled-coil domain, coupled with<br />
bending the coiled-coil itself. This analysis also<br />
shows that the novel right-handed coiled-coil <strong>of</strong><br />
the peripheral stalk can accommodate radial<br />
fluctuations associated with nucleotide binding<br />
states <strong>of</strong> the subunits as they move between<br />
catalytic states, while retaining the stiffness<br />
needed to restrain azimuthal movement generated<br />
by the rotation <strong>of</strong> the central stalk.<br />
Biographies and Abstracts<br />
Monday 5 December - Session 3<br />
Notes<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
55
56<br />
Biographies and Abstracts<br />
Monday 5 December - Session 4<br />
Session 4 – Theatre 2<br />
4.1.1 1:15pm – 1:45pm<br />
Chemistry at the Threshold:<br />
Unexpected Products, Unusual<br />
Mechanisms, and Generally Weird<br />
Things that Happen Near the<br />
Energetic Threshold for a Reaction<br />
Scott H. Kable, Alan T. Maccarone, Klaas<br />
Nauta, Gabrielle de Wit, Mitchell Quinn, Scott A.<br />
Reid, Klaas Nauta, and Meredith J.T. Jordan<br />
School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney, Sydney, NSW, 2006<br />
Biography<br />
Scott Kable is a pr<strong>of</strong>essor <strong>of</strong> chemistry at the<br />
<strong>University</strong> <strong>of</strong> Sydney. He earned his PhD at Griffith<br />
<strong>University</strong> and did 3 years post-doctoral research<br />
at Cornell <strong>University</strong> before returning to Australia at<br />
the <strong>University</strong> <strong>of</strong> Sydney in 1992. His research has<br />
been recognised by the Le Fevre Award <strong>of</strong> the<br />
Australian Academy <strong>of</strong> Science, a JILA Fellowship,<br />
and a Fulbright Senior Fellowship in 2010. He is<br />
also a committed teacher who has been awarded<br />
a Carrick Award and Citation, and several<br />
<strong>University</strong> Teaching Awards.<br />
Abstract<br />
Reactions that occur near the energetic threshold<br />
can be very different to those where energy is in<br />
excess. In a series <strong>of</strong> experiments, we have used a<br />
narrow linewidth laser to provide a small organic<br />
molecule, acetaldehyde (CH3CHO), with a precise<br />
amount <strong>of</strong> energy, and used spectroscopic and<br />
imaging based techniques to probe the reaction<br />
products. In this paper I shall present two different<br />
examples <strong>of</strong> “weird chemistry” that happens at<br />
threshold: i) fragments <strong>of</strong> acetaldehyde, CH3 and<br />
HCO, or CH3CO and H, almost separate but are<br />
trapped in their mutual van der Waals well. Here<br />
they orbit each other and produce different<br />
chemical products; ii) selectively deuterated<br />
acetaldehyde is observed to undergo facile and<br />
unexpected H/D exchange before the reaction is<br />
complete. Near-threshold chemistry is not an<br />
esoteric, narrow window, but one that is prevalent<br />
in chemistry more generally. Examples in<br />
atmospheric and combustion chemistry will be<br />
presented.<br />
4.1.2 1:45pm – 2:00pm<br />
Convergent first principles quantum<br />
dynamics: vMCG and Grow<br />
Terry J. Frankcombe<br />
Research School <strong>of</strong> Chemistry, Australian National <strong>University</strong>, ACT<br />
0200, tjf@rsc.anu.edu.au<br />
Biography<br />
Terry Frankcombe (born 1975, Tasmania) received<br />
his BSc(Hons) with a <strong>University</strong> Medal from the<br />
Australian National <strong>University</strong> in 1997, which<br />
included a period conducting research with Pr<strong>of</strong>.<br />
Sture Nordholm at Göteborgs Universitet. He<br />
completed his PhD in 2001 under the supervision<br />
<strong>of</strong> Pr<strong>of</strong>. Sean Smith at the <strong>University</strong> <strong>of</strong><br />
Queensland. This was followed by postdoctoral<br />
work at the <strong>University</strong> <strong>of</strong> Queensland<br />
(2002--2003) and with Pr<strong>of</strong>. Geert-Jan Kroes at<br />
the <strong>University</strong> <strong>of</strong> Leiden (2004--2006). He spend a<br />
year at Göteborgs Universitet as a Marie Currie<br />
Fellow working with Pr<strong>of</strong>. Gunnar Nyman before<br />
returning to the Australian National <strong>University</strong> to<br />
work with Pr<strong>of</strong>. Michael Collins. In<br />
2010 he was awarded a Future Fellowship by the<br />
Australian Research Council. His theoretical and<br />
computational research interests are diverse, from<br />
modelling solid state materials to astrochemistry<br />
and quantum scattering methodology.<br />
Abstract<br />
Performing accurate quantum dynamics<br />
calculations requires two things: a method to<br />
simulate the properties <strong>of</strong> the nuclear wave<br />
function, and an accurate potential energy surface<br />
(or set <strong>of</strong> coupled surfaces) that determines these<br />
properties. Moving beyond a few atoms using<br />
traditional grid-based<br />
methods is extremely computationally onerous on<br />
both counts, with the configuration space grid<br />
getting very large very quickly.<br />
A novel approach is being developed to<br />
counteract both <strong>of</strong> these issues at once. The<br />
dynamics calculations are performed using a<br />
variational time-dependent wave packet approach<br />
(vMCG) that only depends on the potential and its<br />
derivatives at a reasonably small number <strong>of</strong>
configurations along non-classical trajectories.<br />
The potential is expressed as a modified Shepard<br />
interpolation <strong>of</strong> scattered ab initio data. The key<br />
idea in the overall approach is that these two areas<br />
<strong>of</strong> the calculation must be tightly coupled in an<br />
iterative process, so that the dynamics calculations<br />
specify where ab initio calculations must be<br />
performed and one gets the most “value for<br />
money” out <strong>of</strong> each ab initio calculation.<br />
In this presentation the methodology will be<br />
described in detail, along with demonstrations that<br />
the approach not only works, but that the method<br />
has the unique feature that explicit assessment <strong>of</strong><br />
the convergence <strong>of</strong> the dynamics results<br />
(demonstrable convergence) is inherently built in.<br />
4.1.3 2:00pm – 2:15pm<br />
Quantum mechanical study <strong>of</strong> the<br />
deep well reaction H2+ / D2<br />
M.Hankel 1<br />
1 Centre for Computational Molecular Science, Australian Institute<br />
for Bioengineering and Nanotechnology, The <strong>University</strong> <strong>of</strong><br />
Queensland, Brisbane QLD 4072, Australia<br />
Biography<br />
Marlies Hankel obtained her first degree in<br />
Mathematics in her home country <strong>of</strong> Germany.<br />
She then moved to the <strong>University</strong> <strong>of</strong> Bristol in the<br />
UK for her PhD in Physical Chemistry with Pr<strong>of</strong>.<br />
Gabriel Balint-Kurti to work on the development <strong>of</strong><br />
the real wavepacket approach. After a postdoc in<br />
Manchester with Pr<strong>of</strong>. Jonathan Connor she<br />
moved to the <strong>University</strong> <strong>of</strong> Queensland in 2004.<br />
Since then she has worked in the group <strong>of</strong> Pr<strong>of</strong>.<br />
Sean Smith investigating the reaction dynamics <strong>of</strong><br />
gas phase reactions and further developing her<br />
own reactive scattering code. Apart from her<br />
research Marlies Hankel is also the manager <strong>of</strong> the<br />
local high performance computer cluster with<br />
300+ CPUs.<br />
Abstract<br />
We present integral and differential cross sections<br />
as well as product state distributions for the H+ +<br />
D2 reaction obtained from quantum mechanical<br />
calculations employing the DIFFREALWAVE (DRW)<br />
code [1,2]. We employ the ground adiabatic<br />
Biographies and Abstracts<br />
Monday 5 December - Session 4<br />
electronic state potential energy surface <strong>of</strong><br />
Kamisaka et al. [3]. The integral cross sections<br />
decrease with energy and we find cold vibrational<br />
distributions and highly inverted rotational<br />
distributions. The differential cross sections<br />
oscillate strongly with energy.<br />
We also employ an adapted DRW code where we<br />
can restrict the helicity states to a fixed kmax and<br />
with this the Coriolis couplings involved in the<br />
dynamics [4]. This introduces a significant saving<br />
in the computational effort required for these<br />
demanding calculations which, in the present<br />
case, is about 2/3 compared to the fully coupled<br />
calculations. Coriolis coupling is found to be<br />
important in the H+ + D2 reaction dynamics and<br />
truncating the couplings leads to an overestimation<br />
<strong>of</strong> the exact results. However, we also find that the<br />
features <strong>of</strong> the integral cross section are well<br />
reproduced as well as the product rotational state<br />
distributions if the maximum helicity state is<br />
chosen carefully [4].<br />
References:<br />
[1] M. Hankel, S. C. Smith, R. J. Allan, S. K. Gray and G. G.<br />
Balint-Kurti, J. Chem. Phys. 125, 164303 (2006).<br />
[2] M. Hankel, S. C. Smith, S. K. Gray and G. G. Balint-Kurti,<br />
Comput. Phys. Commun. 179, 569 (2008).<br />
[3] H. Kamisaka, W. Bian, K. Nobusada and H. Nakamura, J.<br />
Chem. Phys. 116, 654 (2002).<br />
[4] M. Hankel, Phys. Chem. Chem. Phys. 13, 7948 (2011).<br />
4.1.4 2:15pm – 2:30pm<br />
Optical Spectroscopy <strong>of</strong> Polycyclic<br />
Aromatic Nitrogen Heterocycle<br />
Cations<br />
Viktoras Dryza 1 , Evan Robertson 2 , Evan<br />
Bieske3 1 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Melbourne, Parkville 3010,<br />
vdryza@unimelb.edu.au<br />
2 Department <strong>of</strong> Chemistry, La Trobe <strong>University</strong>, Bundoora,<br />
Victoria, 3086, e.robertson@latrobe.edu.au<br />
3 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Melbourne, Parkville 3010,<br />
evanjb@unimelb.edu.au<br />
Biography<br />
Viktoras Dryza received his PhD from the<br />
<strong>University</strong> <strong>of</strong> Adelaide in 2008, under the<br />
supervision <strong>of</strong> Gregory Metha, for elucidating the<br />
structures <strong>of</strong> gas-phase metal-carbide clusters via<br />
photoionization efficiency spectroscopy. Currently<br />
57
58<br />
Biographies and Abstracts<br />
Monday 5 December - Session 4<br />
he is a postdoctoral fellow in the research group <strong>of</strong><br />
Evan Bieske at the <strong>University</strong> <strong>of</strong> Melbourne,<br />
characterizing gas-phase molecular ions <strong>of</strong><br />
technological and astrophysical relevance using<br />
infrared and optical photodissociation<br />
spectroscopy.<br />
Abstract<br />
Polycyclic Aromatic Hydrocarbons (PAHs) in their<br />
neutral or ionized forms are believed by some to<br />
be constituents <strong>of</strong> the interstellar medium and<br />
perhaps associated with the diffuse interstellar<br />
bands [1]. An important variation <strong>of</strong> PAHs entails<br />
the incorporation <strong>of</strong> nitrogen atoms to form<br />
Polycyclic Aromatic Nitrogen Heterocycles<br />
(PANHs). To develop a better understanding <strong>of</strong> the<br />
optical properties <strong>of</strong> PANHs we have investigated<br />
the D3 ← D0 and D4 ← D0 transitions <strong>of</strong> the<br />
quinoline and isoquinoline radical cations,<br />
structural isomers <strong>of</strong> the simplest PANH member<br />
C9H7N+. The spectra are obtained by massselecting<br />
quinoline+-Ar and isoquinoline+-Ar<br />
complexes in a tandem mass spectrometer and<br />
monitoring the photo-induced Ar loss channel. The<br />
D3 ← D0 bands <strong>of</strong> quinoline+-Ar and isoquinoline+-<br />
Ar feature origin transitions at 16050 and 15245<br />
cm-1, respectively, and display several strong<br />
vibronic progressions. The analogous transition for<br />
the isoelectronic naphthalene+-Ar complex occurs<br />
at a slightly lower energy (14863 cm-1 [2]). To aid<br />
spectroscopic assignments, the resolved vibronic<br />
structure is modelled by performing timedependent<br />
density functional theory calculations in<br />
conjunction with Franck-Condon simulations.<br />
FIG. 1: Resonance enhanced photodissociation spectrum <strong>of</strong> the<br />
isoquinoline+-Ar complex.<br />
References:<br />
[1] PAHs and the Universe, edited by C. Joblin and A.G.G.M.<br />
Tielens, EAS Publication Series Vol. 46 (2011).<br />
[2] T. Pino, N. Boudin, P. Bréchignac, J. Chem. Phys. 111, 7337<br />
(1999).<br />
4.1.5 2:30pm – 2:45pm<br />
Ab Initio Diabatic Potential Energy<br />
Matrix and Dynamics for OH(2S) +<br />
H2 /D2<br />
Michael A. Collins1 , Oded Godsi2 , Shu Liu<br />
and Dong H. Zhang3 1 Research School <strong>of</strong> Chemistry, Australian National<br />
<strong>University</strong>,Canberra. ACT 0200. Australia<br />
2 Research School <strong>of</strong> Chemistry, Australian National<br />
<strong>University</strong>,Canberra. ACT 0200. Australia<br />
3 Dalian Institute <strong>of</strong> Chemical Physics, Chinese Academy <strong>of</strong><br />
Sciences, Dalian 116023, People’s Republic <strong>of</strong> China<br />
Biography<br />
Mick Collins is a pr<strong>of</strong>essor <strong>of</strong> chemistry at the ANU<br />
Abstract<br />
This talk will briefly review the methodology for<br />
constructing an ab initio quasi-diabatic potential<br />
energy matrix (DPEM) that governs nonadiabatic<br />
chemical dynamics in multiple electronic states. A<br />
DPEM for three electronic states <strong>of</strong> OH3 has been<br />
constructed by interpolation <strong>of</strong> multi-reference<br />
configuration interaction electronic structure data.<br />
The reactive, OH(2S) + H2 ← H2O + H, exchange,<br />
and non-reactive quenching dynamics have been<br />
investigated using surface hopping classical<br />
trajectories and time dependent wavepacket<br />
calculations. The most recent results <strong>of</strong> these<br />
dynamical studies on OH3 and deuterated<br />
analogues will be reported.<br />
4.1.6 2:45pm – 3:00pm<br />
G4(MP2)-6X: Accurate and<br />
Affordable Computational Chemistry<br />
Bun Chan1 , Jia Deng2 , Leo Radom3 1 ARC Centre <strong>of</strong> Excellence for Free Radical Chemistry and<br />
Biotechnology and School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney,<br />
Sydney, NSW 2006, Australia<br />
2 Research School <strong>of</strong> Chemistry, Australian National <strong>University</strong>,<br />
Canberra, ACT 0200, Australia<br />
3 ARC Centre <strong>of</strong> Excellence for Free Radical Chemistry and<br />
Biotechnology and School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney,<br />
Sydney, NSW 2006, Australia
Biography<br />
Bun Chan was trained as an experimental organic<br />
chemist in Otago, New Zealand. During his PhD<br />
he also did molecular modeling to complement his<br />
experimental studies, which triggered his interest<br />
in theoretical chemistry. After graduation, he<br />
spent a year in Academia Sinica as a postdoctoral<br />
experimental chemist. He then moved to the<br />
<strong>University</strong> <strong>of</strong> Sydney to embark on a career as a<br />
computational chemist, where he remains to this<br />
date<br />
Abstract<br />
Computational chemistry is nowadays an<br />
indispensable tool to chemists. Nonetheless,<br />
theoretical methods that are <strong>of</strong> chemical accuracy<br />
are still costly in terms <strong>of</strong> computational resource,<br />
thus rendering them applicable only to small<br />
molecules (up to ~ 10 atoms). The Gn series <strong>of</strong><br />
composite procedures has been <strong>of</strong>fering an<br />
excellent solution to this dilemma. The latest in the<br />
series, G4,1 <strong>of</strong>fers chemical accuracy for a diverse<br />
set <strong>of</strong> energies, and can be applied to larger<br />
chemical systems (up to ~ 20 atoms). Its less<br />
expensive cousin, G4(MP2)2 is applicable to even<br />
larger molecules containing ~40–50 atoms. It<br />
shows good performance but has not yet reached<br />
the same level <strong>of</strong> accuracy. Our new procedure,<br />
G4(MP2)-6X,3 is an alternative to G4(MP2) <strong>of</strong><br />
similar cost but with a performance approaching<br />
that for G4. It achieves chemical accuracy for a<br />
diverse set <strong>of</strong> thermochemical properties including<br />
reaction energies, barriers and weak interactions,<br />
thus it is broadly applicable in chemistry.<br />
1 Curtiss, L. A.; Redfern, P. C.; Raghavachari, K. J. Chem. Phys.<br />
2007, 126, 084108.<br />
2 Curtiss, L. A.; Redfern, P. C.; Raghavachari, K. J. Chem. Phys.<br />
2007, 127, 124105.<br />
3 Chan, B.; Deng, J.; Radom, L. J. Chem. Theory Comput. 2011, 7,<br />
112.<br />
Biographies and Abstracts<br />
Monday 5 December - Session 4<br />
Session 4 – Main Theatre<br />
4.2.1 1:15pm – 1:30pm<br />
Test <strong>of</strong> a Protein Docking Algorithm<br />
on K+ Channel Binding: Validation<br />
and Analysis<br />
Po-chia Chen1 , Serdar Kuyucak 2<br />
1 School <strong>of</strong> Physics, <strong>University</strong> <strong>of</strong> Sydney, NSW, Australia 2006.<br />
poker@physics.usyd.edu.au<br />
2 School <strong>of</strong> Physics, <strong>University</strong> <strong>of</strong> Sydney, NSW, Australia 2006.<br />
serdar@physics.usyd.edu.au<br />
Biography<br />
Po-chia is a long-time resident <strong>of</strong> Sydney<br />
<strong>University</strong> who has recently finished his PhD thesis<br />
on potassium channel binding. This thesis process<br />
has brought Po-chia through multiple aspects <strong>of</strong><br />
biophysical simulation, from ab-initio through<br />
classical dynamics and molecular docking. While<br />
aware <strong>of</strong> the likelihood that his new work is old-hat<br />
to researchers in private pharmaceutical<br />
development, he dreams <strong>of</strong> a day when these<br />
corporations can afford to share their knowledge<br />
with academia at large.<br />
Aside from his day-job running biophysical<br />
simulations, Po-chia also thinks about<br />
incorporating computer simulation and<br />
visualisation into science education. He would<br />
particularly like to devise an effective method to<br />
teach organic chemistry.<br />
Abstract<br />
The active components <strong>of</strong> animal venoms have<br />
historically been used to identify channel<br />
expressions in electro-physiology, and is currently<br />
a prospective candidate for related pharmaceutical<br />
applications. The fitness <strong>of</strong> a particular toxin for<br />
these tasks is determined by its selectivity pr<strong>of</strong>ile<br />
over a range <strong>of</strong> target channels. However,<br />
comprehensive affinity data is not always available,<br />
and are time-consuming to produce.<br />
We present a preliminary method to deduce the<br />
selectivity pr<strong>of</strong>ile <strong>of</strong> a peptide toxin against related<br />
channels by means <strong>of</strong> docking simulations. This is<br />
tested on the family <strong>of</strong> ←-KTx scorpion toxins<br />
versus the Shaker-type potassium channels (Kv1),<br />
for which both structural and affinity data are<br />
available to validate predictions. A total <strong>of</strong> ~25<br />
59
60<br />
Biographies and Abstracts<br />
Monday 5 December - Session 4<br />
toxins were docked to Kv1.1, Kv1.2 and Kv1.3,<br />
resulting in ~75 total docking simulations in the<br />
program<br />
The performance <strong>of</strong> HADDOCK will first be<br />
evaluated, followed by an analysis <strong>of</strong> a selection <strong>of</strong><br />
toxins for which validation can be best provided.<br />
The general selectivity pr<strong>of</strong>iles <strong>of</strong> toxin-subfamilies<br />
can be deduced by a consensus between closely<br />
related toxin-channel pairings, while individual<br />
selectivity pr<strong>of</strong>iles can be identified by comparison<br />
<strong>of</strong> docked-complex quality and program<br />
performance. In particular, HADDOCK was able to<br />
classify αKTX2 toxins as universal binders and<br />
αKTX3 toxins as Kv1.3-selective binders. It was<br />
also able to reproduce the differing characteristics<br />
<strong>of</strong> αKTX6-toxins, which depend on sequence and<br />
di-sulphide patterns. Distinct binding orientations<br />
can be observed between the channels (an<br />
example is shown in the included figure).<br />
4.2.2 1:30pm – 1:45pm<br />
Open channel structure <strong>of</strong> MscL<br />
from restrained MD Simulations<br />
Evelyne Deplazes1, Dylan, Jayatilaka1 , Martti<br />
Louhivuori2 , Siewert J. Marrink 2 , Ben Corry1 .<br />
1 The <strong>University</strong> <strong>of</strong> Western Australia, Perth, ben.corry@uwa.edu.<br />
au<br />
2 <strong>University</strong> <strong>of</strong> Groningen, Groningen, Netherlands<br />
Biography<br />
Evelyne Deplazes is a PhD student at the<br />
<strong>University</strong> <strong>of</strong> Western Australia. Her project<br />
focused on using a combination <strong>of</strong> molecular<br />
dynamics simulations and experimental data to<br />
investigate the structure <strong>of</strong> the mechanosensitive<br />
channel <strong>of</strong> large conductance. In addition she has<br />
used simulations to simulate the behavior <strong>of</strong><br />
fluorescent molecules and to better understand<br />
FRET at the molecular level.<br />
Abstract<br />
Mechanosensitive channels are membrane<br />
proteins that have evolved for osmoregulation in<br />
bacteria and function as mechano-electrical<br />
switches in many physiological processes such as<br />
hearing and touch sensation. The<br />
mechanosensitive channel <strong>of</strong> large conductance<br />
(MscL) is the most studied mechanosensitive<br />
channel and <strong>of</strong>ten serves as a model system to<br />
study mechanosensory transduction. Although a<br />
detailed structure <strong>of</strong> the closed state is known,<br />
many questions about the open pore structure<br />
and the gating mechanism are yet to be answered.<br />
The MscL protein has been characterized using<br />
EPR [1] and FRET [2] spectroscopy providing a<br />
large set structural data. In this study we<br />
incorporate inter-subunit distances and solvent<br />
accessibility data in to coarse grained molecular<br />
dynamics simulations to model the open pore<br />
structure <strong>of</strong> MscL. A series <strong>of</strong> simulations with<br />
different combinations <strong>of</strong> restraints and membrane<br />
tension were carried out to produce a set <strong>of</strong><br />
potential open channel structures.<br />
The open structures show a pore diameter<br />
between 26Å and 32Å and TM1 and TM2 tilts <strong>of</strong><br />
~60º and ~46º, respectively. These<br />
measurements are in good agreement with data<br />
from recent all-atoms restrained simulations [3] as<br />
well as previously reported open pore models. In<br />
all our structures the N-terminal lie along the<br />
membrane surface and is therefore unlikely to<br />
serve as a second gate. The C-terminals does not<br />
dissociate during gating but stays as a helical<br />
bundle pointing into the cytoplasm.<br />
Furthermore, the results suggests that an outward<br />
motion <strong>of</strong> the periplasmic loop in combination with<br />
a change in the relative orientation <strong>of</strong> TM1 and the<br />
periplasmic loop are associated the formation <strong>of</strong><br />
the open pore. The results also indicate that<br />
tension induced thinning <strong>of</strong> the lipid bilayer is<br />
necessary for these structural changes to occur.<br />
These results are consistent with data from<br />
experiments <strong>of</strong> MscL in thinner membranes. To<br />
further investigate these effects we carried out<br />
simulations <strong>of</strong> MscL in short-chain lipids.<br />
[1] Perozo E et al. (2002). Open channel structure<br />
<strong>of</strong> MscL and the gating mechanism <strong>of</strong><br />
mechanosensitive channels. Nature, 418, 942-948<br />
[2] Corry B et al. (2005). Conformational changes<br />
involved in MscL channel gating measured using<br />
FRET spectroscopy, Biophysical Journal, 89,<br />
L49-L51<br />
[3] Corry B et al. (2010). An improved openchannel<br />
structure <strong>of</strong> MscL determined from FRET<br />
confocal microscopy and simulation. Journal <strong>of</strong><br />
General Physiology, 136, 483-494
4.2.3 1:45pm – 2:00pm<br />
Estimation <strong>of</strong> the pKa <strong>of</strong> tri-peptides<br />
using Generalized Multiplicative<br />
ANOVA <strong>of</strong> designed data<br />
Rima Raffoul Khoury 1 , Diako Ebrahimi2 , D.<br />
Brynn Hibbert1 1 School <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> New South Wales, NSW,<br />
2052, s3213363@unsw.edu.au<br />
2 Faculty <strong>of</strong> Medicine, Centre <strong>of</strong> Vascular Research, The <strong>University</strong><br />
<strong>of</strong> New South Wales, NSW, 2052, diako@unsw.edu.<br />
au<br />
1 School <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> New South Wales, NSW,<br />
2052, b.hibbert@unsw.edu.au<br />
Biography<br />
Rima Raffoul Khoury is a current PhD student at<br />
the <strong>University</strong> <strong>of</strong> New South Wales, in the<br />
Chemometrics group under the supervision <strong>of</strong><br />
Pr<strong>of</strong>. Hibbert and Dr. Ebrahimi. Rima is working on<br />
modelling the multi-way interactions between<br />
metal ions and oligopeptides systems using<br />
Generalised Multiplicative Analysis <strong>of</strong> Variance<br />
GEMANOVA. Her research tackles two problems:<br />
the requirements <strong>of</strong> experimental design and the<br />
correct modelling <strong>of</strong> data obtained from complex<br />
systems where the measured response is<br />
dominated by high order interaction between the<br />
influencing factors. Rima is using GEMANOVA to<br />
model data obtained from Potentiometry,<br />
Spectroscopy and Electrochemistry.<br />
Previously, she has completed an Honours degree<br />
from UNSW 2008, where she investigated the<br />
degradation <strong>of</strong> Fatty acids methyl esters in<br />
Biodiesels, under bio-degradation and photooxidation<br />
conditions. More details can be found:<br />
Fuel, Volume 90, Issue 8, August 2011, Pages<br />
2677-2683<br />
Rima has obtained double degrees: Maitrise-es in<br />
Biochemistry, 2004 and General Chemistry, 2003<br />
from the National Lebanese <strong>University</strong>, Lebanon.<br />
Abstract<br />
In chemistry, there are many cases in which the<br />
response <strong>of</strong> a process is affected by the<br />
interaction between the influencing factors rather<br />
than only by the individual and independent<br />
factors. The classical methods for modelling<br />
chemical systems have mainly focussed on the<br />
Biographies and Abstracts<br />
Monday 5 December - Session 4<br />
effects <strong>of</strong> individual factors by assuming that the<br />
interaction effects (in particular high order effects)<br />
are insignificant. This is partly because the<br />
inclusion <strong>of</strong> high order interaction terms in classic<br />
models such as ANOVA requires the estimation <strong>of</strong><br />
a large number <strong>of</strong> parameters, which in turn<br />
makes the interpretation <strong>of</strong> the results very difficult.<br />
When the measured response is mainly affected<br />
by the interactions among factors, disregarding<br />
these interactions leads to erroneous models and<br />
inhibits understanding <strong>of</strong> correct mechanisms.<br />
In this work we use a generalized multiplicative<br />
analysis <strong>of</strong> variance (GEMANOVA) to efficiently<br />
model the interactions among amino acid residues<br />
<strong>of</strong> a tri-peptide system in a designed experiment.<br />
A full factorial design <strong>of</strong> a tri-peptide system<br />
containing Glutamic acid (Glu, E), Glycine (Gly, G)<br />
and Histidine (His, H) was investigated. In this<br />
study the three positions <strong>of</strong> amino acid residues<br />
are considered as three factors each having three<br />
instances <strong>of</strong> E, G and H, thus forming a 33 design.<br />
Each <strong>of</strong> 27 different tri-peptide (GGG, GGE, GGH<br />
...HHH) was subjected to an alkali titration under a<br />
specific experimental condition. The HyperQuad<br />
2008 s<strong>of</strong>tware was used to estimate the pKa<br />
values <strong>of</strong> tripeptides from the titration data<br />
obtained from potentiometry experiments. The<br />
estimated pKa data was then modelled using<br />
GEMANOVA.<br />
The results indicate that the position and the type<br />
<strong>of</strong> amino acid have strong influence on the acidity<br />
<strong>of</strong> the terminal carboxylic acid <strong>of</strong> the tripeptides.<br />
To investigate whether it is possible to predict the<br />
pKa <strong>of</strong> tripeptides using GEMANOVA model, we fit<br />
GEMANOVA models on the data with missing pKa<br />
<strong>of</strong> up to 18 tripeptides. The results indicated that<br />
GEMANOVA is a robust method for modelling the<br />
amino acid interactions <strong>of</strong> peptides as well as for<br />
prediction <strong>of</strong> pKa <strong>of</strong> peptides. The tripeptides<br />
EHH, GHH and HHH which have H at position 2<br />
and 3 had the lowest measured pKa for their<br />
terminal carboxylic acid (pKa = 1.905, 1.93 and<br />
2.05, respectively). The peptides which do not<br />
have a terminal H and contain E (especially at<br />
position 1) were found to have relatively high pKa<br />
values; for example the measured pKa for EEG,<br />
EGE, EHE were 3.480, 3.840 and 3.735,<br />
respectively.<br />
61
62<br />
Biographies and Abstracts<br />
Monday 5 December - Session 4<br />
4.2.4 2:00pm – 2:15pm<br />
The Fouling <strong>of</strong> Hydrophobic<br />
Membranes by Hydrophilic<br />
Alginates: A Molecular Dynamics<br />
Study.<br />
Matthew B. Stewart 1 , Darli Theint Myat1 ,<br />
Stephen R Gray1 and John D. Orbell1,2 1 Institute for Sustainability and Innovation, Victoria <strong>University</strong>, PO<br />
Box 14428, Melbourne, Victoria, 8001, matthew.stewart@vu.<br />
edu.au<br />
2 School <strong>of</strong> Engineering and Science, Victoria <strong>University</strong>, PO Box<br />
14428, Melbourne, Victoria, 8001, john.orbell@vu.edu.au<br />
Biography<br />
Dr. Matthew Stewart is a current postdoctoral<br />
fellow at Victoria <strong>University</strong> in Melbourne. Under<br />
Pr<strong>of</strong>. Orbell, he was awarded his PhD in 2009 for a<br />
thesis which focused on the reconciliation <strong>of</strong><br />
experimental and computational data to provide<br />
insights into the behaviours <strong>of</strong> biologically<br />
interesting molecules. This work included<br />
antioxidant activities, nucleotide-metal interactions<br />
and iron-based radical complexes. His current<br />
work involves computational support for<br />
researchers within the Institute for Sustainability<br />
and Innovation at Victoria <strong>University</strong>, which<br />
includes membrane fouling mechanisms,<br />
shear-induced protein conformation changes and<br />
the development <strong>of</strong> approaches towards the<br />
modelling <strong>of</strong> advanced oxidation processes. He<br />
also enjoys AFL football, cricket, traveling and long<br />
walks on the beach.<br />
Abstract<br />
Natural Organic Matter (NOM) is the collective<br />
name for a wide range <strong>of</strong> compounds routinely<br />
found in surface and waste waters. Many<br />
studies1-5 have found that the hydrophilic fraction<br />
<strong>of</strong> NOM is the most significant with respect to the<br />
fouling <strong>of</strong> hydrophobic micr<strong>of</strong>iltration membranes.<br />
Whilst there is no definable hydrophilic NOM<br />
structure, there are a number <strong>of</strong> molecules that<br />
effectively mimic the general properties <strong>of</strong> NOM.<br />
These compounds are used as analogues for the<br />
study <strong>of</strong> the NOM fouling <strong>of</strong> a number <strong>of</strong><br />
membranes used in water treatment - the most<br />
common model compound being sodium alginate.<br />
Alginate (also known as Alginic acid) is an ionic<br />
polyuronic acid naturally found in a number <strong>of</strong><br />
algae (including seaweed) and bacteria species.<br />
Alginate is a polymer consisting <strong>of</strong> linked<br />
←-D-mannuronic acid (M) and ←-L-guluronic acid<br />
(G) monomers. The general structure <strong>of</strong> algaederived<br />
alginates exhibit ordered sequences <strong>of</strong><br />
poly-G, poly-M and alternating GM6. The lengths<br />
and order <strong>of</strong> these sections are highly variable. In<br />
this regard, the composition <strong>of</strong> alginate chains is<br />
most accurately expressed by the G/M ratio,<br />
which is species dependent. Whilst this structural<br />
uncertainty can be an issue in experimental<br />
investigations <strong>of</strong> alginates, especially from a<br />
mechanistic point-<strong>of</strong>-view, it can be used to<br />
advantage in computational studies <strong>of</strong> these<br />
polymeric molecules. Each section (poly-G,<br />
poly-M and alternating GM) can be individually<br />
studied and combined to infer the behaviour <strong>of</strong><br />
longer chains <strong>of</strong> alginates.<br />
This approach has been used in this study, where<br />
decamers <strong>of</strong> G, M and GM were subjected to<br />
molecular dynamics calculations under a variety <strong>of</strong><br />
conditions that reflect the experimental conditions<br />
<strong>of</strong> membrane fouling experiments currently being<br />
carried out within the research group. Thus, the<br />
effect <strong>of</strong> pH and ionic strength on the<br />
conformation <strong>of</strong> the individual alginate chains, the<br />
interactions between the chains and the<br />
interaction <strong>of</strong> the chains with the membrane<br />
surface have been simulated. This work provides<br />
insights into the solution chemistry <strong>of</strong> alginate<br />
molecules, as well as their specific interactions<br />
with cations (mono- and divalent) and polymer<br />
surfaces. Such insights may also be useful to the<br />
food processing and cosmetics industries, as<br />
alginate is a commonly used constituent.<br />
1. S. R Gray, C. B. Ritchie, T. Tran, B. A. Bolto, P. Greenwood, F.<br />
Busetti, B. Allpike, Effect <strong>of</strong> membrane character and solution<br />
chemistry on micr<strong>of</strong>iltration performance, Water Research, 42<br />
(2008) 743-753<br />
2. L. Fan, L. J. Harris, F. A. Roddick, A. N. Booker, Influence <strong>of</strong><br />
characteristics <strong>of</strong> natural organic matter on the fouling <strong>of</strong><br />
micr<strong>of</strong>iltration membranes, Water Research, 35 (2001)<br />
4455-4463<br />
3. T. Carroll, S. King, S. R. Gray, B. Bolto, A. N. Booker, The fouling<br />
<strong>of</strong> micr<strong>of</strong>iltration membranes by NOM after coagulation<br />
treatment, Water Research, 34 (2000) 2861-2868<br />
4. Jarusutthirak. C, A. Gary, Fouling characteristics <strong>of</strong> wastewater<br />
effluent organic matter (EfOM) isolates on NF and UF<br />
membranes, Desalination, 145 (2002) 247-255<br />
5. S.R. Gray, N. Dow, J.D. Orbell, T. Tran and B.A. Bolto, The<br />
significance <strong>of</strong> interactions between organic compounds and<br />
low pressure membrane fouling, Water Science and
Technology, 64 (2011), 632-639<br />
6. I. Donati and S. Paoletti, Material Properties <strong>of</strong> Alginates, In:<br />
Alginates: Biology and Applications, B.H.A. Rehms (Ed.) (2009)<br />
Springer; Dordrecht, Germany<br />
4.2.5 2:15pm – 2:30pm<br />
HIV1-TAT Peptide modified<br />
nanoparticles: insights from<br />
molecular dynamics simulations.<br />
Nevena Todorova1 , Morgan Mager2 , Molly<br />
Stevens2 , Irene Yarovsky1 1 Health Innovations Research Institute, School <strong>of</strong> Applied<br />
Sciences, RMIT <strong>University</strong>, GPO Box 2476 V, Melbourne, VIC,<br />
Australia.<br />
2 Department <strong>of</strong> Materials, Department <strong>of</strong> Bioengineering, and<br />
Institute for Biomedical Engineering, Imperial College London,<br />
London, U.K., SW7 2AZ<br />
Biography<br />
Nevena Todorova completed her PhD in 2009 at<br />
RMIT <strong>University</strong>, where she investigated the effects<br />
<strong>of</strong> environment on protein folding, misfolding and<br />
aggregation using a range <strong>of</strong> computational<br />
modelling techniques. She is now a post-doctorate<br />
fellow in the Materials Modelling and Simulations<br />
group <strong>of</strong> Pr<strong>of</strong>. Irene Yarovsky at RMIT. Her current<br />
projects involve studies on the effects <strong>of</strong><br />
nanoparticles on protein structure and dynamics<br />
and the application <strong>of</strong> bio-inspired nanomaterials<br />
for drug-delivery, sensing and diagnostics. At the<br />
conference she will present her findings from her<br />
recent work on peptide functionalised<br />
nanoparticles titled “HIV1-TAT Peptide modified<br />
nanoparticles: insights from molecular dynamics<br />
simulations”.<br />
Abstract<br />
Nanoparticle functionalisation with peptides has<br />
been shown to be a good strategy to inhibit<br />
particle aggregation, increase nanoparticle<br />
solubility and develop potential nanocarriers [1]. A<br />
biologically-inspired set <strong>of</strong> peptides collectively<br />
known as cell-penetrating peptides (CPPs) has<br />
become an increasingly popular tool for<br />
transfection and other types <strong>of</strong> cellular delivery [2].<br />
One such example is a peptide derived from the<br />
transcription transactivation (TAT) protein from<br />
human HIV-1 virus (residues 48-60,<br />
GRKKRRQRRRPPQ) [3]. Experimental studies<br />
Biographies and Abstracts<br />
Monday 5 December - Session 4<br />
have shown that TAT-peptide functionalized gold<br />
nanoparticles have the ability to penetrate the cell<br />
membrane and localize in the nucleus [4].<br />
However, the peptides’ behaviour with respect to<br />
their immediate environment and the nanoparticle<br />
surface prior to contact with a membrane is not<br />
well understood. In this work we employed fully<br />
atomistic Molecular Dynamics simulations to shed<br />
some light on the structure and dynamics <strong>of</strong> the<br />
TAT functionalized nanoparticles in solution.<br />
We developed the TAT and CALNN peptide<br />
functionalised nanoparticle models at varying<br />
grafting density. The effect <strong>of</strong> TAT:CALNN surface<br />
ratio on the peptide’s structure and dynamics was<br />
investigated for the 3nm nanoparticle coated with<br />
1.82%, 3.71%, 5.66%, 7.69%, 9.81% <strong>of</strong> TAT, the<br />
concentrations close to those used in experiments<br />
[5]. The nanoparticle-peptide systems were<br />
simulated at 285K, 288K, 296K and 308K to<br />
investigate the effects <strong>of</strong> temperature. For each<br />
concentration, the peptide structure, dynamics<br />
and interactions with the nanoparticle surface and<br />
the environment will be discussed. Our results<br />
identified specific properties that may be<br />
necessary for membrane activity <strong>of</strong> the TAT<br />
modified nanoparticles.<br />
Figure showing the 9.8% TAT coated nanoparticle; starting<br />
structure (TAT expanded) and final conformation (TAT collapsed).<br />
[1] A. Makarucha, N. Todorova, I. Yarovsky “Nanomaterials in<br />
biological environment: a review <strong>of</strong> computer modelling studies”<br />
European Biophysical Journal, 40(2):103 (2011).<br />
[2] N.A. Brooks, et al. “Cell-penetrating peptides: Application in<br />
vaccine delivery”, Biochemica et Biophysica Acta, 1805:25 (2010)<br />
[3] A.D. Frankel and C.O. Pabo “Cellular uptake <strong>of</strong> the tat protein<br />
from human immunodeficiency virus”, Cell, 55(6):1189 (1988)<br />
[4] J.M de la Fuente and C.C. Berry “Tat peptide as an efficient<br />
molecule to translocate gold nanoparticles into the cell nucleus”,<br />
Bioconjugate Chem. 16:1176 (2005)<br />
[5] Unpublished results, Pr<strong>of</strong>. Molly Stevens, Dr. Morgan Mager,<br />
Imperial College, London.<br />
63
64<br />
Biographies and Abstracts<br />
Monday 5 December - Session 4<br />
4.2.6 2:30pm – 2:45pm<br />
Ion Permeation and Selectivity in a<br />
Voltage Gated Sodium Channel<br />
Ben Corry1 , Michael Thomas1 1 School <strong>of</strong> Biomedical, Biomolecular and Chemical Sciences,<br />
The <strong>University</strong> <strong>of</strong> Western Australia, Crawley WA 6009, ben.<br />
corry@uwa.edu.au<br />
Biography<br />
Associate Pr<strong>of</strong>essor Ben Corry has focused his<br />
research on studying ion transport in pores,<br />
particularly in biological ion channels. After gaining<br />
his PhD at the ANU, he has been based at the<br />
<strong>University</strong> <strong>of</strong> Western Australia and during this time<br />
has been awarded the 2005 Young Biophysicist<br />
Award from the Australian Society for Biophysics<br />
and the 2008 Young Scientist <strong>of</strong> the Year at the<br />
WA Premier’s Science Awards.<br />
Abstract<br />
Ion channels regulate electrical signalling in cells<br />
by opening and closing selective pores across the<br />
membrane in response to stimuli. In order to carry<br />
out their function, such channels must be able to<br />
rapidly transport ions at the same time as<br />
effectively discriminating between different ion<br />
types so that the correct signal can be passed<br />
without destroying the electrochemical gradients<br />
across the membrane. Investigations into how ions<br />
permeate through membrane channels have been<br />
focussed on K+ selective channels due to the<br />
availability <strong>of</strong> a number <strong>of</strong> high resolution crystal<br />
structures. The recent publication <strong>of</strong> the first high<br />
resolution crystal structure <strong>of</strong> a voltage gated<br />
sodium channel,1 however, opens the door to<br />
understanding how sodium ions permeate into the<br />
cell and how Na+ can be transported while<br />
blocking the passage <strong>of</strong> K+.<br />
Using molecular dynamics simulations we are able<br />
to demonstrate the multi-ion knock-on basis <strong>of</strong> ion<br />
permeation through the channel and identify the<br />
major binding sites for ions. The nature <strong>of</strong> these<br />
binding sites, and the ability <strong>of</strong> ions to pass<br />
through the channel with the majority <strong>of</strong> their<br />
hydration shell is very different to the situation in<br />
potassium channels. Using free energy<br />
calculations we elucidate the mechanism <strong>of</strong> ion<br />
selectivity in the sodium channel and contrast this<br />
to how K+ selectivity is obtained in potassium<br />
channels.<br />
1. J Payandeh, T Scheuer, N Zheng & WA Catterall,<br />
Nature, 475, 353–358 (2011)<br />
4.2.7 2:45pm – 3:00pm<br />
The Role <strong>of</strong> Molecular Strain in the<br />
Ion Selectivity <strong>of</strong> Biological<br />
Molecules<br />
M. Thomas1 , M. J. Turner 1 , D. Jayatilaka1, B.<br />
Corry1 1 School <strong>of</strong> Biomedical, Biomolecular and Chemical Science,<br />
<strong>University</strong> <strong>of</strong> Western Australia, M313 35 Stirling Hwy Crawley,<br />
WA 6009, thomam05@student.uwa.edu.au<br />
Biography<br />
Michael is a PhD student in the school <strong>of</strong><br />
Biomedical, Biomolecular and Chemical Sciences<br />
at the <strong>University</strong> <strong>of</strong> Western Australia under the<br />
supervisor <strong>of</strong> Dr Ben Corry and A/Pr<strong>of</strong> Dylan<br />
Jayatilaka. He is investigating the mechanisms that<br />
lead to ion selectivity in biological molecules using<br />
computational approaches. Hopefully by now he<br />
has submitted his thesis and is enjoying life to the<br />
fullest.<br />
Abstract<br />
Many biological molecules are able to differentiate<br />
between two extremely similar ions, Na+ and K+.<br />
Different molecules are able to achieve this<br />
remarkable feat by using a combination <strong>of</strong> different<br />
mechanisms.[1] However, these mechanisms<br />
focus on the interactions occuring at the ion<br />
binding site and do not take into account<br />
interactions that may be induced away from the<br />
binding site, such as strain in the molecular<br />
scaffold or induced conformational changes.<br />
These changes may contribute to the selectivity <strong>of</strong><br />
an ion binding site but are difficult to predict<br />
without prior knowledge <strong>of</strong> the structure and<br />
dynamics <strong>of</strong> the molecule. As such, a general<br />
model <strong>of</strong> the role <strong>of</strong> strain in ion selectivity will be<br />
difficult, if not impossible, to construct. [2]<br />
The role <strong>of</strong> strain and conformational changes can<br />
be characterised in specific cases so as to obtain<br />
an understanding <strong>of</strong> what may be occuring in<br />
other molecules. In this investigation, this role is
elucidated for two ion selective bacterial<br />
ionophores: valinomycin and nonactin. Several<br />
intramolecular hydrogen bonds hold valinomycin in<br />
a rigid structure, thought to be necessary for<br />
achieving ion selectivity. Nonactin, on the other<br />
hand, lacks these bonds, making the comparison<br />
between the two particularly interesting.<br />
[1] Thomas, M., Jayatilaka, D., Corry, B., Biophys. J, 2011, 100,<br />
60-69<br />
[2] Bostick, D.L., Brooks, C.L., Biophys. J., 2009, 96, 4470-4492<br />
Keynote Session - Main Theatre<br />
K.7 3:30 – 4:00 pm<br />
Molecular approaches to next<br />
generation photovoltaic energy<br />
conversion<br />
Timothy W. Schmidt1 ,Yuen Yap Cheng1 ,<br />
Burkhard Fückel1 , Murad Tayebjee1 , Raphaël<br />
Clady1 , Nicholas J. Ekins-Daukes 2 , Maxwell J.<br />
Crossley1 , Klaus Lips3 ,<br />
1 School <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> Sydney, NSW 2006<br />
2 Department <strong>of</strong> Physics and Grantham Centre for Climate<br />
Change, Imperial College, London<br />
3 Institut für Silizium-Photovoltaik, Helmholtz Zentrum-Berlin für<br />
Energie und Materialen<br />
Biography<br />
Tim Schmidt gained his BSc (Hons) from this<br />
university in 1998 winning the <strong>University</strong> Medal in<br />
theoretical chemistry. He then studied at Churchill<br />
College, Cambridge, gaining a PhD in chemistry<br />
from the <strong>University</strong> <strong>of</strong> Cambridge in 2001 for work<br />
on the femtosecond dynamics <strong>of</strong> molecules in<br />
intense laser field under the supervision <strong>of</strong> Dr<br />
Gareth Roberts. Postoctoral work was performed<br />
in the group <strong>of</strong> Pr<strong>of</strong>essor John Paul Maier in Basel<br />
on the electronic spectroscopy <strong>of</strong> highly<br />
unsaturated hydrocarbons <strong>of</strong> astrophysical<br />
relevance. Tim returned to Australia in 2003 to<br />
work at the CSIRO (CTIP, Lindfield) on modelling <strong>of</strong><br />
the rubsico enzyme. He commenced as a lecturer<br />
in chemistry in April 2004. From January 2011, Tim<br />
is appointed as Associate Pr<strong>of</strong>essor. He was the<br />
2010 recipient <strong>of</strong> the Coblentz Award, and the<br />
inaugural recipient <strong>of</strong> the Lecturership <strong>of</strong> the<br />
Physical Chemistry Division <strong>of</strong> the RACI.<br />
Biographies and Abstracts<br />
Monday 5 December - Session 4<br />
Abstract<br />
Single threshold solar cells, by their design, are<br />
limited to a maximum energy conversion efficiency<br />
<strong>of</strong> 33% under the standard AM1.5G spectrum.<br />
While this number depends on the absorption<br />
threshold <strong>of</strong> the material, many first generation<br />
technologies such as crystalline silicon are<br />
approaching this limit. By sacrificing efficiency,<br />
cost savings can be made by using cheaper<br />
processing methods and a smaller amount <strong>of</strong><br />
photoactive material. These cheaper cells, referred<br />
to as second generation solar cells, include<br />
technologies such as thin-film silicon and organic<br />
(plastic) photovoltaics. The field <strong>of</strong> third generation<br />
photovoltaics seeks to build on the cost savings <strong>of</strong><br />
the second generation cells while circumventing<br />
the single threshold limit. One strategy in this<br />
direction is to transform the solar spectrum by<br />
upconversion, where long wavelength light used<br />
poorly by a single threshold solar cell is converted<br />
to a shorter wavelength <strong>of</strong> more utility to the cell.<br />
In our laboratory, we have been investigating the<br />
phenomenon known as triplet-triplet annihilation<br />
upconversion (TTA-UC), an upconversion<br />
technology which uses organic molecules to<br />
harvest low energy light, and conjoin the energy <strong>of</strong><br />
two photons to bring about higher energy<br />
radiation. This technique is extremely flexible with<br />
regard to wavelength: Using a range <strong>of</strong> porphyrin<br />
molecules, we have succeeded in upconverting<br />
red to blue, red to green, red to yellow and green<br />
to blue. Experiments have shown that TTA-UC can<br />
proceed with high efficiency. In the tens <strong>of</strong><br />
microseconds following laser excitation, we find as<br />
many as 33% <strong>of</strong> the absorbed quanta take part in<br />
the TTA-UC process, with instantaneous<br />
efficiencies exceeding 40%.[1,2] Moreover, kinetic<br />
analysis reveals that the process would reach its<br />
maximum at a value exceeding 60% (where 100%<br />
represents the maximum quantum efficiency <strong>of</strong><br />
50%). I will present experiments demonstrating<br />
enhancements in the energy conversion <strong>of</strong> an<br />
amorphous silicon solar cell, due to the<br />
upconversion unit.<br />
1. Y. Y. Cheng, T. Khoury, R. Clady, M. J. Y. Tayebjee, N. J.<br />
Ekins-Daukes, M. J. Crossley, and T. W. Schmidt, “On the<br />
efficiency limit <strong>of</strong> triplet-triplet annihilation for photochemical<br />
upconversion,” Physical Chemistry Chemical Physics 12(1), 66-71<br />
(2010).<br />
2. Y. Y. Cheng, B. Fückel, T. Khoury, R. l. G. C. R. Clady, M. J. Y.<br />
Tayebjee, N. J. Ekins-Daukes, M. J. Crossley, and T. W. Schmidt,<br />
65
66<br />
Biographies and Abstracts<br />
Monday 5 December - Session 4<br />
“Kinetic Analysis <strong>of</strong> Photochemical Upconversion by Triplet-Triplet<br />
Annihilation: Beyond Any Spin Statistical Limit,” The Journal <strong>of</strong><br />
Physical Chemistry Letters 1(12), 1795-1799 (2010).<br />
3. N. J. Ekins-Daukes and T. W. Schmidt, “A molecular approach<br />
to the intermediate band solar cell: The symmetric case,” Applied<br />
Physics Letters 93(6)(2008).<br />
K.8 4:00pm – 4:30pm<br />
Charge photo-generation and<br />
recombination in conjugated<br />
polymer donor/ fullerene acceptor<br />
bulk heterojunction solar cells<br />
Attila J. Mozer, Tracey M. Clarke<br />
ARC Centre <strong>of</strong> Excellence for Electromaterials Science, Intelligent<br />
Polymer Research Institute, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>, AIIM<br />
Building, Innovation Campus, Squires way, North <strong>Wollongong</strong>,<br />
NSW 2500, Australia,<br />
attila@uow.edu.au, tclarke@uow.edu.au<br />
Biography<br />
Dr Mozer is an Australian Research Fellow (2011 -)<br />
and a Senior Research Fellow (2010 -) at the<br />
Intelligent Polymer Research Institute at the<br />
<strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>. Dr Mozer is the lead<br />
chief investigator <strong>of</strong> three Australian Research<br />
Council grants focused on developing costefficient<br />
solar energy conversion systems using<br />
organic solar cell technology. His main expertise is<br />
studying solar cell efficiency-determining charge<br />
photo-generation and recombination reactions<br />
using time-resolved optical and electronic probes.<br />
Dr Mozer has a significant track record in two <strong>of</strong><br />
the major organic solar cell architectures: donor /<br />
acceptor polymer bulk heterojunction solar cells<br />
and dye-sensitised photo-electrochemical solar<br />
cells.<br />
He has an h index <strong>of</strong> 14, and has published (since<br />
2004) three book chapters and 33 publications<br />
with total citations exceeding 600. He has<br />
obtained his PhD from Pr<strong>of</strong>. Serdar Sariciftci, the<br />
invertor <strong>of</strong> polymer solar cell technology at the Linz<br />
Institute for Organic Solar Cells, Johannes Kepler<br />
<strong>University</strong>, Linz, Austria in 2004. He has obtained<br />
his MSc in Chemical Engineering from the<br />
Budapest <strong>University</strong> <strong>of</strong> Technology and<br />
Economics in 2002. Dr Mozer has received<br />
research fellowships including the Australian<br />
Research Council ARF Fellowship (2011 -), the<br />
Japanese Society for the Promotion <strong>of</strong> Science<br />
Short-Term Visiting Fellowship (2011 -) and<br />
Postdoctoral Fellowship (2005). He is the Team<br />
Leader <strong>of</strong> the Solar Energy Research Strength at<br />
the Intelligent Polymer Research Institute, and the<br />
member <strong>of</strong> the ARC Centre <strong>of</strong> Excellence for<br />
Electromaterials Science (ACES) Centre Executive.<br />
Abstract<br />
Ultrafast photo-induced electron transfer between<br />
a photo-excited conjugated polymer (electron<br />
donor) and fullerene (acceptor) was first reported<br />
in 1992 by Sariciftci et al.1 This initial discovery has<br />
led to a number <strong>of</strong> important applications in photodetectors,<br />
bio-sensors, light-sensitive field-effect<br />
transistors and plastic solar cells.2 The efficiency<br />
<strong>of</strong> the first bulk-heterojunction (plastic) solar cells,<br />
where an intimate mixing <strong>of</strong> the electron donor /<br />
electron acceptor phases created large surface<br />
area for efficient charge separation, was < 1%.<br />
This year, the best reported certified efficiencies<br />
surpassed 8%3 making this low-cost technology<br />
commercially viable. Two <strong>of</strong> the most<br />
fundamentally important questions still unresolved<br />
are (i) “What is the minimum thermodynamic<br />
driving force (approximately corresponding to the<br />
potential difference between the ionisation<br />
potential <strong>of</strong> photo-excited donor and the electron<br />
affinity <strong>of</strong> the acceptor) required for efficient charge<br />
separation?”; (ii) “What governs charge<br />
recombination <strong>of</strong> the photo-induced charge<br />
carriers and how to minimise charge<br />
recombination in a well-intermixed donor/acceptor<br />
blend with high internal surface area?” Relevant to<br />
answering both <strong>of</strong> these questions is determining<br />
the optimum donor/acceptor domain size leading<br />
to high charge separation yield and slow<br />
recombination.<br />
In this presentation we will show that relatively<br />
large driving force is required for efficient charge<br />
separation in the benchmark regioregular<br />
poly-3-hexylthiophene / PCBM bulk<br />
heterojunction.4 Using sub-ns and femtosecond<br />
transient absorption spectroscopy, the kinetics <strong>of</strong><br />
charge photo-generation in a series <strong>of</strong> low<br />
bandgap polymers with deep lying LUMO energy<br />
and thus small thermodynamic driving force will be<br />
presented.<br />
Furthermore, in 2005 we have shown using<br />
integral mode time-<strong>of</strong>-flight and photo-induced
charge extraction by linearly increasing voltage<br />
(photo-CELIV) techniques that bimolecular<br />
recombination is significantly reduced in<br />
regioregular P3HT/PCBM mixture compared to the<br />
widely-accepted diffusion-controlled Langevin<br />
recombination model.5 This observation was<br />
attributed to the highly ordered, nan<strong>of</strong>ibre-like<br />
morphology <strong>of</strong> P3HT in annealed P3HT/PCBM<br />
blends, reducing the chance for the electron and<br />
hole to meet and recombine. This year, we have<br />
reported reduced recombination in a novel<br />
silole-based low bandgap polymer/PCBM blend,6<br />
which enables the fabrication <strong>of</strong> relatively thick (up<br />
to 300 nm) photoactive layers without a significant<br />
drop in charge collection efficiency. A chemically<br />
very similar polymer mixed with PCBM shows the<br />
typically observed Langevin recombination<br />
kinetics, yet its morphology is virtually identical to<br />
the reduced recombination blend, indicating that<br />
the origin <strong>of</strong> reduced recombination may be much<br />
more complex than originally though. Alternative<br />
explanations will be presented.<br />
1. N.S. Sariciftci, L. Smilowitz, A.J. Heeger and F. Wudl, Science<br />
258 (1992) 1474.<br />
2. C. J. Brabec, N. S. Sariciftci, and J. C. Hummelen, Adv. Funct.<br />
Mater. 11 (2001) 11, 15<br />
3. http://www.konarka.com/index.php/site/pressreleasedetail/<br />
konarkas_power_plastic_achieves_world_record_83_<br />
efficiency_certification_fr<br />
4. T. M. Clarke and J. R. Durrant, Chem. Rev. 110 (2010) 6736.<br />
5. A. Pivrikas, G. Juska, A. J. Mozer, M. Scharber, K. Arlauskas, N.<br />
S. Sariciftci, H. Stubb, and R. Osterbacka, Phys. Rev. Lett. 94<br />
(2005) 176806-1<br />
6. T. M. Clarke, D. B. Rodovsky, A. A. Herzing, J. Peet, G. Dennler,<br />
D. DeLongchamp, C. Lungenschmied, and A. J. Mozer, Adv.<br />
Energy Mater. (2011), published online, DOI: 10.1002/<br />
aenm.201100390<br />
K.9 4:30pm – 5:00pm<br />
Towards High-Efficiency Microalgae<br />
Bi<strong>of</strong>uel Systems<br />
Ben Hankamer<br />
Institute for Molecular Bioscience, The <strong>University</strong> <strong>of</strong> Queensland,<br />
Brisbane, Qld 4072, Australia<br />
Email : b.hankamer@imb.uq.edu.au, Tel : +61 7 334 62012, Fax :<br />
+61 7 334 62103<br />
Biography<br />
PhD: Imperial College London (1990-1994)<br />
Post Doc/Research lecturer: Imperial College<br />
London (1994-2002)<br />
Biographies and Abstracts<br />
Monday 5 December - Session 4<br />
Principle Investigator: Institute for Molecular<br />
Bioscience, The <strong>University</strong> <strong>of</strong> Queensland<br />
(2002-onwards)<br />
Director: Solar Bi<strong>of</strong>uels Consortium (www.<br />
solarbi<strong>of</strong>uels.org) (2006-onwards)<br />
Over the past 10 years, Ben Hankamer has<br />
focused on the development <strong>of</strong> environmentally<br />
friendly high-efficiency bi<strong>of</strong>uel production systems.<br />
This area represents a rapidly expanding<br />
biotechnology. His specialization is in the structural<br />
biology <strong>of</strong> the photosynthetic machinery, which<br />
drives the conversion <strong>of</strong> solar energy into chemical<br />
energy (fuels) and has published extensively on the<br />
water splitting Photosystem II complex, its light<br />
harvesting antenna system and V-type ATPase<br />
(Nature, Nature Structural Biology, TIBS, PNAS).<br />
Using this knowledge <strong>of</strong> the photosynthetic<br />
machinery, he embarked on the targeted<br />
engineering <strong>of</strong> the green alga Chlamydomonas<br />
reinhardtii for high-efficiency bi<strong>of</strong>uel production. To<br />
facilitate the development <strong>of</strong> high efficiency bi<strong>of</strong>uel<br />
systems he founded the Solar Bi<strong>of</strong>uels Consortium<br />
(www.solarbi<strong>of</strong>uels.org) which he now directs. The<br />
consortium includes eight international teams and<br />
conducts economic analysis, bio-discovery,<br />
marine biology, structural biology, molecular<br />
biology, microbiology, genomics, metabolomics,<br />
culture optimisation and bioreactor scale up within<br />
a coordinated research program <strong>of</strong> parallel<br />
research streams.<br />
Abstract<br />
The Stern Review (The Economics <strong>of</strong> Climate<br />
Change) and the reports by the Intergovernmental<br />
Panel on Climate Change have left little doubt that<br />
concerted action is needed to develop CO2 free<br />
energy technologies. Indeed to stabilize global<br />
temperature increases below 2oC as agreed in the<br />
United Nations Climate Change Conference in<br />
Cancun has been reported to require CO2<br />
emissions stabilization by 2010-2015.<br />
The importance <strong>of</strong> CO2 neutral fuels in this<br />
process is highlighted by the fact that fuels make<br />
up ~80% <strong>of</strong> the global energy market. In contrast<br />
global electricity demand accounts for only 17%.<br />
Yet despite the importance <strong>of</strong> fuels, almost all CO2<br />
free energy production systems under<br />
development are designed to drive electricity<br />
generation (e.g. clean-coal technology, nuclear,<br />
67
68<br />
Biographies and Abstracts<br />
Monday 5 December - Session 4<br />
photovoltaic & wind). In contrast, and indeed<br />
almost uniquely, bio-fuels target the much larger<br />
fuel market and so in the future, have clear<br />
potential to play an increasingly important role. The<br />
presentation will provide an overview <strong>of</strong> the<br />
physical constraints <strong>of</strong> photobiological bi<strong>of</strong>uel<br />
production in terms <strong>of</strong> area, solar energy and<br />
nutrient requirements before presenting advances<br />
made in terms <strong>of</strong> developing microalgal bi<strong>of</strong>uel<br />
systems.<br />
Notes<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................
Session 5 – Theatre 2<br />
5.1.1 10:30am – 10:45am<br />
A novel Molecular Dynamics<br />
approach for quantitative prediction<br />
<strong>of</strong> adhesion and wettability:<br />
application to responsive surfaces<br />
George Yiapanis1 , David Henry2 , Evan<br />
Evans3 , Irene Yarovsky1 1 Applied Sciences, RMIT <strong>University</strong>, GPO BOX 2476V, 3001,<br />
Melbourne, Victoria<br />
2 Chemical and Mathematical Sciences, Murdoch <strong>University</strong>,<br />
Western Australia<br />
3 BlueScope Steel Research, Port Kembla, NSW<br />
Biographies and Abstracts<br />
Tuesday 6 December - Session 5<br />
Biography<br />
George Yiapanis received his doctoral degree in<br />
applied physics from RMIT <strong>University</strong> in Melbourne<br />
in 2010. He is currently a postdoctoral fellow at<br />
RMIT <strong>University</strong>, in the Materials Modelling<br />
Simulation group, directed by Pr<strong>of</strong>. Irene Yarovsky.<br />
His research interest is exploring full atomistic<br />
modelling <strong>of</strong> various organic and inorganic<br />
surfaces and interfaces. Presently, he is working<br />
on the design <strong>of</strong> responsive self-cleaning polymer<br />
surfaces, and <strong>of</strong> self-assembled monolayers as<br />
stable evaporation suppressants.<br />
Abstract<br />
Adhesion is an important property that dominates<br />
a multitude <strong>of</strong> processes ranging from fouling to<br />
nano-particle formation. For a two component<br />
system, adhesion can be readily measured<br />
through atomic force microscopy or contact angle<br />
measurements.1,2 However, acquiring a<br />
quantitative prediction <strong>of</strong> adhesion within a<br />
three-component system, is not an easy task. An<br />
example <strong>of</strong> a three-component system includes a<br />
surface interacting with a dirt particle in a liquid<br />
medium. Here, using atomistic Molecular Dynamic<br />
modelling techniques, we developed an approach<br />
to carry out this exact task, allowing us to predict<br />
the dirt-shielding qualities <strong>of</strong> various responsive<br />
polymeric brushes. The surface models have been<br />
carefully designed to mimic the responsive<br />
stay-clean behaviour <strong>of</strong> the Lady’s Mantle leaf,3<br />
and include polyethylene glycol grafted organic<br />
and inorganic substrates.<br />
Our novel approach involves measuring the<br />
contact-angle <strong>of</strong> nano-sized water droplets on s<strong>of</strong>t<br />
deformable surfaces (Figure 1a.). Using this new<br />
approach together with our previously developed<br />
in-silico force measurements4, we investigate the<br />
adhesion <strong>of</strong> the carbon based contaminants5 onto<br />
the adaptive surfaces in both dry and aqueous<br />
environments. Our results show that polyethylene<br />
glycol brushes impart dirt-shielding qualities<br />
provided that the substrate is sufficiently<br />
hydrophilic and robust (Figure 1b.). Analogous to<br />
the flexible hairs <strong>of</strong> the Lady’s Mantle leaf, the<br />
polyethylene glycol molecules provide a repulsive<br />
energy barrier that prevents adhesion <strong>of</strong><br />
contaminants in water. Our new approach can be<br />
applied to test wettability and adhesion <strong>of</strong><br />
essentially any material<br />
Figure 1 a. Contact angle measurements using classical molecular<br />
dynamics. b. Contour map <strong>of</strong> an adaptive surface which<br />
comprises <strong>of</strong> PEG brushes on top <strong>of</strong> a robust silica substrate.<br />
(1) Almazán-Almazán, M. C.; Paredes, J. I.; Pérez-Mendoza, M.; M.<br />
Domingo-García a; López-Garzón, F. J.; Martínez-Alonso, A.;<br />
Tascón, J. M. D. Journal <strong>of</strong> Colloid and Interface Science 2006,<br />
293, 353.<br />
(2) Lee, S. A.; Oh, S. H.; Lee, W. Journal <strong>of</strong> Colloid and Interface<br />
Science 2009, 332, 461.(3) Otten, A.; Herminghaus, S.<br />
Langmuir 2004, 20, 2405.<br />
(4) Yiapanis, G.; Evans, E.; Henry, D. J.; Yarovsky, I. J Phys Chem C<br />
2010, 114, 478.<br />
(5) Yiapanis, G.; Henry, D. J.; Evans, E.; Yarovsky, I. In<br />
Nanotechnology in Australia: Showcase <strong>of</strong> Early Career<br />
Research in Australia; Kane, D. M., Micolich, A. P., Rabeau, J.<br />
R., Eds.; Pan Stanford Publishing: 2011.<br />
69
70<br />
Biographies and Abstracts<br />
Tuesday 6 December - Session 5<br />
5.1.2 10:45am – 11:00am<br />
What can we learn from large-scale<br />
ab initio calculations <strong>of</strong> ionic liquids?<br />
Ekaterina I Izgorodina1 1 School <strong>of</strong> Chemistry, Monash <strong>University</strong>, Wellington Rd,<br />
Clayton, VIC 3800, AUSTRALIA<br />
Biography<br />
Dr Ekaterina (Katya) Pas obtained her PhD from<br />
the <strong>University</strong> <strong>of</strong> Muenster, in the field <strong>of</strong><br />
Theoretical Chemistry in 2004. Immediately after<br />
her PhD Katya accepted a postdoctoral position<br />
with Dr Michelle Coote at ANU to study free radical<br />
and RAFT polymerisation. In 2006 she moved to<br />
Monash <strong>University</strong> as a Research Fellow in the<br />
Ionic Liquid group <strong>of</strong> Pr<strong>of</strong>. Doug MacFarlane. In<br />
2007 she was awarded an ARC APD Fellowship to<br />
study ionic materials from the first principles and<br />
appointed as a lecturer in the School <strong>of</strong> Chemistry.<br />
Currently, Katya is a Senior Lecturer at Monash<br />
Univeristy leading a research group that<br />
specialises in fully ab initio calculations <strong>of</strong> ionic<br />
liquids and liquid electrolytes.<br />
Abstract<br />
Large-scale calculations <strong>of</strong> archetypical ionic<br />
liquids consisting up to eight ion pairs1 were<br />
performed at the MP2 level <strong>of</strong> theory in<br />
combination with a triple-ξ doubly polarised<br />
Ahlrichs type basis set, TZVPP. These calculations<br />
showed that dispersion interactions due to<br />
electron correlation increased rapidly with<br />
increasing number <strong>of</strong> ion pairs in the cluster. For<br />
imidazolium-based ionic liquids the dispersion<br />
contribution was up to 20% <strong>of</strong> the overall energy,<br />
thus emphasising the importance <strong>of</strong> electron<br />
correlation effects as cluster size increases. These<br />
results also reflect the presence <strong>of</strong> all possible<br />
inter-ion interactions, including like-ion dispersion<br />
interactions such as inter-alkyl chains. Using the<br />
Fragment Molecular Orbital approach in<br />
combination with the MP2 level <strong>of</strong> theory2<br />
three-body inter-ion interactions were established<br />
to be necessary in describing Coulomb forces with<br />
accuracy below 1 kJ mol-1, whereas two-body<br />
corrections were already enough for dispersion<br />
forces. A thorough analysis <strong>of</strong> charge transfer<br />
between ions with increasing cluster size was<br />
performed to understand its influence on the<br />
Coulomb and induction forces in ionic systems. To<br />
quantitatively evaluate the fundamental<br />
components <strong>of</strong> the interaction energy such as<br />
Coulomb, induction, repulsion, charge transfer and<br />
dispersion, accurate energy decomposition<br />
schemes are required. Currently available<br />
schemes either cannot be used beyond a single<br />
ion pair such as Symmetry-Adapted Perturbation<br />
Theory (SAPT)3 or are not designed to treat<br />
charged species such as Effective Fragment<br />
Potential (EFP).4 Here we present a detailed<br />
analysis <strong>of</strong> these two widely used decomposition<br />
schemes in attempt to devise a new scheme for<br />
decomposing interaction energies <strong>of</strong> ionic systems<br />
into fundamental components.<br />
5.1.3 11:00am – 11:15am<br />
Formation <strong>of</strong> Radical Products From<br />
Activation <strong>of</strong> Phospholipid Ozonides<br />
Shane Ellis<br />
<strong>University</strong> <strong>of</strong> <strong>Wollongong</strong><br />
Biography<br />
Shane Ellis completed his degree in<br />
Nanotechnology from the <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong><br />
in 2008. He is currently in the 3rd year <strong>of</strong> his PhD<br />
at the <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong> looking at the<br />
applications <strong>of</strong> surface ionization mass<br />
spectrometry approaches for lipid analysis and<br />
imaging. He spent 2 months at Pr<strong>of</strong>essor Graham<br />
Cooks Lab at Purdue <strong>University</strong>, Indiana, USA<br />
looking at the distribution <strong>of</strong> lipids in the human<br />
lens by desorption electrospray ionization mass<br />
spectrometry imaging. His research is partly<br />
funded by the Centre <strong>of</strong> Excellence for Free<br />
Radical Chemistry and Biotechnology.<br />
Abstract<br />
Ozone is responsible for the oxidation <strong>of</strong> many<br />
unsaturated organic compounds in the<br />
atmosphere. These reactions can occur in the gas<br />
phase or on surfaces such as those provided by<br />
organic aerosols. Importantly, ozonolysis can give<br />
rise to radical species (e.g. OH radicals) that are<br />
able to propagate further oxidation, although the<br />
precise mechanisms <strong>of</strong> radical formation are not<br />
well understood. Using desorption electrospray
Biographies and Abstracts<br />
Tuesday 6 December - Session 5<br />
ionisation mass spectrometry we have observed<br />
products arising from the reaction <strong>of</strong> ambient<br />
ozone (present in laboratory air) with unsaturated<br />
lipids. For example, analysis <strong>of</strong> the unsaturated<br />
phosphatidylcholine (PC), PC (18:1n9/16:0) gives<br />
rise to an abundant ion 48 Da above the [M+Na]+<br />
ion <strong>of</strong> the parent lipid. Activation <strong>of</strong> the putative<br />
ozonide ion, [M+Na+O3]+, by collision induced<br />
dissociation (CID) on an ion-trap mass<br />
spectrometer revealed ions corresponding to the<br />
mass <strong>of</strong> aldehydes and carbonyl oxides (i.e.,<br />
Criegee intermediates) although the structure <strong>of</strong><br />
the latter has not been determined. Such ions are<br />
shown to be indicative <strong>of</strong> the double bond position<br />
and have analytical utility in the structure<br />
elucidation <strong>of</strong> naturally occurring unsaturated<br />
lipids. In addition, dissociation <strong>of</strong> the ozonides<br />
yields several product ions with masses<br />
suggestive <strong>of</strong> odd–electron species. These radical<br />
ions are proposed to form following homolytic<br />
cleavage <strong>of</strong> the O-O bond <strong>of</strong> the secondary<br />
ozonide and consistent with this, their mass is<br />
observed to shift with the double bond position(s)<br />
in the parent lipid. The structure <strong>of</strong> these radical<br />
ions has been further probed using subsequent<br />
fragmentation via CID (i.e., MSn) and ion-molecule<br />
reactions with adventitious dioxygen present in the<br />
ion-trap. Both approaches show behaviour<br />
consistent with radical formation. These results<br />
confirm the production <strong>of</strong> radicals directly from the<br />
decomposition <strong>of</strong> activated secondary ozonides in<br />
the gas phase and may have significance for<br />
understanding the fate <strong>of</strong> long-lived ozonides in<br />
the atmosphere. Finally the characteristic<br />
fragmentation behaviour <strong>of</strong> secondary ozonides<br />
(described above) will be compared to<br />
[M+Na+O3]+adducts formed in gas phase<br />
encounters between ozone and ionized,<br />
unsaturated lipids [1] and may provide evidence for<br />
the existence <strong>of</strong> the more elusive primary ozonide<br />
in the latter experiment.<br />
References<br />
(1) Poad, B. L. J., Pham, H. T., Thomas, M. C., Nealon, J. R.,<br />
<strong>Campbell</strong>, J. L., Mitchell, T. W., Blanksby, S. J., Ozone-Induced<br />
Dissociation on a Modified Tandem Linear Ion-Trap: Observations<br />
<strong>of</strong> Different Reactivity for Isomeric Lipids. J. Am. Soc. Mass<br />
Spectrom. 2010, 21, 1989-1999.<br />
5.1.4 11:15am – 11:30am<br />
The Distal Effect <strong>of</strong> Electron-<br />
Withdrawing Groups on the Stability<br />
<strong>of</strong> Peptide Enolates and its<br />
Exploitation in Synthesis<br />
Junming Ho1 , Christopher J. Easton1 , Michelle<br />
L. Coote1 1 ARC Centre <strong>of</strong> Excellence for Free Radical Chemistry and<br />
Biotechnology, Research School <strong>of</strong> Chemistry, Australian<br />
National <strong>University</strong>, Canberra, ACT 0200, Australia<br />
Biography<br />
Junming Ho is a post-doctoral fellow with Michelle<br />
Coote at the ANU where he also recently<br />
completed his PhD. His research involves the use<br />
<strong>of</strong> computational and practical chemistry to<br />
understand chemical reactivity and their<br />
associated applications in synthesis and<br />
biochemistry.<br />
Abstract<br />
Normally amides and esters exhibit much weaker<br />
carbon acidities than ketones mainly as a result <strong>of</strong><br />
the effects <strong>of</strong> the various carbonyl carbon<br />
substituents to attenuate resonance stabilisation <strong>of</strong><br />
the corresponding enolates. In a recent<br />
computational study,1 we predicted that<br />
N-electron-withdrawing substituents, hydrogen<br />
bonding and protonation at amide nitrogen<br />
selectively increase the acidity <strong>of</strong> a distal proton<br />
adjacent to the amide carbonyl, to the extent that<br />
the a-carbonyl acidity <strong>of</strong> some N-substituted<br />
amides exceeds that <strong>of</strong> typical ketones. In this<br />
contribution, the origin <strong>of</strong> the distal effect is<br />
examined using high-level ab initio methods and<br />
its relevance to certain enzymatic reactions is<br />
highlighted. Using a combination <strong>of</strong> theory and<br />
experiment, the synthetic utility <strong>of</strong> the effect is also<br />
demonstrated through the controlled<br />
stereochemical inversion <strong>of</strong> amino acid<br />
derivatives.2<br />
1. Ho, J.; Easton, C. J.; Coote, M. L. J. Am. Chem. Soc. 2010,<br />
132,5515.<br />
2. Ho, J.; Coote, M. L.; Easton, C. J. J. Org. Chem. 2011, 76,<br />
5907.<br />
71
72<br />
Biographies and Abstracts<br />
Tuesday 6 December - Session 5<br />
5.1.5 11:30am – 11:45am<br />
Molecular magnetic properties:<br />
benchmarking and applications<br />
David J. Wilson1 , Trygve Helgaker 2 , Antonio<br />
Rizzo3 1 La Trobe Institute for Molecular Science, La Trobe <strong>University</strong>,<br />
Melbourne, Australia.<br />
2 Centre for Theoretical and Computational Chemistry, <strong>University</strong><br />
<strong>of</strong> Oslo, Norway.<br />
3 ICPF-CNR, UoS di Pisa, Area della Riccerca, via G. Moruzzi 1,<br />
56124 Pisa, Italy.<br />
Biography<br />
David Wilson is a senior lecturer in the Dept <strong>of</strong><br />
Chemistry at La Trobe <strong>University</strong>, which he joined<br />
in 2005. He completed a PhD under Pr<strong>of</strong> Ellak von<br />
Nagy-Felsobuki in Newcastle, before a<br />
postdoctoral position at the <strong>University</strong> <strong>of</strong> Oslo with<br />
Pr<strong>of</strong>. Trygve Helgaker. Dr Wilson is a contributing<br />
author to the Dalton program. His research<br />
interests include theoretical development and<br />
applications, which span from small molecules<br />
(gas-phase chemistry) to ‘medium’ size (metal<br />
chemistry) and very large molecules (biomolecular<br />
modeling).<br />
Abstract<br />
We report benchmark studies <strong>of</strong> DFT and<br />
CCSD(T) calculated magnetizabilities for a series<br />
<strong>of</strong> 22 fluorine-containing molecules, systems for<br />
which the calculation <strong>of</strong> magnetic properties is<br />
particularly challenging. All calculations make use<br />
<strong>of</strong> London atomic orbitals (the GIAO approach),<br />
while basis sets up to aug-cc-pV6Z have been<br />
employed. Statistical analysis <strong>of</strong> results provides<br />
insight into the performance <strong>of</strong> various density<br />
functionals in the calculation <strong>of</strong> magnetic<br />
properties.<br />
Several applications will be presented, with results<br />
compared to available experimental data. This<br />
includes the set <strong>of</strong> fluorine containing molecules,<br />
for which the link with aromaticity will be explored.<br />
An investigation <strong>of</strong> PF3 indicates unusually slow<br />
basis set performance (up to aug-cc-pV6Z), and<br />
the need to explicit core basis functions (up to<br />
aug-cc-pCV5Z). Results for ferrocene will also be<br />
presented. These coupled cluster results represent<br />
the largest and likely most accurate calculations<br />
ever reported for molecular properties <strong>of</strong> this<br />
fascinating molecule.<br />
5.1.6 11:45am – 12:00pm<br />
Molecular Design Rules for<br />
Frequency-Based, Universal<br />
Quantum Computers<br />
Laura K. McKemmish 1,2* , David J.<br />
Kedziora3 , Graham R. White3 , Noel S. Hush4 ,<br />
Jeffrey R. Reimers1 1 School <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> Sydney, Sydney, NSW<br />
2006 Australia<br />
2 Research School <strong>of</strong> Chemistry, Australian National <strong>University</strong>,<br />
Canberra, ACT 2601 Australia<br />
3 School <strong>of</strong> Physics, The <strong>University</strong> <strong>of</strong> Sydney, Sydney, NSW<br />
2006 Australia<br />
4 School <strong>of</strong> Molecular Biosciences, The <strong>University</strong> <strong>of</strong> Sydney,<br />
Sydney, NSW 2006 Australia<br />
* laura.mckemmish@gmail.com<br />
Biography<br />
Laura McKemmish completed her Honours at the<br />
<strong>University</strong> <strong>of</strong> Sydney in 2010, where her research<br />
on frequency-based quantum computers and<br />
quantifying the accuracy <strong>of</strong> adiabatic<br />
approximations was inspired by the benefits that<br />
considering theoretical chemistry and quantum<br />
computation together. She has just started a PhD<br />
at the Australian National <strong>University</strong> on developing<br />
a method <strong>of</strong> quantifying the accuracy <strong>of</strong><br />
computational chemistry calculations. She has<br />
researched a variety <strong>of</strong> topics, including<br />
spectroscopy, biophysics, computational<br />
chemistry (<strong>of</strong> the Creutz-Taube ion and aromatic<br />
dimers), ionic liquids, quantum consciousness<br />
and astronomy. She has chosen to specialise in<br />
theoretical chemistry.<br />
Abstract<br />
NMR is a widely used technique through chemistry<br />
and biochemistry, but the extensive experimental<br />
equipment and nuclear-spin-manipulation<br />
techniques that have been developed for these<br />
applications can also be applied to allow<br />
molecules to function as nuclear-spin-based<br />
quantum registers for quantum computing<br />
applications. Quantum computers and quantum<br />
information processors (QIPs) present a promising
Biographies and Abstracts<br />
Tuesday 6 December - Session 5<br />
paradigm <strong>of</strong> computing, including exponential<br />
speedup to computational simulation <strong>of</strong> quantum<br />
systems [1]. Quantum registers differ from the<br />
classical bits found in modern digital computers as<br />
they do not need to exist only in eigenstates but<br />
can also exist as an arbitrary linear combination <strong>of</strong><br />
eigenstates [2]; the simplest quantum register is a<br />
qubit made from a quantum system that has two<br />
discrete eigenstates, such as the spin <strong>of</strong> a 1H or<br />
13C nucleus. In an NMR-based QIP (or other<br />
molecular-based QIPs such as those built on<br />
rotational, electronic, or vibrational spectroscopy),<br />
systematically controlled electromagnetic pulses<br />
applied to the whole system implement all gates to<br />
allow computation[1]. Two types <strong>of</strong> gates are<br />
required in a QIP, one-qubit gates which act on<br />
single spins, and two-qubit gates which modify<br />
one spin based on the state <strong>of</strong> another.<br />
Over 46 molecules have been used in NMR-based<br />
QIPs, the two largest molecules studied containing<br />
7 and 12 qubits (independently controllable<br />
nuclear spins). Some molecules function well,<br />
others poorly. For example, histidine (12 qubits)<br />
allows the spin <strong>of</strong> an initially polarized chin proton<br />
to be passed to just 7 <strong>of</strong> other 11 qubits. We<br />
provide the first explanation <strong>of</strong> why this is the case,<br />
describing basic molecular design rules for NMR<br />
(etc) quantum register design. We show how<br />
standard GAUSSIAN calculations for the NMR (etc)<br />
spectra properties can be used to make<br />
quantitative predictions <strong>of</strong> functionality, as well as<br />
a series <strong>of</strong> simplistic chemical structure rules for<br />
qualitative prediction and analysis. Our analysis is<br />
based on the effects that spectral congestion has<br />
on inhibiting the operation <strong>of</strong> two-qubit gates.<br />
The ability to scale NMR quantum computers to<br />
larger sizes is severely limited because <strong>of</strong> many<br />
factors. Our analysis <strong>of</strong> the two-qubit gates shows<br />
that in general their construction becomes<br />
exponentially more difficult as the register size<br />
increases. By following our design rules, this<br />
scaling can be reduced to linear or near-linear<br />
scaling, thus enabling much larger registers to be<br />
demonstrated. We use GAUSSIAN calculations<br />
for an acetylene tetramer to show how a fully<br />
functional 25-qubit register could be designed.<br />
Session 5 – Main Theatre<br />
5.2.1 10:30am – 10:45am<br />
Single molecule fluorescence<br />
microscopy: visualising DNA<br />
replication and repair dynamics<br />
within living E. coli cells<br />
Andrew Robinson1 , Meghna Patel2 ,<br />
Elizabeth A. Wood3 , Roger Woodgate 4 , Michael M.<br />
Cox3 , Myron F. Goodman2 and Antoine M. van<br />
Oijen1 1 Zernike Institute for Advanced Materials, <strong>University</strong> <strong>of</strong><br />
Groningen, Groningen 9747 AG, The Netherlands<br />
2 Departments <strong>of</strong> Biological Sciences and Chemistry, <strong>University</strong><br />
<strong>of</strong> Southern California, Los Angeles CA 90089, USA<br />
3 Department <strong>of</strong> Biochemistry, <strong>University</strong> <strong>of</strong> Wisconsin-Madison,<br />
Madison, Wisconsin 53706, USA<br />
4 Laboratory <strong>of</strong> Genomic Integrity, National Institute <strong>of</strong> Child<br />
Health and Human Development, National Institutes <strong>of</strong> Health,<br />
Bethesda, Maryland 20892-3371, USA<br />
Biography<br />
Andrew completed his PhD research in 2007 in<br />
Bridget Mabbutt’s structural biology lab at<br />
Macquarie <strong>University</strong>, Sydney. He then moved to<br />
<strong>Wollongong</strong> to work as a postdoc in Nick Dixon’s<br />
biochemistry lab, where he used xray<br />
crystallography to study the proteins involved in<br />
DNA replication in bacteria. Late 2010 he moved to<br />
Antoine van Oijen’s single molecule biophysics lab<br />
at the <strong>University</strong> <strong>of</strong> Groningen in The Netherlands,<br />
where he is studying DNA replication and repair in<br />
live bacterial cells.<br />
Abstract<br />
Decades <strong>of</strong> genetic studies and in vitro analyses<br />
have painted a remarkably detailed picture <strong>of</strong> how<br />
bacteria maintain their chromosomes. By carefully<br />
coordinating the activities <strong>of</strong> individual DNA<br />
replication and repair proteins, bacteria are<br />
capable <strong>of</strong> faithfully maintaining their genetic<br />
information whilst dividing rapidly and responding<br />
to changing environmental conditions. Underlying<br />
this ability are highly dynamic protein interaction<br />
networks that are constantly remodelled in order<br />
to facilitate discontinuous replication <strong>of</strong> the<br />
chromosomal DNA and to allow any sites that<br />
become damaged to be repaired. While these<br />
networks and their players are now generally well<br />
73
74<br />
Biographies and Abstracts<br />
Tuesday 6 December - Session 5<br />
understood, how these processes are put into play<br />
during the course <strong>of</strong> a cell cycle and how stressed<br />
cells transition from DNA replication to repair<br />
remain to be determined.<br />
We are studying the coordination <strong>of</strong> DNA<br />
replication and repair processes in living E. coli<br />
cells using recently developed methods for<br />
fluorescence microscopy with single molecule<br />
level sensitivity. Such sensitivity is necessary as<br />
most DNA replication and repair proteins are<br />
maintained at very low copy number and are<br />
active at only a few select sites within the cell. We<br />
are using strains <strong>of</strong> E. coli in which subunits <strong>of</strong><br />
DNA pol III and DNA pol V are fused to bright<br />
fluorescent proteins, allowing us to measure<br />
cellular protein concentrations and quantify<br />
proteins localised at replication and repair sites.<br />
We have developed techniques that uniquely allow<br />
us to make such measurements within rapidly<br />
growing and dividing cells. By imaging cells within<br />
flow devices, we can also introduce DNA<br />
damaging agents and determine their effect on the<br />
replication and repair processes. We are using<br />
these techniques to characterise multi-fork<br />
replication in rapidly dividing cells and to monitor<br />
changes in the steady state concentration and<br />
localisation <strong>of</strong> DNA pol V following UV-induced<br />
DNA damage.<br />
5.2.2 10:45am – 11:00am<br />
Structured IlluminationMicroscopy<br />
<strong>of</strong> Living Cells<br />
Liisa M. Hirvonen, Trevor A. Smith<br />
Ultrafast and Microspectroscopy Laboratories, ARC Centre <strong>of</strong><br />
Excellence for Coherent X-Ray Science, School <strong>of</strong> Chemistry,<br />
<strong>University</strong> <strong>of</strong> Melbourne, Australia<br />
email: liisah@unimelb.edu.au<br />
Biography<br />
Liisa did her BsC and Phd in King’s College<br />
London. During her PhD, Liisa built one <strong>of</strong> the first<br />
setups for structured illumination microscopy<br />
under the supervision from Pr<strong>of</strong>essor Rainer<br />
Heintzmann. After completing her PhD, she took a<br />
position as a post-doc in the School <strong>of</strong> Chemistry,<br />
<strong>University</strong> <strong>of</strong> Melbourne, to develop superresolution<br />
microscopy techniques in the Ultrafast<br />
and Microspectroscopy Laboratories under the<br />
direction <strong>of</strong> Associate Pr<strong>of</strong>essor Trevor Smith.<br />
Abstract<br />
In recent years, several new techniques have been<br />
proposed for surpassing the diffraction limit in<br />
fluorescence microscopy [1]. One <strong>of</strong> these<br />
techniques is structured illumination microscopy<br />
(SIM) [2, 3], where a fine grating is<br />
projected onto the sample, and the final image is<br />
reconstructed froma set <strong>of</strong> images taken at<br />
different grating positions. Although the resolution<br />
improvement is limited to a factor <strong>of</strong> two, pulsed<br />
lasers, scanning or special properties <strong>of</strong> the<br />
fluorophores are not needed. It has been shown<br />
that SIM is fast enough to image slowly moving<br />
structures in living<br />
cells [4, 5]. SIM is based on the projection <strong>of</strong> a fine<br />
grating into the sample, and gathering information<br />
in the form <strong>of</strong> Moir´e fringes. When two highfrequency,<br />
i.e. high spatial resolution patterns are<br />
multiplied (superimposed), Moir´e fringes appear.<br />
These fringes carry information in low resolution<br />
format, which is observable through a normal light<br />
microscope. If the known grating pattern is then<br />
removed computationally from the unknown<br />
sample pattern, it is possible to restore previously<br />
unobservable details about the sample. For the<br />
restoration <strong>of</strong> the high resolution information a<br />
minimum <strong>of</strong> three images are needed where the<br />
grating pattern is moved between the images, and<br />
a minimum <strong>of</strong> 3 grating directions are needed for<br />
isotropic resolution improvement in the lateral<br />
plane.<br />
(a) (b) (c)<br />
Figure 1: Moir´e fringes: If an unknown sample<br />
pattern (a) is multiplied by a known illumination<br />
pattern (b), Moir´e fringes appear<br />
(c). If the illumination pattern is removed<br />
computationally, previously unobservable details<br />
can be restored. structured illumination provides a<br />
cheap and relatively easily implementable method<br />
for improving lateral resolution and reducing<br />
out-<strong>of</strong>-focus blur in fluorescence microscopy. No<br />
special properties are required from the<br />
fluorophores, and pulsed lasers and scanning are<br />
not needed. We are currently using this method to<br />
image both living and fixed mammalian cell
samples, as well as living yeast cells, polymers for<br />
solar cells, and botanical samples.<br />
References<br />
[1] L. M. Hirvonen, T. A. Smith, “Imaging on the nanoscale:<br />
Super-resolution fluorescence microscopy,” Australian Journal <strong>of</strong><br />
Chemistry 64, 41-45 (2011).<br />
[2] R. Heintzmann, C. Cremer, “Laterally modulated excitation<br />
microscopy: Improvement <strong>of</strong> resolution by using a diffraction<br />
grating,” Proceedings <strong>of</strong> SPIE 3568, 185-195 (1998).<br />
[3] M. G. L. Gustafsson, “Surpassing the lateral resolution limit by a<br />
factor <strong>of</strong> two using stuctured illumination microscopy,” Journal <strong>of</strong><br />
Microscopy 198, 82-87 (2000).<br />
[4] P. Kner, B. B. Chhun, E. Griffis, L.Winoto, M. G. L. Gustafsson,<br />
“Super-resolution video microscopy <strong>of</strong> live cells by structured<br />
illumination,”Nature Methods 6, 339-342 (2009).<br />
[5] L. M. Hirvonen, K. Wicker, O. Mandula, R. Heintzmann,<br />
“Structured illumination microscopy <strong>of</strong> a living cell,” European<br />
Biophysics Journal38, 807-812 (2009).<br />
5.2.3 11:00am – 11:15am<br />
Differential Dynamic Microscopy and<br />
Dynamic Light Scattering studies <strong>of</strong><br />
Bacterial Motility<br />
R. Nixon-Luke 1 , G. Bryant1 , C. Carnovale1 , V.<br />
A. Martinez 2 , W.C.K Poon2 1 School <strong>of</strong> Applied Sciences, RMIT <strong>University</strong>, GPO Box 2476,<br />
Melbourne, Victoria, 3000<br />
2 SUPA and COSMIC, School <strong>of</strong> Physics & Astronomy, The<br />
<strong>University</strong> <strong>of</strong> Edinburgh, Mayfield Road, Edinburgh EH9 3JZ,<br />
United Kingdom<br />
Biographies and Abstracts<br />
Tuesday 6 December - Session 5<br />
Biography<br />
Reece complete a Bachelor <strong>of</strong> Applied Science<br />
(Honours) at RMIT <strong>University</strong> and has commenced<br />
his PhD. His research interests include<br />
applications <strong>of</strong> light scattering techniques and<br />
development <strong>of</strong> novel instrumentation<br />
Abstract<br />
Cell motility is an important characteristic <strong>of</strong> many<br />
biological processes, and is ubiquitous in<br />
unicellular organisms such as bacteria. The<br />
bacterium Escherichia coli is a well studied model<br />
system for understanding cell motility. These cells<br />
execute random walks by alternating between<br />
swimming (or ‘running’) at speeds <strong>of</strong> tens <strong>of</strong><br />
microns per second (for ~ 1 second), and then<br />
tumbling (changing direction) for 0.1 s.<br />
Dynamic Light Scattering (DLS) has long been<br />
used for the measurement <strong>of</strong> Brownian motion in<br />
colloids, proteins and macromolecules, and is a<br />
routine technique for the determination <strong>of</strong> particle<br />
size in such Brownian systems. Dynamic light<br />
scattering can make accurate measurements over<br />
a very short timescale, is non-invasive, requires<br />
very little sample, and has a high sensitivity,<br />
making it a perfect tool to investigate living<br />
biological cells.<br />
Early in the development <strong>of</strong> DLS, its potential for<br />
studying bacterial motility was investigated [1];<br />
however, these investigations were complicated by<br />
a lack <strong>of</strong> understanding <strong>of</strong> the details <strong>of</strong> cell<br />
motility, as well as equipment limitations, and were<br />
never seriously pursued. Recently, a new<br />
microscope technique has been developed based<br />
on the principles <strong>of</strong> DLS. This technique, called<br />
differential dynamic microscopy (DDM), has been<br />
applied to the study <strong>of</strong> colloidal dispersions [2],<br />
and found to be a suitable tool in the investigation<br />
<strong>of</strong> bacterial motility [3].<br />
We report on preliminary investigations <strong>of</strong> the<br />
motility <strong>of</strong> E. Coli, and other motile bacteria, using<br />
DDM and DLS. We observe that the correlation<br />
functions exhibit two distinct decays: the faster<br />
decay being due to the self-propelled velocity<br />
(motility), and the second, slower decay being due<br />
to the normal diffusive motion.<br />
These functions were analysed using the model<br />
proposed by Nossal, Chen and Lai [1] and revised<br />
by Stock [4], which was found to provide good<br />
agreement under most conditions. This analysis<br />
yields the velocity distributions, the average<br />
velocities and the fraction <strong>of</strong> non-motile cells. The<br />
diffusion coefficients <strong>of</strong> the cells can be either an<br />
input into the model, or a free parameter. We will<br />
discuss the application <strong>of</strong> the techniques to other<br />
motile systems.<br />
75
76<br />
Biographies and Abstracts<br />
Tuesday 6 December - Session 5<br />
5.2.4 11:15am – 11:30am<br />
Revisiting Boltzmann: disentangling<br />
solid-state NMR measurements <strong>of</strong><br />
heterogeneous model membrane<br />
systems<br />
John D. Gehman1 , Marc-Antoine Sani1 ,<br />
Frances Separovic1 , Anil K. Mehta2 1 School <strong>of</strong> Chemistry, Bio21 Institute, <strong>University</strong> <strong>of</strong> Melbourne,<br />
VIC 3010 Australia; jgehman@unimelb.edu.au<br />
2 Chemistry Department, Emory <strong>University</strong>, Atlanta, GA 30322<br />
USA<br />
Biography<br />
After getting his undergraduate degree at Temple<br />
<strong>University</strong> in Philadelphia, John worked for five<br />
years at Merck in the Research Laboratories as a<br />
biochemist in the Department <strong>of</strong> Antiviral<br />
Enzymology. He moved next to Yale for his PhD,<br />
where he shifted to more physical disciplines,<br />
working and studying between the Biophysics<br />
(MB&B) and (Physical) Chemistry departments.<br />
After a short post-doc, also at Yale, John moved<br />
again a bit further to Australia, where he is now an<br />
Australian Research Council Future Fellow in<br />
Chemistry at Melbourne <strong>University</strong>. He has two<br />
beautiful daughters who stay up way too late,<br />
especially when he’s in charge.<br />
Abstract<br />
On the one hand, solid-state NMR is ideal for<br />
studying disordered materials that are difficult or<br />
impossible to study by other means; on the other<br />
hand, analysis <strong>of</strong> the resulting data is <strong>of</strong>ten<br />
challenging, and requires iteration with extensive<br />
modelling. We will discuss two examples:<br />
heterogeneous distributions <strong>of</strong> chemical shift<br />
tensor parameters, and distributions <strong>of</strong><br />
internuclear distances in rotational-echo doubleresonance<br />
(REDOR) data. A method that uses an<br />
adaptation <strong>of</strong> Boltzmann statistics maximum<br />
entropy will be described, which provides for a<br />
model-free approach to analysis <strong>of</strong> this type <strong>of</strong><br />
troublesome data.<br />
In the case <strong>of</strong> REDOR data, the method can reveal<br />
multiple distances with relatively few data points,<br />
which is <strong>of</strong> particular benefit in application to<br />
biological systems where relaxation limits signal<br />
lifetimes. This reverses the practice <strong>of</strong> comparing<br />
REDOR data to dephasing curves simulated for<br />
several preconceived structural models, by<br />
providing the information necessary to construct<br />
models based on unbiased data analysis. In the<br />
case <strong>of</strong> chemical shift tensor analysis, we apply<br />
the method to arbitrarily complex phospholipid<br />
mixtures as model membranes systems for<br />
analysis <strong>of</strong> subtle perturbations, for example by<br />
association with antimicrobial peptides.<br />
The Boltzmann Statistics method also <strong>of</strong>fers<br />
intriguing philosophical implications, as it gives the<br />
broadest, and perhaps most “honest” probability<br />
distribution consistent with the data, but also helps<br />
to determine which data points hold the greatest<br />
information content, such that experimental time<br />
can be focussed on gaining the best signal-tonoise<br />
where it is likely to benefit most.<br />
5.2.5 11:30am – 11:45am<br />
Liposomes: Stable or Kinetically<br />
Trapped?<br />
Adam Mechler1 and Mubin S. Huda2 1 Department <strong>of</strong> Chemistry, La Trobe <strong>University</strong>, Bundoora<br />
Campus, Bundoora, VIC 3086, a.mechler@latrobe.edu.au<br />
2 Department <strong>of</strong> Chemistry, La Trobe <strong>University</strong>, Bundoora<br />
Campus, Bundoora, VIC 3086, m.mubin@students.latrobe.edu.<br />
au<br />
Biography<br />
Dr. Mechler has obtained his PhD at the <strong>University</strong><br />
<strong>of</strong> Szeged, Hungary, in physics/materials science.<br />
His interest in biological nanostructures lead to 3<br />
years <strong>of</strong> postdoctoral work at the <strong>University</strong> <strong>of</strong><br />
Califonia, Santa Barbara on the atomic force<br />
microscopy <strong>of</strong> biomolecules, and then a research<br />
fellowship at Monash <strong>University</strong> with a sharpening<br />
focus on lipid membranes. He joined La Trobe<br />
<strong>University</strong> as a senior lecturer in 2009.<br />
Abstract<br />
Liposomes: spherical vesicles <strong>of</strong> phospholipid<br />
bilayers are among the most studied selfassembled<br />
structures, largely due to their<br />
relevance to biology through mimicking cell<br />
membranes, and the potential applications in<br />
biotechnology to support enzymatic processes in<br />
vitro. From the physical chemistry point <strong>of</strong> view,
Biographies and Abstracts<br />
Tuesday 6 December - Session 5<br />
phospholipid self-assembly might be seen as a<br />
spontaneous process, where the size and shape<br />
<strong>of</strong> the liposomes is the result <strong>of</strong> a free energy<br />
minimizing process. In this view the energy<br />
increase inherent in the curvature tension is<br />
compensated for in entropic terms, such as the<br />
entropic advantage <strong>of</strong> a highly dispersed system<br />
over the well ordered lamellar phase. Thus the<br />
liposome suspension is in equilibrium; the<br />
liposomes are thermodynamically stable. However,<br />
the potential <strong>of</strong> liposomes for reaching an<br />
equilibrium is frequently questioned. Calculations<br />
<strong>of</strong> lipid residence time in the aggregate based on<br />
critical micelle concentration predict that<br />
liposomes would not change on an observable<br />
timescale; they are kinetically trapped.<br />
Controversially, in the literature several articles<br />
empirically support the thermodynamic equilibrium<br />
model.<br />
Here we present the evidence <strong>of</strong> size evolution <strong>of</strong><br />
liposome suspensions from dynamical light<br />
scattering measurements for three different<br />
compositions: neat dimyristoyl phosphatidylcholine<br />
(DMPC), DMPC:cholesterol 9:1 and<br />
DMPC:DM phosphatidyl-glycerol 4:1. In all cases,<br />
an initial broad and highly polydisperse population<br />
evolved into a well defined, stable size distribution,<br />
which was bimodal for DMPC and<br />
DMPC:cholesterol 9:1. We used fluorescent<br />
imaging to confirm that the reduction in<br />
polydispersity was not caused by sedimentation.<br />
We demonstrate that polydispersity can be<br />
caused by the aggregation <strong>of</strong> small liposomes,<br />
and that temperature alone is a sufficient, and<br />
reversible, control <strong>of</strong> this aggregation. It is thus<br />
unclear if the observed reduction in polydispersity<br />
over time is the result <strong>of</strong> the slow separation <strong>of</strong><br />
liposomes from aggregates or an actual evolution<br />
<strong>of</strong> the sizes <strong>of</strong> individual liposomes (or a mix <strong>of</strong> the<br />
two); the final narrow size distributions however<br />
confirm that in either case liposomes appear to<br />
form with a size preference. These results suggest<br />
that the balance between the adhesive van der<br />
Waals and repulsive steric (protrusion, undulation,<br />
peristaltic etc) forces has a strong effect on the<br />
population homogeneity <strong>of</strong> liposome suspensions.<br />
5.2.6 11:45am – 12:00pm<br />
The Effect <strong>of</strong> Crystallization on<br />
Protein Quaternary Structure<br />
Donald G Vanselow 1<br />
1 nativeproteins.blogspot.com 54 Greenways Rd., Glen Waverley<br />
VIC 3150, Australia. dvanselow@hotmail.com<br />
Biography<br />
Dr Vanselow is a Thermodynamicist and<br />
Biochemist interested in the interaction <strong>of</strong> these<br />
fields throughout his career. He was a Lecturer at<br />
the <strong>University</strong> <strong>of</strong> Melbourne from 1975 to 1978 and<br />
Research Scientist in CSIRO from 1979 to 1983.<br />
He then worked in private industry before joining<br />
Monash <strong>University</strong> as a Scientific Manager in<br />
Biochemistry in 1989. His interest in protein<br />
structure and function began there. He left<br />
Monash in 1998 and has continued his research<br />
while engaged in school-teaching.<br />
Abstract<br />
Protein crystallization involves the conversion <strong>of</strong> a<br />
liquid protein-in-water phase into a crystalline<br />
water-in-protein phase. NMR studies support the<br />
view that the tertiary structures <strong>of</strong> proteins are<br />
usually very similar in both phases, but because <strong>of</strong><br />
the size limitation <strong>of</strong> NMR it has been impossible to<br />
accurately measure differences, if any, in the<br />
quaternary structures (arrangements <strong>of</strong> subunits).<br />
For want <strong>of</strong> any counter-proposal in the early days<br />
<strong>of</strong> x-ray crystallography, it is now widely assumed<br />
that crystallization has little or no effect on<br />
quaternary structure.<br />
This presentation will show how the various<br />
measurements <strong>of</strong> the surface energy <strong>of</strong> aqueous<br />
interfaces can be combined to reveal the chemical<br />
potential <strong>of</strong> water as a function <strong>of</strong> distance from an<br />
interface. This will be a refinement <strong>of</strong> an earlier<br />
model [1]. The extra chemical potential <strong>of</strong> water<br />
near an interface is responsible for the<br />
hydrophobic effect, which, in turn, is responsible<br />
for the binding <strong>of</strong> proteins to each other. The other<br />
significant effect at protein-protein interfaces is<br />
electrostatic. Detailed modelling [2] has shown that<br />
this is usually repulsive relative to opening the<br />
77
78<br />
Biographies and Abstracts<br />
Tuesday 6 December - Session 5<br />
interface to water because <strong>of</strong> the affinity <strong>of</strong> water<br />
for charges (solvation energy).<br />
For a model protein it will be shown that the<br />
crowding accompanying crystallization weakens<br />
the hydrophobic effect. Repulsion due to solvation<br />
energy then opens the original interfaces leading<br />
to further crowding elsewhere because the system<br />
cannot expand. Ultimately the parts <strong>of</strong> the protein<br />
that can come into contact are those with minimal<br />
solvation energy - the so-called hydrophobic<br />
patches. This is what is observed in crystals.<br />
An animation <strong>of</strong> the protein dihydrodipicolinate<br />
synthase (DHDPS) transforming from its native<br />
ovoid shape [3] into the torus seen in crystals will<br />
be shown.<br />
Mention will be made <strong>of</strong> the advances in<br />
understanding the mechanisms <strong>of</strong> protein function<br />
that result from reassembly <strong>of</strong> crystal structures<br />
into physically and biologically realistic forms.<br />
Refs:<br />
[1] Vanselow, D. G. 2002. Role <strong>of</strong> constraint in catalysis and<br />
high-affinity binding by proteins, Biophys. J. 82: 2293–2303.<br />
[2] Hendsch, Z. S., B. Tidor. 1994. Do salt bridges stabilize<br />
proteins? A continuum electrostatic analysis. Protein Sci. 3:<br />
211-226.<br />
[3] The data-base <strong>of</strong> native protein structures is at www.<br />
nativeproteins.net76.net/gallery/index.htm<br />
Papers describing the structures and discussing related issues<br />
are at http://nativeproteins.blogspot.com<br />
Both sites are maintained by the author.<br />
Notes<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................
Session 6 – Theatre 2<br />
6.1.1 1:15pm – 1:45pm<br />
Anion Photoelectron Spectroscopy<br />
<strong>of</strong> Ionic Complexes and Clusters<br />
Duncan A. Wild, Kim M. Lapere, Stephen G.<br />
Dale, Allan J. McKinley<br />
Chemistry, School <strong>of</strong> Biomedical, Biomolecular, and Chemical<br />
Sciences, The <strong>University</strong> <strong>of</strong> Western Australia,<br />
M313 35 Stirling Hwy, Crawley, WA, 6009,<br />
duncan.wild@uwa.edu.au<br />
Biographies and Abstracts<br />
Tuesday 6 December - Session 6<br />
Biography<br />
Dr Duncan Wild completed a Bachelor <strong>of</strong> Science<br />
with Honours at the <strong>University</strong> <strong>of</strong> Melbourne,<br />
where he also undertook a PhD researching<br />
infrared spectroscopy <strong>of</strong> size-selected anion<br />
complexes and clusters. In 2003 he received an<br />
Alexander Von Humbolt Fellowship to undertake<br />
research at the Max Planck Institute for<br />
Biophysical Chemistry in Göttingen, Germany.<br />
During this time he investigated the timescales for<br />
energy relaxation <strong>of</strong> carotenoids, spectroscopy <strong>of</strong><br />
neutral stilbene-alkane gas phase complexes, and<br />
assisted in the construction <strong>of</strong> a photoelectron<br />
spectrometer. Dr Wild came to The <strong>University</strong> <strong>of</strong><br />
Western Australia in 2007 where he has<br />
constructed a Time Of Flight Mass Spectrometer<br />
coupled to a photoelectron Spectrometer in order<br />
to study anion-molecule complexes <strong>of</strong><br />
atmospheric and astronomic interest.<br />
Abstract<br />
To understand the reactions that occur between<br />
radicals and molecules one ultimately requires an<br />
intimate knowledge <strong>of</strong> the potential energy surface<br />
governing the reaction. Such surfaces are routinely<br />
produced from ab initio calculations however their<br />
accuracy requires critical assessment. This can be<br />
achieved by applying spectroscopy to radicalmolecule<br />
complexes which are features <strong>of</strong> the<br />
reactive potential energy surface.<br />
At the <strong>University</strong> <strong>of</strong> Western Australia we have<br />
constructed an apparatus which allows us to<br />
perform photoelectron spectroscopy <strong>of</strong> mass<br />
selected anion-molecule complexes and clusters.<br />
The TOF-PES consists <strong>of</strong> a time <strong>of</strong> flight (TOF)<br />
mass spectrometer for selection <strong>of</strong> a specific<br />
anion-molecule complex, and a photoelectron<br />
spectrometer (PES) to monitor the photoelectrons<br />
detached by UV radiation. As mass spectrometry<br />
is applied it is possible to investigate larger clusters<br />
<strong>of</strong> the form X-…Mn in a step wise fashion, thereby<br />
bridging the gap between gas and bulk phase<br />
contexts.<br />
We complement our experiments with high level<br />
ab initio calculations (up to CCSD(T)/aug-cc-pvtz).<br />
We predict structures and energetics for the<br />
anion-molecule and radical-molecule van der<br />
Waals complexes, to aid in interpretation <strong>of</strong> the<br />
spectra. We also produce multi-dimensional<br />
potential energy surfaces. This contribution will<br />
highlight our recent work we have undertaken on<br />
the halogen anion-molecule complexes and<br />
clusters.<br />
6.1.2 1:45pm – 2:00pm<br />
Ion Mobility Mass Spectrometry<br />
Reveals New Insights into Structure<br />
and Assembly <strong>of</strong> Protein Complexes<br />
Tara Pukala1 , Antonio Calabrese1 , Danielle<br />
Williams1 , Yanqin Liu1 1 School <strong>of</strong> Chemistry and Physics, <strong>University</strong> <strong>of</strong> Adelaide, North<br />
Terrace, Adelaide, SA, 5005, tara.pukala@adelaide.edu.au<br />
Biography<br />
Dr Tara Pukala obtained a PhD from the <strong>University</strong><br />
<strong>of</strong> Adelaide (with J.H. Bowie) which was followed<br />
by a postdoctoral role at the <strong>University</strong> <strong>of</strong><br />
Cambridge, UK (with C.V. Robinson). She returned<br />
to a lectureship position at the <strong>University</strong> <strong>of</strong><br />
Adelaide in 2008, where she has established a<br />
research group focussed on the development and<br />
application <strong>of</strong> mass spectrometric and related<br />
technologies for the investigation <strong>of</strong> biomolecular<br />
systems.<br />
Abstract<br />
Protein-protein interactions play important roles in<br />
controlling cellular processes, and <strong>of</strong>fer new<br />
targets for therapeutic intervention. However, the<br />
structural variability and transient nature <strong>of</strong><br />
biological complexes means their architecture and<br />
interactions can be difficult to characterise using<br />
traditional methods. Mass spectrometry has<br />
emerged as a powerful tool for the analysis <strong>of</strong> such<br />
79
80<br />
Biographies and Abstracts<br />
Tuesday 6 December - Session 6<br />
systems, due to its ability to investigate protein<br />
assemblies at all levels <strong>of</strong> structural complexity. In<br />
particular, ion mobility-mass spectrometry (IM-MS)<br />
provides not only mass but shape information <strong>of</strong><br />
native protein assemblies, making it possible to<br />
define protein complex identity, stoichiometry, size,<br />
structural arrangement and subunit interactions in<br />
a single experiment.<br />
Here we describe the design and synthesis <strong>of</strong><br />
novel mass spectrometry amenable cross-linking<br />
reagents, which can be used to stabilise proteinprotein<br />
interactions by forming covalent bonds,<br />
and if cross-linked sites are identified, can yield<br />
clues about protein binding interfaces. In tandem<br />
with an IM-MS based approach, we show that it is<br />
possible to describe low-resolution models <strong>of</strong><br />
protein complexes <strong>of</strong> previously unknown<br />
structure that are not amenable to traditional<br />
structure biology approaches. We will discuss<br />
current results from various systems under study<br />
in our research group, including calciumcalmodulin-peptide<br />
regulatory complexes, the 10<br />
protein DNA transcription factor assembly TFIIH<br />
and aggregation products <strong>of</strong> alpha synuclein<br />
associated with Parkinson’s Disease.<br />
6.1.3 2:00pm – 2:15pm<br />
Nanomechanics <strong>of</strong> live bacterial cells<br />
Michelle L Gee1 1 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Melbourne, Parkville 3010<br />
Australia, mlgee@unimelb.edu.au<br />
Biography<br />
Michelle Gee is a physical chemist with particular<br />
interests in intermolecular and surface nano-scale<br />
forces in cellular and biomimetic systems, and<br />
nanostructures and molecular assemblies. She<br />
received her PhD from the <strong>University</strong> <strong>of</strong> Melbourne<br />
and subsequently held research fellowships at the<br />
<strong>University</strong> <strong>of</strong> California Santa Barbara and<br />
Princeton <strong>University</strong>. Michelle has held external<br />
appointments that include visiting chairs in the<br />
Departments <strong>of</strong> Physics and Chemical Engineering<br />
at Carnegie Mellon <strong>University</strong> and at CNRS. She<br />
currently serves on the editorial board <strong>of</strong> S<strong>of</strong>t<br />
Materials and is an elected member <strong>of</strong> council to<br />
the International Association <strong>of</strong> Colloid and<br />
Interface Scientists. Michelle leads the S<strong>of</strong>t<br />
Condensed matter Labs at the <strong>University</strong> <strong>of</strong><br />
Melbourne with expertise in lipid-peptide<br />
interactions and the action antimicrobial peptides,<br />
the biophysical properties <strong>of</strong> microbes, particularly<br />
the bacterial glycocalyx and cell wall, and<br />
self-assembled nanostructures for targeted drug<br />
delivery, separations and high-density storage.<br />
Recently her group has developed<br />
multidimensional fluorescence spectroscopies and<br />
direct nanomechanical force methodologies for<br />
studying live cells.<br />
Abstract<br />
With the increased prevalence <strong>of</strong> superbugs,<br />
pathogens resistant to antibiotics, research is<br />
focussing on novel ways <strong>of</strong> interrogating the<br />
behaviour <strong>of</strong> bacteria and the mechanisms by<br />
which they protect themselves. It is thought that<br />
bacterial surface polymers (fimbriae,<br />
lipopolysaccharide and capsular glycocalyx) are<br />
key components <strong>of</strong> cell survival. However, the<br />
roles <strong>of</strong> these surface polymers and their<br />
inter-relationships are not understood.<br />
Here, we show how Atomic Force Microscopy<br />
(AFM) can be used to measure the<br />
nanomechanical properties <strong>of</strong> the surface <strong>of</strong><br />
individual, live bacterial cells, in situ, to give insights<br />
into the biophysical behaviour <strong>of</strong> bacterial surface<br />
polymers. Our results focus on Klebsiella<br />
pneumoniae, an opportunistic pathogen that<br />
causes diseases such as pneumonia, urinary tract<br />
infections, surgical wound infections and infection<br />
<strong>of</strong> the blood, known as bacteremia. We show for<br />
the first time that AFM nanomechanical data for<br />
wild type Klebsiella pneumoniae and its genetically<br />
manipulated mutants give insights into the<br />
biophysical structure and role <strong>of</strong> the cell capsule<br />
and fimbriae in cell survival and cell adhesion.
6.1.4 2:15pm – 2:30pm<br />
Gas-phase structures <strong>of</strong> two isomers<br />
<strong>of</strong> deoxyguanosine radical cation:<br />
experiments and theory<br />
Linda Feketeová 1 , Bun Chan2 , George N.<br />
Khairallah1 , Vincent Steinmetz 3 , Elizabeth Yuriev4 ,<br />
Philippe Maitre3 , John D. Orbell5 , Leo Radom2 ,<br />
Richard A.J. O’Hair1 1 ARC Centre <strong>of</strong> Excellence for Free Radical Chemistry and<br />
Biotechnology, School <strong>of</strong> Chemistry and Bio21 Institute <strong>of</strong><br />
Molecular Science and Biotechnology, The <strong>University</strong> <strong>of</strong><br />
Melbourne, 30 Flemington Road, Parkville, VIC, 3010, Australia,<br />
lfe@unimelb.edu.au<br />
2 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney, NSW, 2006,<br />
Australia, chan_b@chem.usyd.edu.au<br />
3 Laboratoire de Chimie Physique, Université Paris Sud, 15,<br />
avenue Jean PERRIN, Orsay Cedex, 91405, France, philippe.<br />
maitre@u-psud.fr<br />
4 Medicinal Chemistry and Drug Action, Monash Institute <strong>of</strong><br />
Pharmaceutical Sciences, Monash <strong>University</strong>, Parkville, VIC,<br />
3052, Australia. Elizabeth.Yuriev@vcp.monash.edu.au<br />
5 School <strong>of</strong> Engineering & Science, Victoria <strong>University</strong>, Werribee,<br />
VIC, 3030, Australia, John.Orbell@vu.edu.au<br />
Biography<br />
Linda studied physics at the Comenius <strong>University</strong><br />
in Bratislava, Slovakia, where she specialized in<br />
plasma physics. As an undergraduate, she visited<br />
Leopold-Franzens <strong>University</strong> in Innsbruck, Austria,<br />
several times, to study electron interactions with<br />
nucleobases. Linda received her PhD in physics<br />
from Leopold-Franzens <strong>University</strong> in 2006 with<br />
focus on ion-surface interactions <strong>of</strong> charged<br />
molecular ions. Afterwards she got awarded a<br />
visiting fellowship to work at Queen�™s<br />
<strong>University</strong> Belfast to build a new experimental<br />
setup to study ion-molecule reactions in the gas<br />
phase with well defined ion kinetic energies.<br />
Afterwards she obtained European Science<br />
Foundation fellowship, to work at Aarhus<br />
<strong>University</strong>, Denmark, on electron scattering<br />
experiments. After arriving in Australia, she joined<br />
synchrotron group at Monash <strong>University</strong> before<br />
she got awarded an ARC International Fellowship<br />
with Pr<strong>of</strong>. O�™Hair from The <strong>University</strong> <strong>of</strong><br />
Melbourne in 2008. In 2010 she was awarded an<br />
ARC Postdoctoral Fellowship to study<br />
biomolecules and biomolecular clusters.<br />
Biographies and Abstracts<br />
Tuesday 6 December - Session 6<br />
Abstract<br />
Electrospray ionisation (ESI) <strong>of</strong> methanolic<br />
solutions containing a mixture <strong>of</strong> the nucleoside<br />
deoxyguanosine, dG, incubated with Cu(NO 3 ) 2<br />
resulted in the formation <strong>of</strong> a range <strong>of</strong> ions,<br />
including doubly charged copper nucleoside<br />
complexes [CuIIdGn]2+, with n ranging from 2 to<br />
10. 1 Collision-induced dissociation (CID) <strong>of</strong> these<br />
complexes proceeds via a number <strong>of</strong> different<br />
pathways that depend on the size <strong>of</strong> the cluster, n.<br />
When n = 3, monomeric radical cations are formed<br />
via redox processes. When n = 4, dimeric radical<br />
cations are formed. A key finding is that the radical<br />
cations <strong>of</strong> dG have fragmentation patterns that<br />
depend on the way they are formed. Thus, dG •+<br />
formed directly in the ESI source or via CID <strong>of</strong><br />
[Cu II dG3] 2+ complex fragment in the same way,<br />
giving the radical cation <strong>of</strong> the guanine base at m/z<br />
151 via cleavage <strong>of</strong> the N-glycosidic bond. In<br />
contrast, the CID spectra <strong>of</strong> radical cation formed<br />
via the sequences [CuIIdG4] 2+ •+ •+ → dG → dG is<br />
2<br />
dominated by the loss <strong>of</strong> CH 2 O and further loss <strong>of</strong><br />
C 2 H 3 O 2 from the sugar moiety. In addition, infrared<br />
(IR) fingerprint spectra <strong>of</strong> the two type <strong>of</strong> dG •+ ,<br />
recorded using a Free Electron Laser coupled with<br />
a quadrupole ion-trap, confirm that two different<br />
isomers are obtained depending on the way they<br />
are formed. These different fragmentation<br />
reactions are attributed to different tautomeric<br />
structures <strong>of</strong> the radical cations. MS/MS, quantum<br />
chemical calculations, gas-phase IR spectroscopy,<br />
and ion-molecule reactions were used to<br />
determine the possible structures <strong>of</strong> these<br />
tautomeric radical cations.<br />
[1] Feketeová, L.; Yuriev, E.; Orbell, J. D.; Khairallah, G. N.; O’Hair,<br />
R. A. J. Int. J. Mass Spectrom. 2010, 304, 74-82<br />
81
82<br />
Biographies and Abstracts<br />
Tuesday 6 December - Session 6<br />
6.1.5 2:30pm – 2:45pm<br />
Attaching Molecular Hydrogen to<br />
Atomic Ions<br />
Evan Bieske 2 , Viktoras Dryza1 ,<br />
1 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Melbourne, Parkville 3010,<br />
vdryza@unimelb.edu.au<br />
2 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Melbourne, Parkville 3010,<br />
evanjb@unimelb.edu.au<br />
Biography<br />
Evan Bieske has been at the School <strong>of</strong> Chemistry,<br />
<strong>University</strong> <strong>of</strong> Melbourne for 15 years. He is<br />
primarily concerned with the spectroscopy <strong>of</strong><br />
molecular ions and ion clusters.<br />
Abstract<br />
This talk will focus on our laser spectroscopic<br />
studies <strong>of</strong> small molecular complexes, in which<br />
one or more hydrogen molecules is attached to an<br />
atomic cation (Li+-H2, Na+-H2, Cr+-H2, B+-H2<br />
and Ag+-H2) or anion (Cl--H2, Br--H2, I--H2).<br />
These simple complexes serve as model systems<br />
for understanding the bonding <strong>of</strong> dihydrogen to<br />
charged sites in solid materials including zeolites<br />
and metal organic frameworks which are currently<br />
being investigated for hydrogen storage.3 The<br />
infrared spectra, which feature full resolution <strong>of</strong><br />
rotational structure, are recorded by monitoring<br />
charged phot<strong>of</strong>ragments, and deliver detailed<br />
information on the manner in which H2 molecules<br />
interact with metal cations and halide anions. In<br />
some cases, the onset <strong>of</strong> dissociation at a<br />
particular rovibrational level allows us to place<br />
extremely narrow bounds on the dissociation<br />
energy <strong>of</strong> the complex, providing a benchmark<br />
against which current computational techniques<br />
can be assessed. We will discuss the manner in<br />
which the properties <strong>of</strong> the complexes, including<br />
dissociation energy, vibrational band shift, and<br />
intermolecular separation, depend on the<br />
electronic structure <strong>of</strong> the atomic ion.<br />
FIG. 1: Binding energies for M+-H2 complexes deduced through<br />
thermochemical measurements. Infrared spectra have been<br />
obtained for complexes with hatched bars, allowing extraction <strong>of</strong><br />
detailed structural information. The vibrational frequencies <strong>of</strong> H2<br />
and D2 are marked on the right, indicating which complexes can<br />
be studied using resonance enhanced photodissociation<br />
spectroscopy.<br />
6.1.6 2:45pm – 3:00pm<br />
Which Density Functionals can be<br />
reliably applied to Main Group<br />
Thermochemistry?<br />
Lars Goerigk1,2 , Stefan Grimme2 1 School <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> Sydney, Sydney, New<br />
South Wales 2006, Australia<br />
2 Theoretical Organic Chemistry, Organisch-Chemisches Institut,<br />
<strong>University</strong> <strong>of</strong> Münster, Corrensstr. 40, 48149 Münster, Germany<br />
Biography<br />
2003-2008: Studies in Chemistry, <strong>University</strong> <strong>of</strong><br />
Münster, Germany<br />
2008-2011: PhD Thesis with Pr<strong>of</strong>. Stefan Grimme,<br />
<strong>University</strong> <strong>of</strong> Münster, Germany<br />
Abstract<br />
Which functional should I use? Many users <strong>of</strong> DFT<br />
are regularly faced with this problem. We present<br />
the results <strong>of</strong> a recent study, in which we tried to<br />
answer this question and tried to shed some light<br />
on the plethora <strong>of</strong> developed DFT methods.[1] Our<br />
aim was to thoroughly investigate which<br />
functionals are generally well applicable and<br />
robust to describe the energetics <strong>of</strong> molecules.<br />
Our study was based on the new GMTKN30<br />
database for general main group thermochemistry,<br />
kinetics and noncovalent interactions.[2,3] In total,<br />
47 functionals were investigated: two LDAs, 14<br />
GGAs, three meta-GGAs, 23 hybrids and five
double-hybrids. Besides the double-hybrids, also<br />
other modern approaches, i.e., the M05 and M06<br />
classes <strong>of</strong> functionals and range-separated<br />
hybrids, were tested. The range <strong>of</strong> the properties<br />
covered by GMTKN30 allowed us to give a reliable<br />
answer to the question stated above.<br />
Biographies and Abstracts<br />
Tuesday 6 December - Session 6<br />
Perdew’s metaphoric picture <strong>of</strong> Jacob’s Ladder for<br />
the classification <strong>of</strong> density functionals’<br />
performance could unbiasedly be confirmed with<br />
GMTKN30. We also showed that there is no<br />
statistical correlation between a functional’s<br />
accuracy for atomization energies and the<br />
performance for chemically more relevant reaction<br />
energies.<br />
For each rung on Jacob’s Ladder we recommend the functionals,<br />
which we regard as the most robust ones in terms <strong>of</strong> accuracy,<br />
reliability and robustness. The recommended methods are the<br />
B97-D3 and revPBE GGAs, the oTPSS[2] meta-GAA, Truhlar’s<br />
PW6B95 hybrid and the PWPB95[3] and DSD-BLYP doublehybrids.<br />
The usage <strong>of</strong> an atom-pairwise London-dispersion<br />
correction (DFT-D3) is shown to be crucial for accuracy.[4] We<br />
discourage from regularly applying the B3LYP method, because<br />
better alternatives are available. We hope that our<br />
recommendations are helpful to the broad community <strong>of</strong> DFT<br />
users.<br />
[1] L. Goerigk, S. Grimme, Phys. Chem. Chem. Phys., 2011, 13,<br />
6670.<br />
[2] L. Goerigk, S. Grimme, J. Chem. Theory Comput. 2010, 6, 107.<br />
[3] L. Goerigk, S. Grimme, J. Chem. Theory Comput. 2011, 7, 291.<br />
[4] S. Grimme, J. Antony, S. Ehrlich, H. Krieg J. Chem. Phys. 2010,<br />
132, 154104.<br />
Session 6 - Main Theatre<br />
6.2.1 1:15pm – 1:30pm<br />
Polarization effects in ion channels<br />
and bulk from ab initio MD<br />
simulations<br />
Serdar Kuyucak, Jeff Timko, Alexandra De<br />
Castro,<br />
School <strong>of</strong> Physics, <strong>University</strong> <strong>of</strong> Sydney, NSW 2006<br />
Biography<br />
2004-present: A/Pr<strong>of</strong>. in Biophysics, <strong>University</strong> <strong>of</strong><br />
Sydney<br />
1992-2003: Senior Fellow in Nuclear Physics and<br />
Biophysics, ANU<br />
1985-1991: Research Fellow in Nuclear Physics,<br />
<strong>University</strong> <strong>of</strong> Melbourne<br />
1983-1985: PDF in Nuclear Physics, <strong>University</strong> <strong>of</strong><br />
Tubingen<br />
1982: PhD in Nuclear Physics from Yale <strong>University</strong><br />
Abstract<br />
Classical force fields employed in molecular<br />
dynamics (MD) simulations <strong>of</strong> biomolecules do not<br />
include the polarization interaction explicitly.<br />
Effects <strong>of</strong> polarization are taken into account in an<br />
average way by boosting the partial charges on<br />
atoms. Because this parametrization is done in<br />
bulk solution, it may not work very well in a<br />
different environment, e.g. membrane proteins, or<br />
with higher charges such as Ca2+ ions. In<br />
particular, polarization is expected to play an<br />
important role in ion permeation across ion<br />
channels. Indeed several calculations <strong>of</strong> the<br />
potential <strong>of</strong> mean force (PMF) <strong>of</strong> K+ ions across<br />
the gramicidin A channel have found a large<br />
central energy barrier <strong>of</strong> height ~20 kT, which is<br />
incompatible with the observed conductance <strong>of</strong><br />
K+ ions near the diffusion rate. Inclusion <strong>of</strong> the<br />
polarization interaction has been shown to reduce<br />
this barrier significantly.<br />
Efforts are under way to construct polarizable<br />
force fields to deal with such problems. However,<br />
relying solely on experiments to constrain the<br />
parameters <strong>of</strong> polarizable force fields is fraught<br />
with difficulties. Ab initio calculations would be<br />
83
84<br />
Biographies and Abstracts<br />
Tuesday 6 December - Session 6<br />
very helpful in this regard by providing extensive<br />
set <strong>of</strong> results for fitting the parameters and further<br />
testing <strong>of</strong> the force fields. Towards this end, we<br />
have performed ab initio MD simulations <strong>of</strong> ion<br />
channels and bulk solution, and characterized<br />
their polarization properties.<br />
Salt solution is the main environment for<br />
biomolecules and its correct description is<br />
essential for their proper simulation. We have<br />
constructed the PMF for the dissociation <strong>of</strong> Na-Cl<br />
and Ca-Cl pairs in water from ab initio MD<br />
simulations [1, 2]. Comparison <strong>of</strong> these ab initio<br />
PMF’s to those obtained from the non-polarizable<br />
force fields illustrates the significance <strong>of</strong> the<br />
polarization. As one would expect the dipole<br />
moments <strong>of</strong> the hydration waters differ significantly<br />
between the ab ignition and classical results.<br />
Analysis <strong>of</strong> the trajectory data further reveals that<br />
the hydration numbers also differ and this has an<br />
impact on the PMF’s.<br />
Ion channels provide the most stringent tests for<br />
the classical force fields. We have studied the<br />
polarization effect in the gramicidin A and<br />
potassium channels via ab inito MD simulations.<br />
Our results indicate that presence <strong>of</strong> ions in the<br />
channel has a significant effect on the dipole<br />
moments carbonyl groups, boosting their dipole<br />
moments appreciably compared to simulations<br />
with water only [3]. This will help to lower the free<br />
energy <strong>of</strong> ions in the channel and solve the<br />
problems encountered in permeation <strong>of</strong> K+ ions<br />
across the gramicidin A and potassium channels.<br />
References<br />
1. J. Timko, D. Bucher and S. Kuyucak. J. Chem. Phys. 132 (2010)<br />
114510.<br />
2. J. Timko, A. De Castro and S. Kuyucak, J. Chem. Phys. 134<br />
(2011) 204510.<br />
3. D. Bucher and S. Kuyucak, Chem. Phys. Lett.477 (2009) 207.<br />
6.2.2 1:30pm – 1:45pm<br />
A critical dual role for the pore helix<br />
in hERG K channel inactivation<br />
Matthew D Perry1 , Jamie I Vandenberg 1,2 ,<br />
1 Molecular Cardiology and Biophysics Division, Victor Chang<br />
Cardiac Research Institute, 405 Liverpool Street, Darlinghurst,<br />
NSW 2010, Australia<br />
2 St Vincent’s Clinical School, <strong>University</strong> <strong>of</strong> New South Wales,<br />
NSW 2052, Australia<br />
Biography<br />
Matt Perry gained his PhD at the <strong>University</strong> <strong>of</strong><br />
Leeds in the UK, before undertaking post-doctoral<br />
positions with John Mitcheson at <strong>University</strong> <strong>of</strong><br />
Leicester and subsequently with Mike Sanguinetti<br />
at the <strong>University</strong> <strong>of</strong> Utah in Salt Lake City. He is<br />
currently pursuing his interests in ion gating and<br />
pharmacology at the Victor Chang Cardiac<br />
Research Institute in Sydney with Jamie<br />
Vandenberg.<br />
Abstract<br />
Human ether-a-go-go related gene (hERG)<br />
potassium channels pass current (IKr) that plays<br />
an important role in repolarization <strong>of</strong> the cardiac<br />
ventricular action potential. Underlying the unique<br />
hERG/IKr phenotype is a rapid, voltage-dependent<br />
C-type inactivation gating mechanism.<br />
Previously, we used ϕ-value analysis to determine<br />
the temporal sequence <strong>of</strong> conformational changes<br />
in the channel protein during the transition from<br />
open to inactivated states (Wang et al, 2011; Nat<br />
Struct & Mol Biol, 18: 35-42). After introducing a<br />
point mutation, a ϕ-value is calculated by<br />
comparing perturbations (versus WT) to the<br />
energetics <strong>of</strong> the transition state, ∆log(k ), to that <strong>of</strong><br />
f<br />
the stable end states, ∆log(K ). The ratio<br />
eq<br />
∆log(k )/∆log(K ) provides a ϕ-value between 0<br />
f eq<br />
and 1, where 0 indicates the last step in the<br />
pathway and 1 indicates a change at the start <strong>of</strong><br />
the reaction. Such analysis revealed an initial loss<br />
<strong>of</strong> potassium ions from the selectivity filter is<br />
followed in turn by conformational changes in the<br />
S5, S4, S4-S5 linker, S6 and pore helix. Finally, the<br />
channel is rendered non-conducting through a<br />
‘collapse’ <strong>of</strong> the selectivity filter. However, a key<br />
question still to be answered is how loss <strong>of</strong><br />
potassium from the selectivity filter leads to
conformational changes in the S5 helix. We<br />
hypothesized that the pore helix may play a dual<br />
role, acting to couple the selectivity filter to the S5<br />
helix (early) and to the S6 helix (late).<br />
A mutagenesis scan identified two residues near<br />
the base <strong>of</strong> the pore helix (Thr618 and Leu622)<br />
where mutants resulted in sufficiently large<br />
∆log(K ) values (>0.5) to permit accurate<br />
eq<br />
estimations <strong>of</strong> ϕ-values. Derived mean ϕ-values<br />
for T618V and T618I were 0.87±0.03 (n=8) and<br />
0.9±0.02 (n=7). The overall ϕ-value for Thr618<br />
mutants (C, V, I, L, M, Q) was 0.89 (R2 =0.99).<br />
Similarly, ϕ-value for L622V was 0.81±0.01 (n=7),<br />
with an overall ϕ-value for Leu622 mutants (C, V,<br />
M, I) <strong>of</strong> 0.85 (R2 =0.91). These results reveal that<br />
during transition from open to inactivated states,<br />
the base <strong>of</strong> the pore helix undergoes a<br />
conformational change just after the exit <strong>of</strong> K+ ions<br />
from the selectivity filter (ϕ ~1) and just prior to a<br />
conformational change in the S5 domain (ϕ ~0.75).<br />
Our study suggests that the pore helix plays a<br />
critical dual role in C-type inactivation gating <strong>of</strong><br />
hERG channels, undergoing conformational<br />
changes both early (coupling loss <strong>of</strong> K+ ions to S5<br />
motion) and late (coupling S6 motion to selectivity<br />
filter collapse) in the transition pathway.<br />
6.2.3 1.45pm – 2.00pm<br />
Bimodal Regulation <strong>of</strong> hERG Gating<br />
by the N-Terminal Tail as Revealed<br />
by Voltage Clamp Fluorometry<br />
<strong>Peter</strong> S.P. Tan1 , Adam P. Hill1 , Chai Ann Ng1 ,<br />
Matthew Perry1 , Jamie I. Vandenberg 1<br />
1 Molecular Cardiology and Biophysics Division, Victor Chang<br />
Cardiac Research Institute, Darlinghurst, New South Wales<br />
Biography<br />
<strong>Peter</strong> is a postdoctoral researcher at the Victor<br />
Chang Cardiac Research Institute working with Dr<br />
Adam Hill and Pr<strong>of</strong> Jamie Vandenberg<br />
Abstract<br />
The cytoplasmic N-terminal domain <strong>of</strong> humanether-a-go-go<br />
related gene (hERG) K+ channels<br />
has been shown to modulate the unusually slow<br />
deactivation <strong>of</strong> this channel. We have previously<br />
Biographies and Abstracts<br />
Tuesday 6 December - Session 6<br />
solved the solution NMR structure <strong>of</strong> this region<br />
showing it to contain a Per-Arnt-Sim (PAS) domain<br />
(residues 26-135), an amphipathic α-helix (residues<br />
13-23) and an unstructured tail segment (residues<br />
2-9). Deletion <strong>of</strong> the tail and α-helical regions<br />
(∆2-25) or the tail alone (∆2-9) both enhanced rates<br />
<strong>of</strong> deactivation and slowed the rate <strong>of</strong> activation.<br />
However, while ∆2-9 shifted the V0.5 <strong>of</strong> activation<br />
in the depolarized direction, ∆2-25 did not,<br />
suggesting a differential effect on the voltage<br />
dependence <strong>of</strong> activation.<br />
In this study we have used voltage clamp<br />
fluorometry (VCF) to investigate the mechanism <strong>of</strong><br />
how these N-terminal deletions affect the<br />
activation process. The fluorophore MTSR was<br />
attached to position E518C in the voltage sensing<br />
domain to track movement <strong>of</strong> this region during<br />
activation in each <strong>of</strong> the N-terminal deletions. The<br />
V0.5 <strong>of</strong> the fluorescent-voltage (FV) relationship for<br />
∆2-9 was significantly shifted to more depolarized<br />
potentials (18.2 ± 1.5 mV, n= 9) when compared to<br />
control (0 ± 1.8 mV, n=12, one way ANOVA)<br />
indicating that ∆2-9 affects activation gating by<br />
shifting the voltage range over which the voltage<br />
sensor moves. In contrast the V0.5 <strong>of</strong> ∆2-25 (1 ±<br />
2.9 mV, n=7) was not significantly different from<br />
control.<br />
In contrast, both ∆2-9 and ∆2-25 have slow rates<br />
<strong>of</strong> current activation (τ = 208 ± 9.6 ms and 169.2 ±<br />
13.5 ms at 20kjmol-1 respectively, one-way<br />
ANOVA) compared to control (τ = 90.5 ± 13.2 ms<br />
at 20kjmol-1) with no observable effect on the<br />
fluorescent report <strong>of</strong> the rate <strong>of</strong> voltage sensor<br />
movement (τ = 89.6 ± 9.8 ms, 85.1 ± 7.2 ms and<br />
81.8 ± 4.1 ms at 10kjmol-1for control, ∆2-9 and<br />
∆2-25 respectively, one-way ANOVA). This data<br />
shows that both ∆2-9 and ∆2-25 slowed rates <strong>of</strong><br />
activation independent <strong>of</strong> voltage sensor<br />
movement. A likely explanation for these results is<br />
that deletion <strong>of</strong> the N-terminal tail alters the rate <strong>of</strong><br />
opening/closing <strong>of</strong> the cytoplasmic activation gate<br />
in the pore domain.<br />
Our data suggests a bimodal regulation <strong>of</strong><br />
activation gating by the N-terminal domain <strong>of</strong><br />
hERG channels. While the α-helical region<br />
modulates activation gating via an effect on<br />
movement <strong>of</strong> the voltage sensor, the N-terminal tail<br />
(residues 1-9) interact with the cytoplasmic<br />
activation gate <strong>of</strong> the pore domain to regulate the<br />
rates <strong>of</strong> opening and closing.<br />
85
86<br />
Biographies and Abstracts<br />
Tuesday 6 December - Session 6<br />
6.2.4 2:00pm – 2:15pm<br />
Studies <strong>of</strong> Bacterial<br />
Mechanosensitive (MS) Channels<br />
under High Hydrostatic Pressure<br />
Evgeny Petrov1 , Paul R Rohde1 and Boris<br />
1, 2 Martinac<br />
1 Victor Chang Cardiac Research Institute, Lowy-Packer Building,<br />
405 Liverpool St, Darlinghurst, 2010.<br />
2 St Vincent’s Clinical School, The <strong>University</strong> <strong>of</strong> New South<br />
Wales, Victoria St, Darlinghurst, 2010 E.Petrov@victorchang.<br />
edu..au; P.Rohde@victorchang.edu.au; B.martinac@<br />
victorchang.edu.au<br />
Biography<br />
Senior Research Fellow, JAN2009 – Present<br />
The Victor Chang Cardiac Research Institute, 405<br />
Liverpool St, Darlinghurst, NSW 2010<br />
Effect <strong>of</strong> high hydrostatic pressure on<br />
mechanosensitive ion channels<br />
Research Officer, JAN2005-DEC2008<br />
School <strong>of</strong> Biomedical Sciences, Skerman Building<br />
65, <strong>University</strong> <strong>of</strong> Queensland,<br />
St Lucia, QLD 4067<br />
Research Officer, SEP2004-DEC2004<br />
Pharmacology Unit M510, School <strong>of</strong> Medicine and<br />
Pharmacology, QEII Medical Centre,<br />
The <strong>University</strong> <strong>of</strong> Western Australia, Crawley, WA<br />
6009<br />
Effect <strong>of</strong> static magnetic field on mechanosensitive<br />
channels, reconstituted into phospholipids bilayer<br />
Postdoctoral Fellow, SEP2001-MAR2003<br />
<strong>University</strong> <strong>of</strong> California at Davis, Center for<br />
Neuroscience, Dept. <strong>of</strong> Otolaryngology,<br />
1544 Newton Ct., Davis, CA 95616 USA<br />
Whole-cell Ca-current under membrane potential<br />
oscillations in hair cells.<br />
Ph.D., 1993-1995, Department <strong>of</strong> Biophysics,<br />
SSMU, Russia.<br />
Epithelium-derived relaxation in rat tracheal<br />
smooth muscles mediated by second<br />
messengers.<br />
M.S., 1992-1993, Department <strong>of</strong> Biophysics,<br />
SSMU, Russia.<br />
Influence <strong>of</strong> aorta endothelium and trachea<br />
epithelium on rats smooth muscle contractility<br />
Abstract<br />
High hydrostatic pressure (HHP) is present in<br />
natural environments where it impacts on<br />
biophysical properties <strong>of</strong> cell membranes and<br />
protein quaternary structure [1, 2]. We have<br />
investigated the effect <strong>of</strong> high hydrostatic pressure<br />
on E. coli mechanosensitive (MS) channels <strong>of</strong> small<br />
conductance MscS/MscK and G22E-MscL, a<br />
spontaneously opening mutant <strong>of</strong> E. coli MscL.<br />
Patch-clamp technique combined with a<br />
flying-patch device and hydraulic setup allowed<br />
the study <strong>of</strong> the effects <strong>of</strong> HHP up to 90 MPa on<br />
both MS channels in situ from the channels<br />
expressed in E. coli giant spheroplasts [3, 4, 5]. In<br />
addition, the G22E-MscL mutant channels were<br />
reconstituted into artificial liposome membranes<br />
[6] and examined for the effects HHP on their<br />
gating. Pressure affected MscS channel kinetics<br />
but not conductance [3]. At negative pipette<br />
voltages (corresponding to membrane<br />
depolarization in the inside-out patch configuration<br />
used in our experiments) the channel exhibited a<br />
reversible reduction in activity with increasing<br />
hydrostatic pressure between 0.1 and 90 MPa (1<br />
and 900 atm) at room temperature (�230C). The<br />
reduced activity was characterized by a significant<br />
reduction in the channel opening probability<br />
resulting from a shortening <strong>of</strong> the channel<br />
openings with increasing pressure. Thus HHP<br />
generally favoured channel closing in accordance<br />
with thermodynamic predictions. In contrast,<br />
against thermodynamic predictions, HHP in the<br />
same range <strong>of</strong> 0.1–90 MPa increased G22E-MscL<br />
channel open probability by favouring the open<br />
state <strong>of</strong> the channel [5]. Furthermore, hydrostatic<br />
pressure affected the channel kinetics, as<br />
manifested by the propensity <strong>of</strong> the channel to<br />
gate at subconducting levels with an increase in<br />
pressure. In the MscS case we postulate that<br />
lateral compression <strong>of</strong> the bilayer, under high<br />
hydrostatic pressure, is responsible for reduction<br />
in the channel open probability with increasing<br />
hydrostatic pressure. In the G22-MscL case we<br />
propose that the presence <strong>of</strong> water molecules<br />
around the hydrophobic gate <strong>of</strong> the G22E-MscL<br />
channel induce hydration <strong>of</strong> the hydrophobic lock<br />
under HHP causing frequent channel openings<br />
and preventing the channel closure in the absence<br />
<strong>of</strong> membrane tension. In conclusion, our studies<br />
indicate that HHP can be used as a valuable<br />
experimental approach towards better<br />
understanding <strong>of</strong> the gating mechanism in<br />
complex channels such as MscS and MscL.
References:<br />
[1] Macdonald, A. G. (1984) The effects <strong>of</strong> pressure on the<br />
molecular structure and physio- logical functions <strong>of</strong> cell<br />
membranes. Philos. Trans. R. Soc. Lond. B Biol. Sci. 304:47–68.<br />
[2] Mozhaev, V. V., K. Heremans, J. Frank, P. Masson, and C.<br />
Balny. (1996) High pressure effects on protein structure and<br />
function. Proteins 24:81-91.<br />
[3] Martinac, B., Buechner, M., Delcour, A.H.., Adler, J. and Kung,<br />
C. (1987) Pressure-sensitive ion channel in Escherichia coli. Proc.<br />
Natl. Acad. Sci. USA 84: 2297-2301.<br />
[4] Macdonald, A.G. and Martinac, B. (2005) Effect <strong>of</strong> high<br />
hydrostatic pressure on the bacterial mechanosensitive channel<br />
MscS. Eur. Biophys. J. 34: 434-442.<br />
[5] Petrov, E., Rohde, P.R. and Martinac, B. (2011) “Flying-patch”<br />
patch-clamp study <strong>of</strong> G22E MscL mutant under high hydrostatic<br />
pressure. Biophys. J. 100: 1635-1641. (Published as a Featured<br />
Article in Research Highlights <strong>of</strong> the 2nd March issue <strong>of</strong> BJ.)<br />
[6] Martinac, B., Rohde, P.R., Battle, A.R., Petrov, E., Pal, P., Fook<br />
Weng Foo, A., Vásquez, V., Huynh, T. and Kloda, A. (2010)<br />
Studying mechanosensitive ion channels using liposomes.<br />
Methods Mol. Biol. 606: 31-53.<br />
6.2.5 2:15pm – 2:30pm<br />
SPontaneous Oscillatory<br />
Contractions (SPOC): Assessing the<br />
Contractile Performance <strong>of</strong> Human<br />
Cardiomyopathies<br />
Amy Li1 , James Wolfe2 , Eleanor Kable3 , Filip<br />
Braet4 , Tatsuya Kagemoto 5 , <strong>Peter</strong> S Macdonald4 ,<br />
Shinichi Ishiwata3 , Cristobal dos Remedios8 1 Department <strong>of</strong> Anatomy & Histology, The <strong>University</strong> <strong>of</strong> Sydney,<br />
Sydney, NSW, 2006, amli7468@uni.sydney.edu.au<br />
2 Department <strong>of</strong> Anatomy & Histology, The <strong>University</strong> <strong>of</strong> Sydney,<br />
Sydney, NSW, 2006, jamesr@anatomy.usyd.edu.au<br />
3 Australian Centre for Microscopy & Microanalysis, The<br />
<strong>University</strong> <strong>of</strong> Sydney, Sydney, NSW, 2166, eleanor.kable@<br />
sydney.edu.au<br />
4 Australian Centre for Microscopy & Microanalysis, The<br />
<strong>University</strong> <strong>of</strong> Sydney, Sydney, NSW, 2166, filip.braet@sydney.<br />
edu.au<br />
5 Department <strong>of</strong> Physics, School <strong>of</strong> Science & Engineering,<br />
Waseda <strong>University</strong>, 3-4-1 Okubo, Shinjuku-ku, Tokyo 169-8555,<br />
Japan, yadara-fumia@ruri.waseda.jp<br />
6 Heart & Lung Transplant Unit, St. Vincent’s Hospital,<br />
Darlinghurst, NSW, pmacdonald@stvincents.com.au<br />
7 Department <strong>of</strong> Physics, School <strong>of</strong> Science & Engineering,<br />
Waseda <strong>University</strong>, 3-4-1 Okubo, Shinjuku-ku, Tokyo 169-8555,<br />
Japan, ishiwata@waseda.jp<br />
8 Department <strong>of</strong> Anatomy & Histology, The <strong>University</strong> <strong>of</strong> Sydney,<br />
Sydney, NSW, 2006, crisdos@anatomy.usyd.edu.au<br />
Biography<br />
Ms Li is currently completing her B.Sc. Honours<br />
degree under the supervision <strong>of</strong> Pr<strong>of</strong>essor Cris<br />
Biographies and Abstracts<br />
Tuesday 6 December - Session 6<br />
dos Remedios. In her third year she completed a<br />
double major in physiology and psychology and<br />
has an excellent track record. Her supervisor in<br />
confident she will be awarded first class honours<br />
and it is her intention to continue in 2012 as a PhD<br />
student.<br />
Abstract<br />
SPontaneous Oscillatory Contraction (SPOC) is a<br />
novel state <strong>of</strong> muscle contraction that can be<br />
observed in partially activated striated muscle<br />
fibres. SPOC is characterised by auto-oscillations<br />
between repetitive cycles <strong>of</strong> rapid lengthening and<br />
slow shortening which correlates with diastole and<br />
systole. There are two conditions by which<br />
cardiomyocytes can be activated; ADP-induced<br />
SPOC and Ca2+-induced SPOC. Ca-SPOC which<br />
has been shown to highly correlate with heart beat<br />
in a number <strong>of</strong> animal models.<br />
This study aims to quantify the contractile<br />
performance <strong>of</strong> cardiomyocytes isolated from<br />
human dilated and hypertrophic cardiomyopathies<br />
(some with known mutations) and compared with<br />
aged matched non-failing donors. The parameters<br />
<strong>of</strong> contractile performance include contraction and<br />
relaxation velocities, period <strong>of</strong> contraction and<br />
relaxation, SPOC velocity and SPOC period. We<br />
will compare contractile changes between (1)<br />
failing and non-failing hearts, (2) age-dependent<br />
progression and (3) differences between ADP- and<br />
Ca-SPOC in these hearts. These observations are<br />
then related to their patient data (e.g. ejection<br />
fraction, fractional shortening).<br />
Interestingly, we were able to re-activate human<br />
hearts that have been flash frozen at -200oC<br />
within minutes <strong>of</strong> loss <strong>of</strong> coronary blood flow<br />
(cross-clamping). Some samples have been stored<br />
for more than twenty years. Cardiac muscle fibres<br />
from the left atria and left ventricles, intraventricular<br />
septa and papillary muscles were treated with a<br />
series <strong>of</strong> washes prior to micro-dissection. These<br />
samples were then measured and averaged at<br />
very high spatial and temporal resolution using the<br />
Leica SP5 multi-photon microscope.<br />
Preliminary data so far have indicated that<br />
measurable changes in contractile performance<br />
were observed between non-failing and failing<br />
hearts. Samples <strong>of</strong> hypertrophic cardiomyopathy<br />
exhibited decreased rates <strong>of</strong> relaxation, consistent<br />
87
88<br />
Biographies and Abstracts<br />
Tuesday 6 December - Session 6<br />
with the clinically-described phenotype <strong>of</strong> diastolic<br />
dysfunction. These observations reveal SPOC to<br />
yield a range <strong>of</strong> functional parameters that can be<br />
used to evaluate the functional state <strong>of</strong> human<br />
heart muscle fibres. It may prove to provide a<br />
valuable set <strong>of</strong> parameters that can objectively<br />
assess the state <strong>of</strong> human heart failure.<br />
6.2.6 2:30pm – 2:45pm<br />
Does GLUT4 queue to get to the<br />
plasma membrane?<br />
Adelle C. F. Coster1 1 School <strong>of</strong> Mathematics & Statistics, <strong>University</strong> <strong>of</strong> New South<br />
Wales, Sydney NSW and Garvan Institute for Medical Research,<br />
Darlinghurst, NSW, A.Coster@unsw.edu.au<br />
Biography<br />
Adelle is an applied mathematician from UNSW<br />
who, in collaboration with the Diabetes & Obesity<br />
program at the Garvan Institute <strong>of</strong> Medical<br />
Research, has been modelling the spatio-temporal<br />
dynamics <strong>of</strong> the insulin signalling pathway and the<br />
ultimate translocation <strong>of</strong> the glucose transporter<br />
GLUT4 in response to differential levels <strong>of</strong> insulin.<br />
In between this and trying to get mathematicians<br />
into biology and biologists into mathematics she is<br />
also the Treasurer <strong>of</strong> the Australian Society for<br />
Biophysics.<br />
Abstract<br />
It is thought that the insulin-responsive glucose<br />
transporter protein GLUT4, which is a membrane<br />
embedded protein, recycles to the plasma<br />
membrane (PM) via the endosomes but is also<br />
sequestered in storage compartments. The<br />
sequestered GLUT4 can then be released into the<br />
recycling pathway in an insulin-dose dependent<br />
manner.<br />
The GLUT4 is exocytosed via small vesicles, which<br />
fuse with the PM. The application <strong>of</strong> insulin to a<br />
cell in the basal state causes an initial burst <strong>of</strong><br />
fusion events and then the rate returns to a steady<br />
lower level, in some cases similar to that observed<br />
in the basal case. It has been observed that the<br />
total amount recycling to the PM in the presence <strong>of</strong><br />
insulin is higher than in the basal state. It is also<br />
been observed that the endocytosis rate remains<br />
relatively constant irrespective <strong>of</strong> the insulin dose.<br />
Conversely, the transferrin receptor is thought to<br />
recycle to the PM, but when endocytosed only<br />
enters the early endosomes, being sorted away<br />
from the GLUT4 which is sequestered. Thus the<br />
transferrin receptor dynamics is controlled via<br />
fusions <strong>of</strong> the recycling pathway alone.<br />
Perturbation with insulin again causes an initial<br />
burst <strong>of</strong> transferring receptor fusion events, which<br />
then return to the near basal level.<br />
So we have two types <strong>of</strong> burst phenomena, that<br />
due to an additional entry <strong>of</strong> vesicles from a<br />
sequestered store and that due to a change in the<br />
recycling in an existing dynamic system.<br />
One possible model that could characterise this<br />
behaviour utilises queuing theory. This idea stems<br />
from the presence <strong>of</strong> the microtubules that cross<br />
the cytoplasm <strong>of</strong> the cells. Microtubules are<br />
implicated in the sorting <strong>of</strong> different endocytic<br />
vesicular contents and have well characterised<br />
molecular motors which control the movement <strong>of</strong><br />
vesicles down their lengths.<br />
Could these microtubules act as a scaffold for<br />
vesicles, which would then form queues waiting<br />
for fusion? The possible dynamics for such a<br />
system are explored for GLUT4 and transferrin<br />
receptor exocytosis in adipocytes.<br />
6.2.7 2:45pm – 3:00pm<br />
Dynamics <strong>of</strong> Protein Hydration Water<br />
Kathleen Wood1 , Francois-Xavier Gallat2 ,<br />
Douglas Tobias3 , Giuseppe Zaccai2 , Martin Weik4 1 Bragg Institute, Australian Nuclear Science and Technology<br />
Organisation, Locked Bag 2001, Kirrawee DC, NSW 2232,<br />
kwo@ansto.gov.au<br />
2 Institut Laue Langevin, Grenoble, France<br />
3 <strong>University</strong> <strong>of</strong> California, Irvine, California, USA<br />
4 Institut de Biologie Structurale, Grenoble, France<br />
Biography<br />
Katy Wood performed her PhD at the Institut Laue<br />
Langevin, Grenoble, France, where she studied<br />
protein and hydration water dynamics using<br />
neutron spectroscopy. She then moved to the<br />
Biological NMR group at the <strong>University</strong> <strong>of</strong><br />
Groningen in the Netherlands, studying intrinsically
Biographies and Abstracts<br />
Tuesday 6 December - Session 6<br />
unfolded proteins. She is currently jointly<br />
responsible for the small angle neutron scattering<br />
instrument at the Bragg Institute.<br />
Katy Wood performed her PhD at the Institut Laue<br />
Langevin, Grenoble, France, where she studied<br />
protein and hydration water dynamics using<br />
neutron spectroscopy. She then moved to the<br />
Biological NMR group at the <strong>University</strong> <strong>of</strong><br />
Groningen in the Netherlands, studying intrinsically<br />
unfolded proteins. She is currently jointly<br />
responsible for the small angle neutron scattering<br />
instrument at the Bragg Institute.<br />
Abstract<br />
Proteins are dynamical entities, interconverting<br />
between different structures to perform their<br />
functions. In a cellular context, the vast majority <strong>of</strong><br />
protein interactions are mediated by water, which<br />
can be considered a biomolecule in its own right.<br />
Here we characterise dynamics <strong>of</strong> proteins and<br />
their surrounding hydration layer.<br />
The influence <strong>of</strong> solvent dynamics on protein<br />
dynamics is so important that some have used the<br />
word ‘slaving’ to describe that water ‘imposes’ its<br />
dynamics on proteins. Combining neutron<br />
spectrosocpy with isotope labelling allowed us to<br />
study separately the dynamics <strong>of</strong> water and<br />
proteins in three systems: a soluble folded protein<br />
[2], an intrinsically disordered protein [3] and a<br />
membrane system [4]. We find a different<br />
relationship between water and protein dynamics.<br />
In the three systems.<br />
The results presented are the work <strong>of</strong> all authors<br />
on references [2-4].<br />
1. Frauenfelder, H., P.W. Fenimore, G. Chen, and B.H. McMahon,<br />
Protein folding is slaved to solvent motions. Proc Natl Acad Sci<br />
U S A, 2006. 103(42): p. 15469-72.<br />
2. Wood, K., A. Frolich, A. Paciaroni, M. Moulin, M. Hartlein, G.<br />
Zaccai, D.J. Tobias, and M. Weik, Coincidence <strong>of</strong> Dynamical<br />
Transitions in a Soluble Protein and Its Hydration Water: Direct<br />
Measurements by Neutron Scattering and MD Simulations. J<br />
Am Chem Soc, 2008. 130(14): p. 4586-4587.<br />
3. Gallat, F.X., A. Laganowski, K. Wood, F. Gabel, L. van Eijk, J.<br />
Wuttke, M. Moulin, M. Haertlein, D. Eisenberg, J-P. Colletier, G.<br />
Zaccai, M. Weik, under review.<br />
4. Wood, K., M. Plazanet, F. Gabel, B. Kessler, D. Oesterhelt, D.J.<br />
Tobias, G. Zaccai, and M. Weik, Coupling <strong>of</strong> protein and<br />
hydration-water dynamics in biological membranes. Proc Natl<br />
Acad Sci U S A, 2007. 104(46): p. 18049-54.<br />
Keynote Session - Main Theatre<br />
K.10 4:20pm - 4:50pm<br />
Dephosphorylation <strong>of</strong> the calcium<br />
pump – an infrared spectroscopy<br />
and density functional theory study<br />
Andreas Barth1 , Maria Rudbeck1 , Margareta<br />
Blomberg2 1 Department <strong>of</strong> Biochemistry and Biophysics, Stockholm<br />
<strong>University</strong>, Arrhenius Laboratories, 10691 Stockholm, Sweden,<br />
maria.rudbeck@gmail.com<br />
2 Department <strong>of</strong> Physics, Albanova, Stockholm <strong>University</strong>,<br />
Arrhenius Laboratories, 10691 Stockholm, Sweden, mb@fysik.<br />
su.se<br />
Biography<br />
Andreas Barth is Pr<strong>of</strong>essor in Experimental<br />
Molecular Biophysics at the Department <strong>of</strong><br />
Biochemistry and Biophysics <strong>of</strong> Stockholm<br />
<strong>University</strong> / Sweden. He is a trained physicist with<br />
a Diploma degree from the Technical <strong>University</strong> <strong>of</strong><br />
Darmstadt / Germany. Courses and research<br />
practice given by Hans-Joachim Galla made him<br />
switch to biophysics for his PhD which he did at<br />
the <strong>University</strong> <strong>of</strong> Freiburg /Germany with Werner<br />
Mäntele. He became then Wellcome fellow at the<br />
National Institute for Medical Research in London /<br />
UK with <strong>Peter</strong> Bayley and moved via Erlangen to<br />
Frankfurt am Main / Germany where he became<br />
Assistant Pr<strong>of</strong>essor. Since 2002 he has a position<br />
at Stockholm <strong>University</strong>.<br />
The common theme in his research has been the<br />
application <strong>of</strong> spectroscopic techniques to the<br />
study <strong>of</strong> proteins, with a particular focus on<br />
infrared spectroscopy. Current topics are: the<br />
mechanism <strong>of</strong> the Ca2+-ATPase, aggregation <strong>of</strong><br />
the Alzheimer’s peptide, membrane effects on<br />
membrane proteins, infrared spectroscopy as a<br />
tool for ligand binding studies and for analysing the<br />
effect <strong>of</strong> poisons on marine organisms.<br />
Abstract<br />
The sarcoplasmic reticulum Ca2+-ATPase<br />
(SERCA1a) actively pumps Ca2+ at the expense <strong>of</strong><br />
ATP hydrolysis, which leads to muscle relaxation.<br />
During the pump cycle, ATP phosphorylates the<br />
ATPase at Asp351 and the enzyme adopts two<br />
phosphoenzyme intermediates Ca2E1P and E2P.<br />
An important feature <strong>of</strong> E2P is its fast hydrolysis.<br />
89
90<br />
Biographies and Abstracts<br />
Tuesday 6 December - Session 6<br />
To understand the underlying mechanism, we<br />
performed an isotope exchange experiment which<br />
identified the absorption <strong>of</strong> the phosphate group <strong>of</strong><br />
the phosphorylated intermediates [1]. Major bands<br />
at 1194 and 1137 cm-1 are assigned to E2P, minor<br />
bands at 1175 and 1115 cm-1 to Ca2E1P.<br />
These data were evaluated by density functional<br />
theory (DFT) calculations (B3LYP/6-31++G**,<br />
6-311++G(3df, 3pd), where the aspartyl phosphate<br />
<strong>of</strong> the ATPase was modeled by acetyl phosphate<br />
and its interactions with the environment by HF<br />
molecules. These were placed at several locations<br />
and fixed distances with respect to acetyl<br />
phosphate. For the various model environments, a<br />
correlation was found between the average<br />
antisymmetric P-O stretching vibration and the<br />
bond length <strong>of</strong> the P-O bond that is cleaved during<br />
dephosphorylation. However, the data points<br />
scatter considerably and the models that most<br />
closely reproduce the experimental wavenumbers<br />
for acetyl phosphate in water and the ATPase<br />
phosphoenzymes do not indicate a lengthening <strong>of</strong><br />
the scissile P-O bond due to the protein<br />
environment.<br />
Protein models <strong>of</strong> the catalytic site derived from<br />
structural data were also analysed. They show that<br />
the reactant state <strong>of</strong> the dephosphorylation<br />
reaction is different from the E2P state adopted in<br />
solution. The reactant state has a longer scissile<br />
P-O bond but the expected wavenumbers <strong>of</strong> the<br />
reactant state are not observed in the experimental<br />
spectrum, indicating that the reactant state is<br />
considerably higher in free energy than the E2P<br />
ground state. The dephosphorylation reaction was<br />
modeled and its mechanism will be presented.<br />
Reference<br />
[1] A. Barth, N. Bezlyepkina (2004), J. Biol. Chem. 279,<br />
51888-51896<br />
K.11 4:50pm – 5:10pm<br />
Molecular Mechanisms <strong>of</strong> K+<br />
Selectivity <strong>of</strong> the Na/K Pump<br />
Haibo Yu1 1 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>, NSW 2522,<br />
Australia<br />
Biography<br />
Dr Haibo Yu joined the School <strong>of</strong> Chemistry at the<br />
<strong>University</strong> <strong>of</strong> <strong>Wollongong</strong> in July 2011. He attended<br />
the <strong>University</strong> <strong>of</strong> Science and Technology <strong>of</strong> China,<br />
where he received his B.Sc. in 2000. In Dec 2004,<br />
He completed his PhD in Chemistry at ETH Zurich,<br />
Switzerland with Pr<strong>of</strong> Wilfred F. van Gunsteren.<br />
Afterwards, he worked with Pr<strong>of</strong> Qiang Cui at the<br />
<strong>University</strong> <strong>of</strong> Wisconsin-Madison and Pr<strong>of</strong> Benoit<br />
Roux at the <strong>University</strong> <strong>of</strong> Chicago in the USA. His<br />
recent interests center on developing theoretical<br />
and computational models to study complex<br />
biomolecular systems.<br />
Abstract<br />
The sodium-potassium (Na/K) pump is a P-type<br />
ATPase that generates Na+ and K+ concentration<br />
gradients. For each ATP molecule, the pump<br />
extrudes three Na+ and imports two K+ across the<br />
cell membrane by alternating between outwardand<br />
inward-facing conformations that preferentially<br />
bind K+ or Na+, respectively. Remarkably, the<br />
selective K+ and Na+ binding sites share several<br />
residues, and how the pump is able to achieve the<br />
different selectivities at the shared sites required<br />
for the functional cycle is unclear. This talk will<br />
present our latest findings on molecular<br />
mechanisms <strong>of</strong> K+ selectivity <strong>of</strong> Na/K pump from<br />
a joint computational and electrophysiological<br />
study. Molecular simulations based on the crystal<br />
structures <strong>of</strong> the Na/K pump in a K+-loaded state<br />
reveal that protonation state <strong>of</strong> the acidic<br />
side-chains involved in the binding sites is critical<br />
to achieve the proper K+ selectivity. This prediction<br />
is tested with electrophysiological experiments<br />
showing that the selectivity <strong>of</strong> the E2P state for K+<br />
over Na+ is affected by extracellular pH.<br />
K.12 5:10pm – 5:30pm<br />
Towards rational control <strong>of</strong> the<br />
bacterial flagellar motor<br />
Lawrence Lee<br />
The Victor Chang Cardiac Research Institute<br />
Biography<br />
Lawrence Lee is a group leader at the Victor
Biographies and Abstracts<br />
Tuesday 6 December - Session 6<br />
Chang Cardiac Research Institute (VCCRI). He<br />
received his PhD from the Faculty <strong>of</strong> Pharmacy at<br />
the <strong>University</strong> <strong>of</strong> Sydney in 2008 and completed a<br />
postdoctoral fellowship in the Structural and<br />
Computational Biology Division <strong>of</strong> the VCCRI in<br />
August 2011 before establishing a research group<br />
in his current role. Using the bacterial flagellar<br />
motor as a model system, his group uses an<br />
interdisciplinary approach to determine molecular<br />
mechanisms that are fundamental to biology and<br />
to translate discovery into nano-technological<br />
advance in biomimicry.<br />
Abstract<br />
The bacterial flagellar motor (BFM) is a reversible<br />
biological nanorotor, which converts a flux <strong>of</strong><br />
cations into mechanical rotation. For over 30 years<br />
the BFM has been a canonical system in molecular<br />
and cellular biology as an example <strong>of</strong> a large,<br />
sophisticated protein complex and the endpoint <strong>of</strong><br />
a well-studied sensory network that facilitates<br />
bacterial chemotaxis. The latter has provided a<br />
paradigm for sensory networks in general. The<br />
BFM is also arguably the best in vivo-characterized<br />
molecular machine. However, the complete lack <strong>of</strong><br />
high-resolution structural data <strong>of</strong> functional<br />
subcomplexes hampers the BFMs promise to<br />
provide enormous insight into molecular<br />
processes that are fundamental to biology,<br />
including biological energy conversion and<br />
cooperative protein complex assembly and<br />
function. We recently determined the structure <strong>of</strong><br />
the rotor protein most directly involved in torque<br />
generation and rotational switching. This led to a<br />
detailed molecular picture <strong>of</strong> both the BFM’s ~1.5<br />
megadalton torque generating ring and a<br />
cooperative mechanism <strong>of</strong> rotational switching[1].<br />
Here, I describe some <strong>of</strong> our recent progress in<br />
taking this molecular picture into live cells to<br />
rationally probe the structure and function <strong>of</strong> the<br />
BFM in vivo.<br />
1. Lee LK, Ginsburg MA, Crovace C, Donohoe M, Stock D:<br />
Structure <strong>of</strong> the torque ring <strong>of</strong> the flagellar motor and the<br />
molecular basis for rotational switching. Nature (2010)<br />
466(7309):996-1000.<br />
Notes<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
91
92<br />
Poster Authors<br />
P1 Melatonin and serotonin in characeae<br />
Sabah Al Khazaaly 1 , Mary Jane Beilby 1 , Susan Murch 2 , Faisal Albisherawy 1<br />
1 The <strong>University</strong> <strong>of</strong> NSW, School <strong>of</strong> Physics, Kensington, Sydney, NSW, 2052, m.j.beilby@unsw.edu.au<br />
2 <strong>University</strong> <strong>of</strong> British Columbia, Okanagan , Kelowna, British Columbia, Canada, V1V 1V7 , susan.murch@ubc.ca<br />
P2 Zn2+ inactivates H+/OH- channels <strong>of</strong> Chara australis<br />
Sabah Al Khazaaly 1 , Mary Jane Beilby 1<br />
1 The <strong>University</strong> <strong>of</strong> NSW, School <strong>of</strong> Physics, Kensington, Sydney, NSW, 2052, s.alkhazaaly@unsw.edu.au<br />
1 The <strong>University</strong> <strong>of</strong> NSW, School <strong>of</strong> Physics, Kensington, Sydney, NSW, 2052, m.j.beilby@unsw.edu.au<br />
P3 Phenol and p-Chorophenol Adsorption onto Alumina-Grafted Different<br />
Polymers<br />
Hadi S. Al-Lami 1 , Ammar H. Al-Dujiali 2 , Maha T. Sultan 3<br />
1 Department <strong>of</strong> Chemistry, College <strong>of</strong> Science, <strong>University</strong> <strong>of</strong> Basra-Iraq<br />
2,3 Department <strong>of</strong> Chemistry, College <strong>of</strong> Education/ Ibn Al-Haitham, <strong>University</strong> <strong>of</strong> Baghdad-Iraq.<br />
P4 Experimental and Computational Investigations into Size Selected<br />
Gold Clusters on Semiconductor Supports as Photoelectrochemical<br />
Catalysts<br />
Jason F. Alvino1 , Greg F. Metha2 1 The <strong>University</strong> <strong>of</strong> Adelaide, North Terrace Campus, SA, 5005, Jason.alvino@adelaide.edu.au<br />
2 The <strong>University</strong> <strong>of</strong> Adelaide, North Terrace Campus, SA, 5005, greg.metha@adelaide.edu.au<br />
P5 High-resolution FTIR spectroscopy <strong>of</strong> the ground state, v8, v7, v6 and<br />
Coriolis “perturbation allowed” v12 and v10 modes <strong>of</strong> ketenimine.<br />
M. K. Bane1 , C. D. Thompson1 , E. G. Robertson 2 , D. R. T. Appadoo3 , C. Medcraft1, D.<br />
McNaughton1 .<br />
1 School <strong>of</strong> Chemistry, Monash <strong>University</strong>, Victoria 3800 Australia<br />
2 Department <strong>of</strong> Chemistry, La Trobe <strong>University</strong>, Victoria 3083 Australia<br />
3 Australian Synchrotron, 800 Blackburn Rd, Clayton, 3168, Victoria. Australia<br />
P6 An improved method, with theoretical analyses, for the simple<br />
experimental measurement <strong>of</strong> liquid junction potentials<br />
<strong>Peter</strong> H Barry1 , Trevor M Lewis1 , Andrew J Moorhouse1 1 Dept <strong>of</strong> Physiology, School <strong>of</strong> Medical Sciences, <strong>University</strong> <strong>of</strong> New South Wales, Sydney, NSW 2052, Australia. Email:<br />
p.barry@unsw.edu.au; t.lewis@unsw.edu.au; a.moorhouse@unsw.edu.au<br />
P7 Computational and Experimental investigations into catalytic<br />
applications <strong>of</strong> gas-phase gold-niobium bimetallic clusters<br />
Trystan Bennett1 , Robert A. Hardy1 , Gregory F. Metha1 1. The <strong>University</strong> <strong>of</strong> Adelaide, North Terrace Campus, Adelaide SA 5005. Trystan.bennett@adelaide.edu.au
P8 Designing single-molecule assays to image the dynamics <strong>of</strong> molecular<br />
chaperones<br />
Quill Bowden1 and Till Böcking2 1 Centre for Vascular Research, UNSW, Sydney 2052, q.bowden@unsw.edu.au<br />
2 Centre for Vascular Research, UNSW, Sydney 2052, till.boecking@unsw.edu.au<br />
P9 In silico modeling <strong>of</strong> protein dynamics and drug design<br />
Melissa J. Buskes1 , David J. D. Wilson2 , Belinda M. Abbott3 1 La Trobe Institute for Molecular Science, La Trobe <strong>University</strong>, Plenty Rd, Bundoora, VIC, 3086, M.Buskes@latrobe.edu.au<br />
2 La Trobe Institute for Molecular Science, La Trobe <strong>University</strong>, Plenty Rd, Bundoora, VIC, 3086, David.Wilson@latrobe.edu.au<br />
3 La Trobe Institute for Molecular Science, La Trobe <strong>University</strong>, Plenty Rd, Bundoora, VIC, 3086, B.Abbott@latrobe.edu.au<br />
P10 Differential Dynamic Microscopy Studies <strong>of</strong> Bacterial Motility<br />
C. Carnovale 1 , R. Nixon-Luke 1 , G. Bryant1 .<br />
1 School <strong>of</strong> Applied Sciences, RMIT <strong>University</strong>, GPO Box 2476, Melbourne, Victoria, 3000<br />
P11 Exciton Migration in Conjugated Polymer Dots<br />
Scott N. Clafton 1 , Dr David M. Huang2 , Ming Chiu3 and Dr Tak W. Kee4 1 <strong>University</strong> <strong>of</strong> Adelaide, North Terrace Adelaide, South Australia, 5005, scott.clafton@adelaide.edu.au<br />
2 <strong>University</strong> <strong>of</strong> Adelaide, North Terrace Adelaide, South Australia, 5005, david.huang@adelaide.edu.au<br />
3 <strong>University</strong> <strong>of</strong> Adelaide, North Terrace Adelaide, South Australia, 5005, ming.chiu@adelaide.edu.au<br />
4 <strong>University</strong> <strong>of</strong> Adelaide, North Terrace Adelaide, South Australia, 5005, tak.kee@adelaide.edu.au<br />
P12 Species differences in the kinetics <strong>of</strong> the Na+,K+-ATPase<br />
Ronald J. Clarke and Flemming Cornelius<br />
School <strong>of</strong> Chemistry,<strong>University</strong> <strong>of</strong> Sydney, Sydney, NSW 2001, Australia, and Department <strong>of</strong> Physiology and Biophysics,<br />
<strong>University</strong> <strong>of</strong> Aarhus, Aarhus, Denmark<br />
Correspondence.: r.clarke@chem.usyd.edu.au<br />
Poster Authors<br />
P13 Cardiac troponin: a paramagnetic relaxation enhancement NMR study<br />
Nicole M Cordina1 , C K Liew2 , D A Gell2 , J P Mackay2 , T M Logan3 , L J Brown1 1 Department <strong>of</strong> Chemistry and Biomolecular Sciences, Macquarie <strong>University</strong>, NSW 2109, Australia<br />
2 School <strong>of</strong> Molecular and Microbial Biosciences, <strong>University</strong> <strong>of</strong> Sydney, NSW 2006, Australia<br />
3 Institute <strong>of</strong> Molecular Biophysics, Florida State <strong>University</strong>, Tallahassee, FL 32306, USA<br />
P14 Unimolecular Reaction Chemistry <strong>of</strong> Atmospheric Peroxyl Radicals<br />
Gabriel da Silva<br />
Chemical and Biomolecular Engineering, The <strong>University</strong> <strong>of</strong> Melbourne, Parkville 3010, Australia<br />
P15 Structural Analysis <strong>of</strong> Missense Mutations in the CLIC2 Chloride<br />
Intracellular Ion Channel Protein<br />
E L Daniel1 , J E Hare2 , S C Goodchild3 , N M Cordina4 , L J Brown5 1 Department <strong>of</strong> Chemistry and Biomolecular Science, Macquarie <strong>University</strong>, Sydney, New South Wales, 2109, elizabeth.<br />
daniel@students.mq.edu.au<br />
93
94<br />
Poster Authors<br />
P16 The Role <strong>of</strong> Spin in Triplet-Triplet Upconversion<br />
A. Danos 1 , Y. Y. Cheng 1 , D. R. McCamey 2 , T. W. Schmidt 1<br />
1 School <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> Sydney, NSW 2006, Australia<br />
2 School <strong>of</strong> Physics, The <strong>University</strong> <strong>of</strong> Sydney, NSW 2006, Australia<br />
P18 Bonding between the uracil monomers <strong>of</strong> the cyclobutane pyrimidine<br />
dimer radical anion by means <strong>of</strong> quantum chemical calculations<br />
Linda Feketeová 2 , Andreas Mauracher1 ,David Gschliesser 1 , <strong>Peter</strong> Bartl1 , Violaine Vizcaino1 ,<br />
Lukas An der Lan1 , Catrin Goeschen2 , Stephan Denifl1 , Richard A.J. O’Hair2 , T. D. Märk1 , P.<br />
Scheier1 , U. Wille2 1 Institut für Ionenphysik und Angewandte Physik, Leopold-Franzens-Universität, Technikerstr. 25, Innsbruck, 6020, Austria,<br />
andreas.mauracher@uibk.ac.at<br />
2 ARC Centre <strong>of</strong> Excellence for Free Radical Chemistry and Biotechnology, School <strong>of</strong> Chemistry and Bio21 Institute <strong>of</strong><br />
Molecular Science and Biotechnology, The <strong>University</strong> <strong>of</strong> Melbourne, 30 Flemington Road, Victoria, 3010, Australia, lfe@unimelb.<br />
edu.au<br />
P19 Structure and binding energies <strong>of</strong> the doubly charged zwitterionic<br />
betaine clusters<br />
Linda Feketeová 2 , Emilie Cauët1 , William A. Donald2 , Richard A.J. O’Hair2 ,<br />
1 Service de Chimie Quantique et Photophysique, Université Libre de Bruxelles, avenue F.D. Roosevelt 50 CPi 160/09,<br />
Brussels, 1050, Belgium, ecauet@ulb.ac.be<br />
2 ARC Centre <strong>of</strong> Excellence for Free Radical Chemistry and Biotechnology, School <strong>of</strong> Chemistry and Bio21 Institute <strong>of</strong><br />
Molecular Science and Biotechnology, The <strong>University</strong> <strong>of</strong> Melbourne, 30 Flemington Road, Victoria, 3010, Australia, lfe@unimelb.<br />
edu.au<br />
P20 Parametric Sensitivity Analyses <strong>of</strong> the Insulin Signalling Pathway<br />
Catheryn Gray 1 , Adelle C. F. Coster 1,2<br />
1 School <strong>of</strong> Mathematics & Statistics, <strong>University</strong> <strong>of</strong> New South Wales<br />
2 Garvan Institute <strong>of</strong> Medical Research, Darlinghurst NSW<br />
P21 Energy and Charge Transfer Interactions in a Mixed Porphyrin Cosensitized<br />
TiO2 Electrode: A sub-ns Transient Absorption<br />
Spectroscopy Study<br />
M. J. Griffith, 1 A. J. Mozer, 1 P.Wagner, 1 G. G. Wallace, 1 D. L. Officer, 1 and R. Katoh 2,3<br />
1. ARC Centre <strong>of</strong> Excellence for Electromaterials Science and Intelligent Polymer Research Institute, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>,<br />
Innovation Campus, Squires Way, North <strong>Wollongong</strong>, NSW, 2500, Australia.<br />
Emails: mjg48@uow.edu, attila@uow.edu.au, pawel@uow.edu.au, gwallace@uow.edu.au, davido@uowmail.edu.au<br />
2. National Institute <strong>of</strong> Advanced Industrial Science and Technology, Tsukuba Central 5, Tsukuba, Ibaraki, 305-8565, Japan.<br />
3. Department <strong>of</strong> Chemical Biology and Applied Chemistry, College <strong>of</strong> Engineering, Nihon <strong>University</strong>, Tamura, Koriyama,<br />
Fukushima, 963-8642, Japan.<br />
Email: rkatoh@chem.ce.nihon-u.ac.jp<br />
P22 ACE I/D genotypes and their impact on heart rate variability: a limited<br />
meta-analysis<br />
Brett Hambly6 , Ethan Ng 1 , Yaxin Lu 2 , Slade Matthews3 , Herbert Jelinek4 , Craig McLachlan 5<br />
1 Pathology, Univ <strong>of</strong> Sydney, NSW 2006, etng1324@uni.sydney.edu.au<br />
2 Pathology, Univ <strong>of</strong> Sydney, NSW 2006, yalu4496@uni.sydney.edu.au<br />
3 Pharmacology, Univ <strong>of</strong> Sydney, NSW 2006, sladem@med.usyd.edu.au
4 School <strong>of</strong> Community Health, Charles Sturt Univ, NSW 2640, HJelinek@csu.edu.au<br />
5 Rural Clinical School, Univ <strong>of</strong> NSW, NSW, 2650, reperfusion@hotmail.com<br />
6 Pathology, Univ <strong>of</strong> Sydney, NSW 2006, bretth@pathology.usyd.edu.au<br />
Poster Authors<br />
P23 UV-Vis Action Spectroscopy <strong>of</strong> Room Temperature Protonated<br />
Aromatics<br />
Christopher S. Hansen1 , Ben B. Kirk1 , Richard A.J. O’hair 2 , Stephen J. Blanksby1, Adam<br />
J. Trevitt 1<br />
1 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>, NSW 2522 Australia<br />
2 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Melbourne, VIC 3010 Australia<br />
P24 Stabilisation and Delivery <strong>of</strong> Medicinal Pigment Curcumin by<br />
g-Cyclodextrin Dimers<br />
Takaaki Harada1 , Duc-Truc Pham1 , Huy Tien Ngo1 , Tiffany Harris2 , Eleanor Need2 , Grant<br />
Buchanan2 , Mandy Leung1 , Brendon Coventry, 3 Stephen F. Lincoln1 , Tak W. Kee1 1 School <strong>of</strong> Chemistry and Physics, The <strong>University</strong> <strong>of</strong> Adelaide, North Terrace Campus, Adelaide, SA, 5005, Australia, takaaki.<br />
harada@adelaide.edu.au<br />
2 Molecular Ageing Laboratory, School <strong>of</strong> Medicine, The <strong>University</strong> <strong>of</strong> Adelaide, Basil Hetzel Institute, The Queen Elizabeth<br />
Hospital, 28 Woodville Road, Woodville South, SA, 5011, Australia<br />
3 Discipline <strong>of</strong> Surgery, The <strong>University</strong> <strong>of</strong> Adelaide, Royal Adelaide Hospital, Adelaide, SA, 5005, Australia<br />
P25 Effective Core Potential Benchmarking for Cerium Oxide Clusters<br />
Using Density Functional Theory<br />
Robert A. Hardy1 , Birte Reichers2 and Gregory F. Metha3 1 Department <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> Adelaide, South Australia 5005, robert.hardy@adelaide.edu.au<br />
2 Department <strong>of</strong> Chemistry, Bielefeld <strong>University</strong>, 33615 Bielefeld, Germany, birte.reichers@uni-bielefeld.de<br />
3 Department <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> Adelaide, South Australia 5005, gregory.metha@adelaide.edu.au<br />
P26 Probing the Integral Membrane Form <strong>of</strong> Clic1 Using Fluorescence<br />
Resonance Energy Transfer<br />
Joanna E Hare1 , Sophia C Goodchild2, Louise J Brown3 1 Department <strong>of</strong> Chemistry and Biomolecular Science, Macquarie <strong>University</strong>, Sydney, New South Wales, 2109, joanna.hare@<br />
students.mq.edu.au<br />
2 Department <strong>of</strong> Chemistry and Biomolecular Science, Macquarie <strong>University</strong>, Sydney, New South Wales, 2109, sophia.<br />
goodchild@mq.edu.au<br />
3 Department <strong>of</strong> Chemistry and Biomolecular Science, Macquarie <strong>University</strong>, Sydney, New South Wales, 2109, louise.brown@<br />
mq.edu.au<br />
P27 Single Molecule Widefield Fluorescence Studies <strong>of</strong> Conjugated<br />
Polymers<br />
Emma N. Hooley1 , Toby D. M. Bell2 , Kenneth P. Ghiggino3 1 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Melbourne, Victoria 3010, e.hooley@student.unimelb.edu.au<br />
2 School <strong>of</strong> Chemistry, Monash <strong>University</strong>, Victoria, 3800, toby.bell@monash.edu<br />
3 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Melbourne, Victoria 3010, ghiggino@unimelb.edu.au<br />
95
96<br />
Poster Authors<br />
P28 Potential role <strong>of</strong> forbidden disulfide motifs in Zn fingers.<br />
Hulugalle D V K 1,3 , Haworth N L 2 , Ballouz S 1,4 , Jason Y. Liu 2 , Fan S W 1,3 , Wouters M A 2,3<br />
1 Structural and Computational Biology Program, Victor Chang Cardiac Research Institute, Sydney, Australia.<br />
2 School <strong>of</strong> Life and Environmental Sciences, Deakin <strong>University</strong>, Geelong, Australia<br />
3 School <strong>of</strong> Medical Sciences, <strong>University</strong> <strong>of</strong> New South Wales, Sydney, Australia<br />
4 School <strong>of</strong> Computer Science & Engineering, <strong>University</strong> <strong>of</strong> New South Wales, Sydney, Australia<br />
P29 Cryopreservation – The Beginning <strong>of</strong> a DSC (Differential Scanning<br />
Calorimetric) Understanding.<br />
Taavi Hunt 1 , Anja Kaczmarczyk 2, 3 , Bryn Funnekotter 2, 3, Shane Turner 3, 4 , Eric Bunn 3, 4 , Gary<br />
Bryant1 , Ricardo L. Mancera2 1 Applied Physics, School <strong>of</strong> Applied Sciences, RMIT <strong>University</strong>, Melbourne, VIC, Australia.<br />
2 Curtin Health Innovation Research Institute, Western Australian Biomedical Research Institute, Curtin <strong>University</strong>, Perth WA,<br />
Australia.<br />
3 Botanic Gardens and Parks Authority, Fraser Avenue, West Perth WA , Australia.<br />
4 School <strong>of</strong> Plant Biology, Faculty <strong>of</strong> Natural and Agricultural Sciences, <strong>University</strong> <strong>of</strong> Western Australia, Crawley WA, Australia.<br />
P30 A DNA-Based Assay for Chemical Toxicity in Wastewater and<br />
Drinking Water<br />
Vangelis George Kanellis1 , Amy Foreman1 , Leo Phillips1 , Cris dos Remedios1 , David<br />
Hibbert2 , John Chapman3 , Moreno Julli3 , Ronald Patra3 1 Anatomy and Histology, The <strong>University</strong> <strong>of</strong> Sydney, Sydney, Anderson Stuart Building (F13), Sydney <strong>University</strong>, NSW, 2006,<br />
Australia, cris.dosremedios@sydney.edu.au<br />
2 School Chemistry , <strong>University</strong> <strong>of</strong> New South Wales, Sydney, Dalton Building, <strong>University</strong> <strong>of</strong> New South Wales, Kensington,<br />
2052, b.hibbert@unsw.edu.au<br />
3 New South Wales Government,<br />
P31 Experimental and Computational Studies on Gas Phase Bimetallic<br />
Holmium-Rhodium Clusters<br />
Aidan M. Karayilan 1 , Gregory F. Metha1 1 <strong>University</strong> <strong>of</strong> Adelaide, North Terrace Campus Adelaide, SA, 5005, aidan.karayilan@adelaide.edu.au<br />
2 <strong>University</strong> <strong>of</strong> Adelaide, North Terrace Campus Adelaide, SA, 5005, greg.metha@adelaide.edu.au<br />
P32 Neutron membrane diffraction measurements <strong>of</strong> dehydrated DOPC<br />
bilayers with introduced sugars<br />
Ben Kent1 , Gary Bryant2 , Taavi Hunt2 and Christopher J. Garvey1 1 Bragg Institute, Australian Nuclear Science and Technology Organisation, Lucas Heights, Australia<br />
2 Applied Physics, School <strong>of</strong> Applied Sciences, RMIT <strong>University</strong>, Melbourne, Australia<br />
P33 •NO ejection from R• + •NO2. Does •NO2 add through N or O?<br />
Benjamin B. Kirk1 , Adam J. Trevitt 1 , Stephen J. Blanksby1 1 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>, NSW, 2522
P34 Non-Markovian Memory from Time-Local Stochastic Trajectories<br />
Werner Koch 1 , Frank Grossmann 1<br />
1 Research School <strong>of</strong> Chemistry, ANU, Canberra, ACT, 0200, Australia<br />
2 Institut für Theoretische Physik, TU Dresden, 01062 Dresden, Germany<br />
P35 Quantum Effects in H2-Li+-Benzene: A Model For Hydrogen Storage<br />
Materials<br />
Stephen J. Kolmann 1 , Meredith J. T. Jordan2 1 School <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> Sydney, NSW, 2006. email: s.kolmann@chem.usyd.edu.au<br />
2 School <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> Sydney, NSW, 2006. email: m.jordan@chem.usyd.edu.au<br />
P36 Anion photoelectron spectra <strong>of</strong> the halide-(N2)n and -(N2O)n clusters<br />
Kim M. Lapere1 , Allan J. McKinley1 & Duncan A. Wild1 1 Chemistry, <strong>University</strong> <strong>of</strong> Western Australia, M313 35 Stirling Hwy Crawley, WA 6009, 20169424@student.uwa.edu.au<br />
P37 Transient Absorption Spectroscopy <strong>of</strong> Curcumin-Copper Complex<br />
Hei Man Mandy Leung1 , Dr Tak W. Kee2 1 School <strong>of</strong> Chemistry and Physics, <strong>University</strong> <strong>of</strong> Adelaide, Adelaide, South Australia, 5005, Australia,<br />
hei.leung@adelaide.edu.au<br />
2 School <strong>of</strong> Chemistry and Physics, <strong>University</strong> <strong>of</strong> Adelaide, Adelaide, South Australia, 5005, Australia, tak.kee@adelaide.edu.au<br />
P38 Optimising the responsive behaviour <strong>of</strong> polymer surfaces using<br />
Molecular Dynamics<br />
Kamron Ley 1 , George Yiapanis 1 , Irene Yarovsky 1 , Evan Evans 2<br />
1 Applied Sciences, RMIT <strong>University</strong>, GPO BOX 2476V, Victoria, 3001, Australia<br />
2 BlueScope Steel Research, Port Kembla, NSW, Australia<br />
Poster Authors<br />
P39 Laser Spectroscopic Studies <strong>of</strong> Conformation in Neurotransmitters<br />
Isabella Antony Lobo1 , David Wilson2 , Evan Bieske3 , Evan G Robertson 4<br />
1 Department <strong>of</strong> Chemistry, La Trobe <strong>University</strong>, Bundoora, Victoria, 3086, ialobo@students.latrobe.edu.au<br />
2 Department <strong>of</strong> Chemistry, La Trobe <strong>University</strong>, Bundoora, Victoria, 3086, david.wilson@latrobe.edu.au<br />
3 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Melbourne, Victoria, 3010, evanjb@unimelb.edu.au<br />
4 Department <strong>of</strong> Chemistry, La Trobe <strong>University</strong>, Bundoora, Victoria, 3086, e.robertson@latrobe.edu.au<br />
P40 Single Cell Membrane Analysis by TERS is Reaching Nanometer Scale<br />
Christian Löbbe1 , Marc Richter2 , Heiko Haschke2 , Martin Hedegaard 3 , Tanja Deckert-<br />
Gaudig4 , <strong>Peter</strong> Lampen5 , Volker Deckert 4,6<br />
1 Scitech P/L, 4/72-74 Chifley Dr, Preston 3072, Australia<br />
2 JPK Instruments AG, Bouchestr. 12, 12435 Berlin, Germany<br />
3 Technical Faculty <strong>University</strong> <strong>of</strong> Southern Denmark, Institute <strong>of</strong> Sensors, Signals and Electrotechnics (SENSE), Campusvej 55,<br />
5230 Odense M, Denmark<br />
4 Institute <strong>of</strong> Photonic Technology (IPHT), Albert-Einstein-Straße 9, 07745 Jena, Germany<br />
5 Leibnitz-Institut für Analytische Wissenschaften – ISAS e.V., Bunsen-Kirchh<strong>of</strong>f-Str. 11, Dortmund 44139, Germany<br />
6 Friedrich-Schiller-Universität Jena, Institute <strong>of</strong> Physical Chemistry, Helmholzweg 4, 07743 Jena, Germany<br />
97
98<br />
Poster Authors<br />
P41 Reinvestigating the 308 nm Photodissociation Dynamics <strong>of</strong><br />
Acetaldehyde: A Velocity Map Imaging Study Examining Two Distinct<br />
Pathways Producing CO + CH4<br />
Alan T. Maccarone 1 , Mitchell S. Quinn2 , Gabi de Wit3 , Scott A. Reid4 , B. Klaas Nauta5 ,<br />
Scott H. Kable6 1 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney, New South Wales 2006, alanmac@uow.edu.au<br />
2 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney, New South Wales 2006, mqui5334@uni.sydney.edu.au<br />
3 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney, New South Wales 2006, g.dewit@chem.usyd.edu.au<br />
4 Department <strong>of</strong> Chemistry, Marquette <strong>University</strong>, Milwaukee, WI, USA, scott.reid@marquette.edu<br />
5 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney, New South Wales 2006, k.nauta@chem.usyd.edu.au<br />
6 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney, New South Wales 2006, s.kable@chem.usyd.edu.au<br />
P42 Understanding the immune interactions <strong>of</strong> an unstructured protein<br />
antigen: the malaria surface protein MSP2<br />
Christopher A. MacRaild1 , Marie Ø. Pedersen1 , Christopher G. Adda2 , Robin F. Anders2 and Raymond S. Norton1 1 Department <strong>of</strong> Medicinal Chemistry and Drug Action, Monash <strong>University</strong>, Melbourne, Australia.<br />
2 Department <strong>of</strong> Biochemistry, La Trobe <strong>University</strong>, Melbourne, Australia<br />
P43 Computational investigation <strong>of</strong> binding <strong>of</strong> kappa conotoxin to voltagegated<br />
potassium channels<br />
S. Mahdavi 1 and S. Kuyucak1 1-School <strong>of</strong> Physics, <strong>University</strong> <strong>of</strong> Sydney, NSW2006, Australia<br />
P44 Ab initio and classical molecular dynamics study <strong>of</strong> the aggregation<br />
propensities <strong>of</strong> amyloidogenic peptides in the presence <strong>of</strong><br />
nanomaterials<br />
A.J. Makarucha 1 , N. Todorova1 , A. Most<strong>of</strong>i 2 , I. Yarovsky 1<br />
1 Health Innovations Research Institute, School <strong>of</strong> Applied Sciences, RMIT <strong>University</strong>, GPO Box 2476 V, Melbourne, VIC,<br />
Australia.<br />
2 Department <strong>of</strong> Material s & Physics, Imperial College London, London, U.K., SW7 2AZ<br />
P45 Competitive N-O and O-C homolysis in TEMPO-based alkoxyamines<br />
David L. Marshall 1 , Martin R. L. Paine1 , Ganna Gryn’ova2 , Philip J. Barker3, Michelle L.<br />
Coote2 , Stephen J. Blanksby1 1 ARC Centre <strong>of</strong> Excellence for Free Radical Chemistry and Biotechnology & School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>,<br />
NSW, 2522<br />
2 ARC Centre <strong>of</strong> Excellence for Free Radical Chemistry and Biotechnology & Research School <strong>of</strong> Chemistry, Australian<br />
National <strong>University</strong>, Canberra ACT, 0200<br />
3 BlueScope Steel Research, P.O Box 202, Port Kembla, NSW, 2502<br />
P46 Thermodynamics and Metabolomics integration into metabolic<br />
Genome Scale Model to resolve metabolic flux directions<br />
Veronica Martinez1 , Stefanie Dietmair1 , Lake-Ee Quek1 , Lars Keld Nielsen1 1 Australian Institute for Bioengineering and Nanotechnology, The <strong>University</strong> <strong>of</strong> Queensland Australia , Corner College and<br />
Cooper Rds (Bldg 75) Brisbane, Qld, 4072
P47 Diagnostics for Orbital and Wavefunction Quality<br />
Poster Authors<br />
Laura K McKemmish 1 *, Jia Deng 1 and <strong>Peter</strong> Gill 1<br />
1 Research School <strong>of</strong> Chemistry, Australian National <strong>University</strong>, Science Road, Acton 2601 ACT Australia<br />
* laura.mckemmish@gmail.com<br />
P48 Far-Infrared spectroscopy <strong>of</strong> water aerosols using synchrotron<br />
radiation<br />
Chris Medcraft1,2 , Evan G. Robertson 3 , Dominque R.T. Appadoo2 , Sigurd Bauerecker 4 and<br />
Don McNaughton1 1 School <strong>of</strong> Chemistry, Monash <strong>University</strong>, Victoria 3800 Australia, chris.medcraft@monash.edu, don.mcnaughton@monash.<br />
edu<br />
2 Australian Synchrotron, 800 Blackburn Rd, Clayton, 3168, Victoria. Australia, dominque.appadoo@synchrotron.org.au<br />
3 Department <strong>of</strong> Chemistry, La Trobe <strong>University</strong>, Victoria 3083 Australia, E.Robertson@latrobe.edu.au<br />
4 Institut für Physikalische und Theoretische Chemie, Technische Universität Braunschweig, Hans-Sommer-Strasse 10,<br />
D-38106 Braunschweig, Germany<br />
P49 Quantum Chemical Studies <strong>of</strong> the Catalytic Mechanism <strong>of</strong> E3<br />
Hydrolysis <strong>of</strong> Organophosphates in the Australian Sheep Blowfly L.<br />
cuprina<br />
Tamara Meirelles1 , Junming Ho1 , Michelle Coote1 , Colin Jackson1 *<br />
1 Research School <strong>of</strong> Chemistry, Australian National <strong>University</strong> ACT 0200<br />
*cjackson@rsc.anu.edu.au<br />
P50 Ab Initio Folding <strong>of</strong> Helical Peptides Using Adaptive Hydrogen Bond-<br />
Specific Charge Scheme<br />
Siti Raudah Mohamed Lazim1 , Dawei Zhang2 Division <strong>of</strong> Chemistry and Biological Chemistry, School <strong>of</strong> Physical and Mathematical Sciences, Nanyang Technological<br />
<strong>University</strong>, 21 Nanyang Link, Singapore, 637371,<br />
1 SITI13@e.ntu.edu.sg<br />
2 zhangdw@ntu.edu.sg<br />
P51 Amyloid-like fibrillization and the structure <strong>of</strong> glucagon in the presence<br />
<strong>of</strong> anionic bicelles<br />
Ayano Momose 1 , Izumi Yamane 1 , Hideki Fujita 1 , Eri Yoshimoto 1 , Izuru Kawamura1 ,<br />
Marc-Antoine Sani2 , Frances Separovic2 , Akira Naito1 1 Graduate School <strong>of</strong> Engineering, Yokohama National <strong>University</strong>, Hodogaya-ku, Yokohama 240-8501, Japan naito@ynu.ac.jp<br />
2 School <strong>of</strong> Chemistry, Bio21 Institute, <strong>University</strong> <strong>of</strong> Melbourne, VIC 3010<br />
P52 Modelling Molecular Response in Anisotropic Electric Fields<br />
Michael Morris1 , Meredith Jordan2 1 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney, NSW, 2006, morris_m@chem.usyd.edu.au<br />
2 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney, NSW, 2006, mjtj@chem.usyd.edu.au<br />
99
Poster Authors<br />
P53 Examining the Electronic Interactions <strong>of</strong> Ligated Copper Nanoparticles<br />
Generated by Laser Ablation Synthesis in Solution (LASiS)<br />
Ashley Mulder1 , Mark Buntine1 , Aidon Slaney1 , Sean Long1 , Max Massi1 , Franca Jones1 ,<br />
Mark Ogden1 1 Department <strong>of</strong> Chemistry, Curtin <strong>University</strong>, GPO BOX U1987, Perth WA 6845<br />
P54 Patch Fluorimetry to Measure the Membrane Tension Required to Gate<br />
Mechanosensitive Ion Channels<br />
Takeshi Nomura1 , Charles Cranfield1,2 , Boris Martinac1,2 1 Victor Chang Cardiac Research Institute, Lowy-Packer Building, 405 Liverpool St, Darlinghurst, 2010.<br />
2 St Vincent’s Clinical School, The <strong>University</strong> <strong>of</strong> New South Wales, de Lacy, Victoria St, Darlinghurst, 2010<br />
T.nomura@victorchang.edu.au; C.cranfield@victorchang.edu.au; B.martinac@victorchang.edu.au<br />
P55 Structural characterization <strong>of</strong> triacylglycerols by radical directed<br />
dissociation<br />
Huong T. Pham 1 , Stephen J. Blanksby 2 , Gavin E. Reid 3<br />
1 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>, Australia, thp658@uow.edu.au<br />
2 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>, Australia, blanksby@uow.edu.au<br />
3 Department <strong>of</strong> Chemistry, Michigan State <strong>University</strong>, United States, reid@chemistry.msu.edu<br />
P56 Organic Monolayers for Water Evaporation Suppression: A Molecular<br />
Dynamic Study<br />
Michael Plazzer, George Yiapanis, Irene Yarovsky1 1 Applied Physics, School <strong>of</strong> Applied Sciences, RMIT <strong>University</strong>, Melbourne 3000 irene.yarovsky@rmit.edu.au<br />
P57 Langevin dynamics modelling <strong>of</strong> water diffusion in anisotropic<br />
biophysical structures.<br />
Sean Powell1 , Konstantin Momot1 1 Discipline <strong>of</strong> Physics, Queensland <strong>University</strong> <strong>of</strong> Technology, GPO Box 2434, Brisbane, QLD, 4001, sean.powell@qut.edu.au<br />
P58 Photodissociation Dynamics <strong>of</strong> Acetaldehyde at 308 nm: A Comparison<br />
<strong>of</strong> Experimental Studies and a Classical Trajectory Study <strong>of</strong> the<br />
Transition State Mechanism<br />
Mitchell S. Quinn1 , Gabi de Wit2 , Scott A. Reid3 , B. Klaas Nauta4 , Alan T. Maccarone 5 ,<br />
Scott H. Kable6 , Meredith J. T. Jordan7 P59 Alanine scan <strong>of</strong> an immunosuppressive peptide: Surface plasmon<br />
resonance analysis and structure-function relationships<br />
Laura Raguine1 , Marina Ali1 , Veronika Bender1 , Eve Diefenbach2 , Nicholas Manolios1 1 Department <strong>of</strong> Rheumatology, Westmead Hospital, Westmead, NSW 2145, Australia.<br />
2 Protein Production Facility, Westmead Millennium Institute, Westmead, NSW 2145, Australia.<br />
100
P60 Affinity and selectivity <strong>of</strong> sea anemone toxin ShK for the Kv1.1, Kv1.2<br />
and Kv1.3 channels from free energy simulations.<br />
M.H. Rashid and S. Kuyucak<br />
School <strong>of</strong> Physics, <strong>University</strong> <strong>of</strong> Sydney, NSW 2006, Australia,<br />
harun@physics.usyd.edu.au, serdar@physics.usyd.edu.au.<br />
P61 Large-scale fully ab initio calculations <strong>of</strong> ionic liquids using the<br />
Fragment Molecular Orbital approach<br />
Jason D. Rigby, Douglas R. MacFarlane, Ekaterina I. Izgorodina<br />
School <strong>of</strong> Chemistry, Monash <strong>University</strong>, Wellington Road, Victoria, 3800, E-Mail: jason.rigby@monash.edu<br />
P62 Structural studies <strong>of</strong> osmoregulatory ABC transporters<br />
Stephanie J. Ruiz12 , , Maaike Jansen, and Bert Poolman<br />
1 Department <strong>of</strong> Biochemistry, <strong>University</strong> <strong>of</strong> Groningen, The Netherlands<br />
2 s.j.ruiz@rug.nl<br />
P63 Determination <strong>of</strong> Threshold Dissociation Energies <strong>of</strong> Li+, Na+, K+ and<br />
Cs+ Cationized Dimers <strong>of</strong> 3-Hydroxyflavone, 5-Hydroxyflavone and<br />
5-Methoxyflavone by FTICR Mass Spectrometry and DFT Calculations<br />
Chowdhury Hasan Sarowar 1 , Michael Guilhaus2 , Gary David Willett3 , Grainne Moran4 1 School <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> New South Wales, Sydney, NSW 2052, Australia, c.h.sarowar@unsw.edu.au<br />
2 Bioanalytical Mass Spectrometry Facility, The <strong>University</strong> <strong>of</strong> New South Wales, Sydney, NSW 2052, Australia<br />
3 School <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> New South Wales, Sydney, NSW 2052, Australia, g.willett@unsw.edu.au<br />
4 Mark Wainwright Analytical Centre, The <strong>University</strong> <strong>of</strong> New South Wales, Sydney, NSW 2052, Australia, g.moran@unsw.edu.<br />
au<br />
P64 The impact <strong>of</strong> the ab initio/DFT method used for geometry<br />
optimisations on reaction enthalpies<br />
D. L. A. Scarborough, C. D. Thompson, E. I. Izgorodina<br />
School <strong>of</strong> Chemistry, Monash <strong>University</strong>, Victoria, Australia, 3800<br />
Email: David.Scarborough@monash.edu<br />
Poster Authors<br />
P65 Spectroscopy <strong>of</strong> resonance-stabilized hydrocarbon radicals<br />
Timothy W. Schmidt, Tyler P. Troy, Nahid Chalyavi, Zijun Ge, Gerard D. O’Connor,<br />
Masakazi Nakajima, Neil J. Reilly, Damian L. Kokkin, Klaas Nauta, Scott H. Kable<br />
School <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> Sydney, NSW 2006, Australia<br />
P66 Surface-enhanced Raman Spectroscopy <strong>of</strong> Is<strong>of</strong>lavones on Alternative<br />
Substrates<br />
Ryo Sekine1 , Evan G. Robertson, 2 Leone Spiccia3 , Richard A. Dluhy4 , Don McNaughton 5<br />
1 Centre for Biospectroscopy, Monash <strong>University</strong>, Clayton, VIC, 3800, Australia, ryo.sekine@monash.edu<br />
2 School <strong>of</strong> Molecular Sciences, La Trobe <strong>University</strong>, VIC, 3086, Australia, e.robertson@latrobe.edu.au<br />
3 School <strong>of</strong> Chemistry, Monash <strong>University</strong>, Clayton, VIC, 3800, Australia, leone.spiccia@monash.edu<br />
4 Department <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> Georgia, Athens, GA, USA, dluhy@uga.edu<br />
5 Centre for Biospectroscopy, Monash <strong>University</strong>, Clayton, VIC, 3800, Australia, don.mcnaughton@monash.edu<br />
101
Poster Authors<br />
P67 Dynamic Characterisation <strong>of</strong> Surfaces Using In-Silico Nano-<br />
Indentation<br />
102<br />
Lachlan Shaw 1 , George Yiapanis 1 , David Henry 2 , Evan Evans 3 , Irene Yarovsky 2<br />
1 School <strong>of</strong> Applied Sciences, RMIT <strong>University</strong>, GPO Box 2476, Vic, 3001<br />
2 School <strong>of</strong> Chemical and Mathematical Sciences, Murdoch <strong>University</strong>, South Street, WA, 6150<br />
3 BlueScope Steel Research Laboratories, Islands Rd, Port Kembla, NSW, 2505<br />
P68 CyDNA: A versatile photoswitchable biopolymer for advanced<br />
fluorescence microscopy applications.<br />
Darren A. Smith 1,2 ,*, Philipp Holliger 3 , Cristina Flors 1 ,*<br />
1 EaStChem School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Edinburgh, Edinburgh, EH9 3JJ, United Kingdom.<br />
2 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Melbourne, Melbourne, 3010, Australia.<br />
3 MRC Laboratory <strong>of</strong> Molecular Biology, Cambridge, CB2 0QH, United Kingdom.<br />
P69 Computational characterisation <strong>of</strong> an unusual metallacalix[4]arene<br />
dinitrogen activator<br />
Richard Terrett1 , Germán E. Cavigliasso1 , Rob Stranger1 , B.F. Yates 2<br />
1 Research School <strong>of</strong> Chemistry, Australian National <strong>University</strong>, ACT, 0200, email: rterrett@rsc.anu.edu.au<br />
2 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Tasmania, Private Bag 75, Hobart, TAS 7001<br />
P70 Recombination <strong>of</strong> photolytically generated iodine in single iodoalkane<br />
microdroplets<br />
Phillip J. Tracey1 , Bartholomew S. Vaughn1 , Adam J. Trevitt 1<br />
1 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>, NSW, 2522<br />
pjt105@uowmail.edu.au<br />
P71 Symmetry, Pseudo-symmetry and Evolution in Protein Structures<br />
Donald G Vanselow 1<br />
1 nativeproteins.blogspot.com 54 Greenways Rd., Glen Waverley VIC 3150, Australia. dvanselow@hotmail.com<br />
P72 Single Microdroplet Laser Spectroscopy<br />
Bartholomew S. Vaughn1 , Phillip J. Tracey 1 , Adam J. Trevitt 1 .<br />
1 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>, NSW 2522<br />
bv703@uowmail.edu.au<br />
P73 Molecular Dynamics <strong>of</strong> Curcumin and Linked Cyclodextrin Dimers<br />
Samuel J. Wallace 1 , David M. Huang2 , Takaaki Harada, 3 Tak W. Kee4 1 <strong>University</strong> <strong>of</strong> Adelaide, North Terrace, South Australia, 5005, sam.wallace@adelaide.edu.au<br />
2 <strong>University</strong> <strong>of</strong> Adelaide, North Terrace, South Australia, 5005, david.huang@adelaide.edu.au<br />
3 <strong>University</strong> <strong>of</strong> Adelaide, North Terrace, South Australia, 5005, takaaki.harada@adelaide.edu.au<br />
4 <strong>University</strong> <strong>of</strong> Adelaide, North Terrace, South Australia, 5005, tak.kee@adelaide.edu.au
Poster Authors<br />
P74 De novo Structure prediction <strong>of</strong> Peptide Based Biomimetic<br />
Carbohydrate Receptors<br />
Mark Waller1 1 Organic Chemistry Institute, <strong>University</strong> <strong>of</strong> Münster, Corrensstraße 40, D-48149 Münster, Germany<br />
P75 Modelling Cellulase Activity upon Cellulose Surfaces using Cellular<br />
Automata<br />
Andrew C. Warden1 , Bryce A. Little2 , Victoria S. Haritos1 1 CSIRO Ecosystem Sciences, Clunies Ross St, Acton, Canberra, ACT, 2601<br />
2 CSIRO Livestock Industries, Queensland Biosciences Precinct, 306 Carmody Road, St. Lucia, QLD, 4067<br />
P76 Lipid Composition Regulates the Conformation and Insertion <strong>of</strong> the<br />
Antimicrobial Peptide Maculatin 1.1<br />
Thomas Whitwell1 , Marc-Antoine Sani1 , Frances Separovic1 1 School <strong>of</strong> Chemistry, Bio21 Institute, <strong>University</strong> <strong>of</strong> Melbourne, VIC 3010<br />
P77 The Quokka Small Angle Neutron Scattering Instrument at OPAL<br />
K. Wood1 , C. J. Garvey1 , E. P. Gilbert1 1 Bragg Institute, Australian Nuclear Science and Technology Organisation, Locked Bag 2001, Kirrawee DC, NSW 2232,<br />
Australia<br />
P78 Structure, dynamics and interactions <strong>of</strong> malaria surface antigens<br />
Tessa R. Young 1 , David K. Chalmers1 , Christopher A. MacRaild1 , Marie O. Pedersen1 , Robin<br />
F. Anders2 , Raymond S. Norton1 1 Monash Institute <strong>of</strong> Pharmaceutical Sciences, Monash <strong>University</strong>, 381 Royal Parade, Parkville 3052, Australia<br />
2 Department <strong>of</strong> Biochemistry, La Trobe <strong>University</strong>, Bundoora, VIC 3086, Australia<br />
P79 New insights into the chemical reactivity <strong>of</strong> the deazaflavin c<strong>of</strong>actor<br />
F420 through quantum chemical calculations.<br />
Peng Yuan1 , Junming Ho1 , Colin J. Jackson1 , Michelle L. Coote1 1 Research School <strong>of</strong> Chemistry, Australian National <strong>University</strong>, ACT, 0200, coote@rsc.anu.edu.au<br />
P80 Utilization <strong>of</strong> Ambient Ozone for Determining Double Bond Positions in<br />
Unsaturated Lipids<br />
Shane R. Ellis1 , Marc in het Panhuis2 , Todd W. Mitchell3 , Stephen J. Blanksby1 ,<br />
1 ARC Centre <strong>of</strong> Excellence for Free Radical Chemistry and Biotechnology, School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>,<br />
<strong>Wollongong</strong>, NSW, 2522<br />
2 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>, <strong>Wollongong</strong>, NSW, 2522<br />
3 School <strong>of</strong> Health Sciences, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>, <strong>Wollongong</strong>,<br />
P81 Thi<strong>of</strong>lavin T and its derivatives: Revealing their spectroscopic<br />
properties in the absence and presence <strong>of</strong> insulin amyloid fibrils<br />
Eric H.-L. Chen 1 , Jack C.-C. Hsu 1 , Frederick Y. Luh 1 , T.-S. Lim2 , Rita P.-Y. Chen1 1 Institute <strong>of</strong> Biological Chemistry, Academia Sinica, Taipei, 11529, Taiwan<br />
2 Department <strong>of</strong> Physics, Tunghai <strong>University</strong>, Taichung 407, Taiwan<br />
103
Poster Presentations<br />
Sunday 4 December - Session 1<br />
P1<br />
Melatonin and serotonin in<br />
characeae<br />
Sabah Al Khazaaly 1 , Mary Jane Beilby1 ,<br />
Susan Murch2 , Faisal Albisherawy 1<br />
1 The <strong>University</strong> <strong>of</strong> NSW, School <strong>of</strong> Physics, Kensington, Sydney,<br />
NSW, 2052, m.j.beilby@unsw.edu.au<br />
2 <strong>University</strong> <strong>of</strong> British Columbia, Okanagan , Kelowna, British<br />
Columbia, Canada, V1V 1V7 , susan.murch@ubc.ca<br />
Abstract<br />
Neurohormone melatonin (N-acetyl-5methoxytryptamine)<br />
is secreted principally by the<br />
pineal gland <strong>of</strong> mammals including humans. The<br />
concentration levels exhibits a circadian cycle with<br />
a peak during the night. Other physiological<br />
functions may depend on the melatonin signal,<br />
such as immune antioxidative defences,<br />
hemostasis and glucose regulation (Claustrat et al.<br />
2005, Sleep Medicine Reviews 9: 11). Disturbed<br />
melatonin secretion is likely to predispose the<br />
organism to disease. Serotonin, which forms in the<br />
same metabolic pathway as melatonin, functions<br />
as a neurotransmitter in brains <strong>of</strong> animals.<br />
Recent research found melatonin and serotonin in<br />
higher plants (Murch and Saxena, 2002, In Vitro<br />
Cellular & Developmental Biology - Plant, 38, 531;<br />
Posmyk and Janas, 2009, Acta Physiologia<br />
Plantarum 31: 1). Some medicinal plants, such as<br />
St. John’s wort, contained up to gs per g <strong>of</strong> tissue.<br />
The structure <strong>of</strong> melatonin and serotonin is closely<br />
related to an important plant growth hormone<br />
auxin. It appears that these ancient molecules are<br />
highly conserved across biological kingdoms and<br />
might also act in plants as growth hormones,<br />
antioxidants and chronobiological substances.<br />
The giant-celled characeae are extant relatives <strong>of</strong><br />
ancestors <strong>of</strong> all land plants and are used<br />
extensively to model physiology and<br />
electrophysiology <strong>of</strong> land plant cells. Thus the<br />
presence or absence <strong>of</strong> melatonin and serotonin is<br />
<strong>of</strong> interest. Preliminary experiments with salt<br />
sensitive Chara australis and salt tolerant<br />
Lamprothamnium succinctum yielded<br />
comparatively high amounts <strong>of</strong> melatonin and<br />
serotonin <strong>of</strong> up to 4 g/g and 50 g/g, respectively.<br />
Melatonin and serotonin were extracted by<br />
104<br />
employing ethanol, purified by high performance<br />
liquid chromatography (HPLC) and indentified by<br />
mass spectrometry.<br />
The effects <strong>of</strong> varying day-night regimes, light <strong>of</strong><br />
different wavelength and Ca2+ concentration on<br />
the melatonin and serotonin levels will be tested at<br />
single cell level. Later experiments will include<br />
exogenously applied melatonin and inhibitors <strong>of</strong><br />
indoleamine metabolism, such as Ritalin and<br />
Prozac.<br />
P2<br />
Zn2+ inactivates H+/OH- channels <strong>of</strong><br />
Chara australis<br />
Sabah Al Khazaaly 1 , Mary Jane Beilby1 1 The <strong>University</strong> <strong>of</strong> NSW, School <strong>of</strong> Physics, Kensington, Sydney,<br />
NSW, 2052, s.alkhazaaly@unsw.edu.au<br />
1 The <strong>University</strong> <strong>of</strong> NSW, School <strong>of</strong> Physics, Kensington, Sydney,<br />
NSW, 2052, m.j.beilby@unsw.edu.au<br />
Abstract<br />
Zn2+ inactivates many types <strong>of</strong> animal H+<br />
channels by binding to histidine residues <strong>of</strong> the<br />
channel proteins (DeCoursey, 2010, Physiology,<br />
25: 27). Mercaptoethanol (ME) reverses this<br />
inhibition by scavenging the zinc ions. Are plant H+<br />
(or OH-) channels sufficiently similar to be affected<br />
by Zn2+?<br />
Giant-celled characeae, extant relatives to<br />
ancestors <strong>of</strong> all land plants, exhibit acid and<br />
alkaline banding with circulating external currents<br />
between the zones (Spear et al 1969, Journal <strong>of</strong><br />
General Physiology, 54: 397; Walker and Smith,<br />
1977, Journal <strong>of</strong> Experimental Botany, 28: 1190). In<br />
the acid zone the proton pump exports H+ out <strong>of</strong><br />
the cell, while in the alkaline zone H+/OHchannels<br />
facilitate either passive H+ influx or OHefflux.<br />
Different schemes have been proposed<br />
involving CO2 and HCO3- transport, but there is<br />
agreement that banding facilitates carbon<br />
assimilation in slightly alkaline ponds, where<br />
freshwater characeae live. Cells transferred into a<br />
medium <strong>of</strong> pH above 10.0 exhibit high<br />
conductance and the membrane PD (potential<br />
difference) behaves like a pH electrode: the whole<br />
cell becomes an alkaline band (Bisson and Walker,<br />
1980, Journal <strong>of</strong> Membrane Biology 56: 1). Upon
exposure to 1 mM ZnCl2, this high pH state is<br />
abolished, but can be restored by exposure to 0.5<br />
mM mercaptoethanol. Banding also disappears in<br />
response to ZnCl2.<br />
Beilby and Al Khazaaly (2009, Journal <strong>of</strong><br />
Membrane Biology, 230: 21) and Al Khazaaly et al<br />
(2009, European Biophysics Journal 39: 167)<br />
postulated that H+/OH- channels open transiently<br />
at the onset <strong>of</strong> saline stress in salt sensitive Chara<br />
australis, causing membrane PD noise; and remain<br />
open in latter stages <strong>of</strong> saline stress, contributing<br />
to cell deterioration. Zn2+ inhibited the saline noise<br />
and the upwardly concave I/V characteristics<br />
associated with the H+/OH- channel opening after<br />
several hours <strong>of</strong> saline stress. Both these effects<br />
could be reversed by ME. We will discuss the<br />
similarity <strong>of</strong> plant and animal H+/OH- channels and<br />
the increasing evidence for role <strong>of</strong> these channels<br />
in plant salinity stress.<br />
P3<br />
Phenol and p-Chorophenol<br />
Adsorption onto Alumina-Grafted<br />
Different Polymers<br />
Hadi S. Al-Lami 1 , Ammar H. Al-Dujiali 2 ,<br />
Maha T. Sultan 3<br />
1 Department <strong>of</strong> Chemistry, College <strong>of</strong> Science, <strong>University</strong> <strong>of</strong><br />
Basra-Iraq<br />
2,3 Department <strong>of</strong> Chemistry, College <strong>of</strong> Education/ Ibn<br />
Al-Haitham, <strong>University</strong> <strong>of</strong> Baghdad-Iraq.<br />
Abstract<br />
The ability <strong>of</strong> different alumina-grafted polymers<br />
was examined for adsorption <strong>of</strong> phenol and<br />
p-chlorophenol under different conditions (i.e.<br />
concentrations and temperatures). Adsorption<br />
behavior for standard alumina, alumina-graft<br />
acrylic acid monomer and other three polymers (A,<br />
B, C) in relation to adsorption <strong>of</strong> phenol and<br />
p-chlorophenol showed that adsorption follows<br />
Freundlich equation for phenol and p-chlorophenol<br />
while substances behavior was different in this<br />
process as the standard alumina adsorbed phenol<br />
better than others while the adsorption <strong>of</strong><br />
p-chlorophenol onto alumina-graft acrylic acid<br />
monomer and polymer A was better. Both<br />
polymers B and C showed a good adsorption for<br />
phenol and p-chlorophenol.<br />
Poster Presentations<br />
Sunday 4 December - Session 1<br />
P4<br />
Experimental and Computational<br />
Investigations into Size Selected<br />
Gold Clusters on Semiconductor<br />
Supports as Photoelectrochemical<br />
Catalysts<br />
Jason F. Alvino1 , Greg F. Metha2 1 The <strong>University</strong> <strong>of</strong> Adelaide, North Terrace Campus, SA, 5005,<br />
Jason.alvino@adelaide.edu.au<br />
2 The <strong>University</strong> <strong>of</strong> Adelaide, North Terrace Campus, SA, 5005,<br />
greg.metha@adelaide.edu.au<br />
Abstract<br />
For many years it has been known that<br />
semiconductor surfaces or particles such as TiO2<br />
can absorb light in order to directly drive the<br />
processes required to generate H2. It has been<br />
shown in recent years that doping TiO2 with<br />
various noble metal nanoparticles such as Au or Pt<br />
can promote H2 generation by acting as a<br />
‘reservoir’ for photo-generated electrons.<br />
Experimental studies are being developed to<br />
investigate the photo-catalytic potential <strong>of</strong> various<br />
configurations <strong>of</strong> size selected gold clusters on<br />
semiconductor surfaces using an experimental<br />
apparatus that can provide immediate feedback<br />
on the gas composition <strong>of</strong> the reaction.<br />
Computational studies have been performed on<br />
various rutile (110) and anatase (101) TiO2 surface<br />
models utilizing the ONIOM partitioning scheme.<br />
This computational technique can model large<br />
systems by defining two or three layers within the<br />
structure that are treated with varying levels <strong>of</strong><br />
accuracy. In this work the high layer was treated<br />
with density functional theory and the low layer<br />
surrounding lattice structure was treated with<br />
molecular mechanics methods. This has allowed<br />
for the modelling <strong>of</strong> a large area <strong>of</strong> the TiO2<br />
surface with minimal computational expense while<br />
utilizing atomic centred basis sets for the<br />
interaction site. Various types <strong>of</strong> defect sites were<br />
modelled including but not limited to i) oxygen<br />
vacancies, ii) smooth step-edges, and iii) rough<br />
step edges. Au, Au2 and Au3 clusters were then<br />
added to these defect sites to determine the<br />
binding energy, reaction pr<strong>of</strong>iles, density <strong>of</strong> states<br />
and morphological changes to the TiO2 surface.<br />
105
Poster Presentations<br />
Sunday 4 December - Session 1<br />
The experimental apparatus and an explanation <strong>of</strong><br />
the computational procedure, including preliminary<br />
results, will be presented in this poster.<br />
P5<br />
High-resolution FTIR spectroscopy<br />
<strong>of</strong> the ground state, v8, v7, v6 and<br />
Coriolis “perturbation allowed” v12<br />
and v10 modes <strong>of</strong> ketenimine.<br />
M. K. Bane1 , C. D. Thompson1 , E. G.<br />
Robertson2 , D. R. T. Appadoo3 , C. Medcraft1, D.<br />
McNaughton1 .<br />
1 School <strong>of</strong> Chemistry, Monash <strong>University</strong>, Victoria 3800 Australia<br />
2 Department <strong>of</strong> Chemistry, La Trobe <strong>University</strong>, Victoria 3083<br />
Australia<br />
3 Australian Synchrotron, 800 Blackburn Rd, Clayton, 3168,<br />
Victoria. Australia<br />
Abstract<br />
Ketenimine (CH2CNH) is a transient molecule that<br />
is <strong>of</strong> interest in interstellar chemistry and about<br />
which little is known. This tautomer <strong>of</strong> acetonitrile<br />
has been identified in the star forming region<br />
Sagittarius B2(N) by observing microwave<br />
emissions (1). In this study far- and mid-IR high<br />
resolution spectra were recorded using a Bruker<br />
HR125 coupled to the far-IR beam line <strong>of</strong> the<br />
Australian synchrotron and both pure ground state<br />
rotational transitions and ro-vibrational transitions<br />
were recorded.<br />
The excited vibrational states exhibit a complex<br />
ro-vibrational structure, due primarily to strong<br />
Coriolis interactions. As a consequence <strong>of</strong> this<br />
coupling, some <strong>of</strong> these modes exhibit a novel<br />
intensity sharing effect, with the relatively weak v10<br />
and v12 (2) bands being analyzed completely<br />
using perturbation allowed transitions, which<br />
exhibit intensities independent <strong>of</strong> their own natural<br />
dipole moment derivative.<br />
The analysis <strong>of</strong> v7 (3), v10 and v6 (4) were also<br />
complicated by the presence <strong>of</strong> local Fermi and<br />
Coriolis resonances with the higher order<br />
excitations <strong>of</strong> v12 and v8, which are themselves<br />
strongly Coriolis coupled. Analysis <strong>of</strong> ground state<br />
combination differences <strong>of</strong> v7 also uncovered<br />
second order Coriolis interactions between the<br />
106<br />
ground state and both v12 and v8 at high Ka, and<br />
a global fit including the ground state, v12, v8, v7,<br />
2v12, v12 + v8, 2v8, v10 and v6 was achieved. The<br />
combination and overtones are included in the fit<br />
as dark-states, since they were too weak to be<br />
observed.<br />
1. F. J. Lovas, J. M. Hollis, A. J. Remijan and P. R. Jewell,<br />
Astrophys. J., 2006, 645, L137<br />
2. M. K. Bane, C. D. Thompson, E. G. Robertson, R. T. Appadoo<br />
and D. McNaughton, Phys. Chem. Chem. Phys., 2011, 13, 6793<br />
3. M. K. Bane, E. G. Robertson, C. D. Thompson, R. T. Appadoo,<br />
C. Medcraft and D. McNaughton, J. Chem. Phys., 2011, 134,<br />
234306<br />
4. Analysis <strong>of</strong> v10 and v6 is currently unpublished<br />
P6<br />
An improved method, with<br />
theoretical analyses, for the simple<br />
experimental measurement <strong>of</strong> liquid<br />
junction potentials<br />
<strong>Peter</strong> H Barry1 , Trevor M Lewis1 , Andrew J<br />
Moorhouse1 1 Dept <strong>of</strong> Physiology, School <strong>of</strong> Medical Sciences, <strong>University</strong> <strong>of</strong><br />
New South Wales, Sydney, NSW 2052, Australia. Email: p.<br />
barry@unsw.edu.au; t.lewis@unsw.edu.au; a.moorhouse@<br />
unsw.edu.au<br />
Abstract<br />
In electrophysiological experiments, accurate<br />
potential measurements require corrections for<br />
liquid junction potentials (LJPs). Under certain<br />
conditions, as in patch clamp experiments and<br />
dilution potential measurements, their magnitude<br />
can <strong>of</strong>ten be ~5-10 mV or more. In most cases,<br />
where the ion mobilities are known, LJP<br />
corrections can be simply calculated. However, in<br />
order to confirm such calculations, or if ion<br />
mobilities are not known accurately, it is necessary<br />
to measure LJPs experimentally. We describe here<br />
an improved simple and accurate method for<br />
doing this using a freshly-cut 3M KCl-agar
salt-bridge (in polyethylene tubing) as a reference<br />
electrode. Critical to success, is cutting <strong>of</strong>f at least<br />
the last 5 mm <strong>of</strong> this KCl-agar salt-bridge just<br />
before it is placed into a different test solution. This<br />
ensures a fresh 3M KCl-agar surface in contact<br />
with the test solution, and eliminates the major<br />
history-dependent problem that normally arises<br />
with 3M KCl reference salt-bridges (e.g., [1] and<br />
[2]). The measured potentials also need to be<br />
corrected for a small, well-defined and easily<br />
calculable, shift in LJPs at this 3M KCl salt-bridge.<br />
We directly demonstrate the extent <strong>of</strong> the<br />
history-dependent problems and how the<br />
freshly-cut tip eliminates them, supporting these<br />
experiments with new theoretical analyses <strong>of</strong> NaCl<br />
diffusing into a 3M KCl-agar salt bridge and the<br />
KCl diffusing out <strong>of</strong> it, with diffusion coefficients<br />
corrected for the 4% agar gel used in the<br />
salt-bridges [3]. Using this improved technique, we<br />
have measured LJPs for diluted solutions <strong>of</strong> NaCl<br />
(with and without CaCl2), LiCl, KCl and CsCl and<br />
for biionic combinations <strong>of</strong> the undiluted salts<br />
(some earlier measurements are in [4, 5]). The<br />
measured values give excellent agreement<br />
(generally within expected errors <strong>of</strong> 0.1 mV) with<br />
LJPs theoretically calculated with the Henderson<br />
equation [6], and incorporated into a liquid junction<br />
potential calculation program (JPCalc; [7]). We also<br />
demonstrate that ion activities rather than<br />
concentrations should be used for dilution LJP<br />
calculations.<br />
1. Barry, PH & Diamond, JM (1970) J Membrane Biol. 3: 393-122<br />
2. Neher E (1992) Meth. Enzym. 207: 123-131<br />
3. Slade AL, Cremers AE, Thomas HC (1966). J. Phys.Chem. 70:<br />
2840-2844.<br />
4. Sugiharto S, Carland JE, Lewis TM, Moorhouse AJ, Barry PH<br />
(2010). Pflügers Archiv 460: 131-152.<br />
5. Barry PH, Sugiharto S, Lewis TM, Moorhouse AJ. (2010)<br />
Channels 4: 142-149<br />
6. Barry PH, Lynch JW. (1991) J. Membrane Biol. 121: 101-117.<br />
7. Barry, PH (1994) J. Neurosci. Meth. 51: 107-116<br />
Poster Presentations<br />
Sunday 4 December - Session 1<br />
P7<br />
Computational and Experimental<br />
investigations into catalytic<br />
applications <strong>of</strong> gas-phase goldniobium<br />
bimetallic clusters<br />
Trystan Bennett1 , Robert A. Hardy1 , Gregory<br />
F. Metha1 1. The <strong>University</strong> <strong>of</strong> Adelaide, North Terrace Campus, Adelaide SA<br />
5005. Trystan.bennett@adelaide.edu.au<br />
Abstract<br />
Mass Spectra <strong>of</strong> oxygen-poor bimetallic Au/Nb<br />
clusters (AunNbxOy; n=0-2,x=1-5,y=0-3) were<br />
recorded from clusters produced in the gas phase<br />
using laser ablation, and ionised via multi-photon<br />
ionisation with 220 nm light.<br />
Computational studies were undertaken on all<br />
species observed, using density functional theory<br />
at the M06-L/SDD/aug-cc-pVTZ level <strong>of</strong> theory.<br />
Gold atoms incorporated into these molecules<br />
were found to attain δ- charge build-up, with<br />
correlation between charge magnitude and the<br />
oxidation state <strong>of</strong> the niobium atoms. Evidence <strong>of</strong><br />
gold substitution <strong>of</strong> both niobium and oxygen<br />
atoms was found within predicted geometries.<br />
Benchmarking <strong>of</strong> eleven density functionals was<br />
undertaken across ionisation energies and<br />
electron affinity calculations, with M06-L found to<br />
be superior for calculations <strong>of</strong> niobium oxide<br />
clusters.<br />
This poster will present findings from the<br />
benchmarking <strong>of</strong> eleven density functionals with<br />
niobium oxide clusters, modelling both electron<br />
affinities and ionisation energies. The functionals<br />
benchmarked represent a wide array <strong>of</strong><br />
approaches, from pure DFT approaches such as<br />
B97-D and M06-L, to hybrid DFT approaches<br />
such as B3LYP, B3P86, and B98.<br />
Further results are presented from the<br />
experimental/computational application <strong>of</strong> the best<br />
performing functional found during benchmarking,<br />
M06-L. Structure calculations and Hirschfeld<br />
charge investigations were performed upon 16<br />
bimetallic gold-niobium oxide clusters, ranging in<br />
size from 3 to 9 atoms. Examinations <strong>of</strong> electronic<br />
structure, motif development and structure trends<br />
107
Poster Presentations<br />
Sunday 4 December - Session 1<br />
trend are undertaken, with the end goal <strong>of</strong><br />
assessing the bimetallic clusters’ suitability for<br />
further surface-deposition catalytic studies.<br />
P8<br />
Designing single-molecule assays to<br />
image the dynamics <strong>of</strong> molecular<br />
chaperones<br />
Quill Bowden1 and Till Böcking2 1 Centre for Vascular Research, UNSW, Sydney 2052,<br />
q.bowden@unsw.edu.au<br />
2 Centre for Vascular Research, UNSW, Sydney 2052, till.<br />
boecking@unsw.edu.au<br />
Abstract<br />
Molecular chaperones maintain protein<br />
homeostasis in the cell by catalysing a wide variety<br />
<strong>of</strong> processes throughout a protein’s life cycle. Their<br />
functions include protein folding, remodelling <strong>of</strong><br />
protein assemblies, protection from denaturation<br />
and resolubilisation <strong>of</strong> proteins from aggregates<br />
that accumulate during cell stress. The aim <strong>of</strong> our<br />
research is to elucidate the molecular mechanisms<br />
<strong>of</strong> how chaperones from the Hsp70 family interact<br />
with proteins that have assembled into oligomers<br />
or are on the pathway towards aggregate<br />
formation. Utilisation <strong>of</strong> single-molecule<br />
fluorescence microscopy techniques will allow us<br />
to observe individual molecules in the dynamic<br />
chaperone-substrate interactions and thus resolve<br />
reaction pathways that are obscured in traditional<br />
ensemble approaches. Here we present our initial<br />
results on developing strategies for protein<br />
labelling and design <strong>of</strong> the fluorescence imaging<br />
assay. This research has implications for a range<br />
<strong>of</strong> neurodegenerative diseases that are<br />
characterised by the accumulation <strong>of</strong> insoluble<br />
protein aggregates (amyloid fibrils). Disease states<br />
were previously attributed to the presence <strong>of</strong> these<br />
amyloids in the neuronal regions, however current<br />
research suggests that toxicity is actually exerted<br />
by soluble pre-fibrillar species. A better<br />
understanding <strong>of</strong> the interactions <strong>of</strong> chaperones<br />
with assemblies <strong>of</strong> disease proteins will shed light<br />
on the role <strong>of</strong> chaperones in modulating the<br />
progression <strong>of</strong> these diseases.<br />
108<br />
P9<br />
In silico modeling <strong>of</strong> protein<br />
dynamics and drug design<br />
Melissa J. Buskes1 , David J. D. Wilson2 ,<br />
Belinda M. Abbott3 1 La Trobe Institute for Molecular Science, La Trobe <strong>University</strong>,<br />
Plenty Rd, Bundoora, VIC, 3086, M.Buskes@latrobe.edu.au<br />
2 La Trobe Institute for Molecular Science, La Trobe <strong>University</strong>,<br />
Plenty Rd, Bundoora, VIC, 3086, David.Wilson@latrobe.edu.au<br />
3 La Trobe Institute for Molecular Science, La Trobe <strong>University</strong>,<br />
Plenty Rd, Bundoora, VIC, 3086, B.Abbott@latrobe.edu.au<br />
Abstract<br />
This presentation reports current results from an<br />
ongoing study aimed at identifying selective and<br />
potent inhibitors <strong>of</strong> target kinases through<br />
molecular modeling – computer aided drug<br />
design. The study <strong>of</strong> the interaction <strong>of</strong> small<br />
molecules with proteins is a long held interest in<br />
the Molecular Modeling group at La Trobe<br />
<strong>University</strong>.<br />
The work presented here builds on previous<br />
studies designed to develop a potent inhibitor <strong>of</strong><br />
the target kinase, with a major focus on the issue<br />
<strong>of</strong> selectivity <strong>of</strong> inhibitors. This was achieved by<br />
modeling inhibitors through selected protein<br />
kinases. Biological assay results <strong>of</strong> our lead series<br />
<strong>of</strong> compounds highlight selectivity matters<br />
between these particular kinases, and these<br />
kinases were therefore selected for analysis. Our<br />
goal was to optimize the lead compounds as a<br />
result <strong>of</strong> modeling selectivity.<br />
Results <strong>of</strong> molecular dynamics modeling <strong>of</strong><br />
inhibitors <strong>of</strong> the protein kinases will be presented.<br />
In this work we have extensively used the AMBER<br />
package in conjunction with the MM-PBSA and<br />
MM-GBSA methods. Analysis <strong>of</strong> model<br />
preparation, benchmarking and validation <strong>of</strong><br />
methodology, in comparison with experimental<br />
data, was carried out in order to provide an<br />
indication <strong>of</strong> accuracy, reproducibility and reliability<br />
<strong>of</strong> this approach to the calculation <strong>of</strong> binding<br />
energies.
P10<br />
Differential Dynamic Microscopy<br />
Studies <strong>of</strong> Bacterial Motility<br />
C. Carnovale 1 , R. Nixon-Luke 1 , G. Bryant1 .<br />
1 School <strong>of</strong> Applied Sciences, RMIT <strong>University</strong>, GPO Box 2476,<br />
Melbourne, Victoria, 3000<br />
Abstract<br />
The importance <strong>of</strong> cell motility is apparent in many<br />
facets <strong>of</strong> biology. It plays a fundamental role in the<br />
process <strong>of</strong> internal fertilisation in vivo as well as<br />
disease transmission and the spread <strong>of</strong> infection.<br />
Used <strong>of</strong>ten as a model for understanding bacterial<br />
motility, Escherichia coli generally exhibits random<br />
walk movement in isotropic conditions.<br />
Responsible for the motility <strong>of</strong> E. Coli, are<br />
appendages called flagella which are arranged<br />
laterally and distributed over the entire external cell<br />
surface. This peritrichous arrangement <strong>of</strong> flagella<br />
allow E. Coli to alternate forward motion driven by<br />
counter clockwise flagella rotation for ~1 second<br />
before reversal to clockwise movement for ~0.1<br />
second which prompts a change in direction<br />
through a brief tumbling motion [1].<br />
The use <strong>of</strong> dynamic light scattering to analyse<br />
bacterial motility has been explored for many<br />
decades. In 1971 a seminal study performed by<br />
Nossal, Chen & Lai [2] established one <strong>of</strong> the<br />
earliest autocorrelation functions to interpret the<br />
swimming speed distributions <strong>of</strong> E. Coli. While this<br />
method <strong>of</strong> analysis provides accurate data for<br />
diffusion rate and the fraction <strong>of</strong> non motile cells,<br />
limitations <strong>of</strong> the model mean that non linear<br />
movement such as tumbling cannot be accounted<br />
for. Whilst using a low scattering angle (and<br />
effectively a low scattering vector), lessens<br />
undesirable contribution from the tumbling<br />
motions, limitations in equipment capability<br />
prevent this from being a possibility in most<br />
laboratory settings.<br />
Initially tested with colloidal suspensions [3],<br />
differential dynamic microscopy (DDM) is a new<br />
microscope technique based on the principles <strong>of</strong><br />
DLS which has recently been tested using<br />
bacterial samples [4]. The main advantage over<br />
DLS is that measurements can be taken using a<br />
very small scattering vector which effectively<br />
Poster Presentations<br />
Sunday 4 December - Session 1<br />
negates the contributions from the irregular<br />
movement and tumbling motions <strong>of</strong> E.Coli.<br />
In this abstract, we report on measurements <strong>of</strong><br />
bacterial motility through using DDM. We analyse a<br />
number <strong>of</strong> strains <strong>of</strong> bacteria with different flagella<br />
arrangements, and compare the results with<br />
traditional methods.<br />
We discuss the application to other motile<br />
micro-organisms <strong>of</strong> varying sizes as well as future<br />
generations <strong>of</strong> synthesised self motile molecules<br />
1. Berg, H.C. and D.A. Brown, Chemotaxis in Escherichia coli<br />
analyzed by three-dimensional tracking. Antibiotics and<br />
chemotherapy, 1974. 19: p. 55-78.<br />
2. Nossal, R., S.H. Chen, and C.C. Lai, Use <strong>of</strong> laser scattering for<br />
quantitative determinations <strong>of</strong> bacterial motility. Optics<br />
Communications, 1971. 4(1): p. 35-39.<br />
3. Cerbino, R. and V. Trappe, Differential dynamic microscopy:<br />
Probing wave vector dependent dynamics with a microscope.<br />
Physical Review Letters, 2008. 100(18).<br />
4. Wilson, L.G., et al., Differential Dynamic Microscopy <strong>of</strong> Bacterial<br />
Motility. Physical Review Letters, 2011. 106(1).<br />
P11<br />
Exciton Migration in Conjugated<br />
Polymer Dots<br />
Scott N. Clafton 1 , Dr David M. Huang2 , Ming<br />
Chiu3 and Dr Tak W. Kee4 1 <strong>University</strong> <strong>of</strong> Adelaide, North Terrace Adelaide, South Australia,<br />
5005, scott.clafton@adelaide.edu.au<br />
2 <strong>University</strong> <strong>of</strong> Adelaide, North Terrace Adelaide, South Australia,<br />
5005, david.huang@adelaide.edu.au<br />
3 <strong>University</strong> <strong>of</strong> Adelaide, North Terrace Adelaide, South Australia,<br />
5005, ming.chiu@adelaide.edu.au<br />
4 <strong>University</strong> <strong>of</strong> Adelaide, North Terrace Adelaide, South Australia,<br />
5005, tak.kee@adelaide.edu.au<br />
Abstract<br />
Energy transfer dynamics <strong>of</strong> the conjugated<br />
polymer poly[2-methoxy-5-(2-ethylhexyloxy)-1,4phenylene-vinylene]<br />
(MEH-PPV) in two different<br />
solvent environments have been investigated using<br />
femtosecond fluorescence upconversion<br />
spectroscopy. The two environments are that <strong>of</strong> a<br />
reasonably good solvent (tetrahydr<strong>of</strong>uran) in which<br />
MEH-PPV adopts a partially extended<br />
conformation and a very poor solvent (water),<br />
which drives the polymer chains into spherical and<br />
highly compact nanoparticles (CPDots). Isotropic<br />
fluorescence kinetic data show energy transfer<br />
behaviour in both solvent environments that is<br />
109
Poster Presentations<br />
Sunday 4 December - Session 1<br />
consistent with previous studies <strong>of</strong> conjugated<br />
polymers. Time resolved fluorescence anisotropy<br />
decay has previously been used to show that<br />
energy transfer in partially extended polymer<br />
conformations occurs both through space and<br />
along the polymer backbone with approximate<br />
time constants <strong>of</strong> 1.4 ps and 110 ps, respectively.<br />
By comparing this result to the anisotropy decay <strong>of</strong><br />
CPDots it is clear that energy transfer in this<br />
system occurs exclusively in a through space<br />
manner. This is consistent with the hypothesis that<br />
the compact structures <strong>of</strong> CPDots provide many<br />
chromophores capable <strong>of</strong> participating in energy<br />
transfer in close proximity to the excited<br />
chromophore.<br />
P12<br />
Species differences in the kinetics <strong>of</strong><br />
the Na+,K+-ATPase<br />
Ronald J. Clarke and Flemming Cornelius<br />
School <strong>of</strong> Chemistry,<strong>University</strong> <strong>of</strong> Sydney, Sydney, NSW 2001,<br />
Australia, and Department <strong>of</strong> Physiology and Biophysics,<br />
<strong>University</strong> <strong>of</strong> Aarhus, Aarhus, Denmark<br />
Correspondence.: r.clarke@chem.usyd.edu.au<br />
Abstract<br />
Crystal structures <strong>of</strong> the Na+,K+-ATPase from two<br />
animal species (pig1 and shark2) have now been<br />
resolved. The 3D structures are very similar and<br />
the amino acid sequences <strong>of</strong> the alpha subunits <strong>of</strong><br />
the two enzymes display ~90% homology.<br />
However, in spite <strong>of</strong> the similar structures,<br />
stopped-flow investigations <strong>of</strong> the partial reactions<br />
<strong>of</strong> the same preparations used for structure<br />
determination have shown that the pig and shark<br />
enzymes display significant differences in their<br />
kinetics3,4. In the case <strong>of</strong> the pig enzyme, the<br />
major rate-determining step <strong>of</strong> the reaction cycle<br />
under saturating substrate conditions is the E2 →<br />
E1 conformational transition associated with the<br />
deocclusion <strong>of</strong> K+ ions and their release to the<br />
cytoplasm. For the shark enzyme the major<br />
rate-determining step appears to be the occlusion<br />
<strong>of</strong> K+ ions from the extracellular medium by the<br />
phosphorylated enzyme, E2P. These differences in<br />
rate-determining step could have major<br />
consequences for the physiology <strong>of</strong> the animals<br />
concerned. For example, although both enzymes<br />
110<br />
have similar turnovers when all substrates are at<br />
saturating concentrations, under physiological<br />
conditions the turnovers could be quite different.<br />
Furthermore, based on the different ratedetermining<br />
steps, one would expect the turnover<br />
<strong>of</strong> the shark enzyme to be much more sensitive to<br />
the extracellular K+ concentration than that <strong>of</strong> the<br />
pig. The apparent paradox <strong>of</strong> significantly different<br />
kinetics in spite <strong>of</strong> very similar structures leads one<br />
to ask what it is that really determines the kinetics<br />
<strong>of</strong> the Na+,K+-ATPase. Are subtle differences in<br />
the structure <strong>of</strong> the alpha-subunit sufficient to<br />
explain the different kinetics? Could the kinetic<br />
differences be due to differences in the structure<br />
<strong>of</strong> the beta-subunit or the gamma (or FXYD)<br />
subunit? Could their origin lie in different lipid<br />
compositions <strong>of</strong> the surrounding membrane or be<br />
associated with differences in protein-protein<br />
interactions (i.e. between alpha-beta protomers)<br />
within the membrane?<br />
1. Morth JP et al (2007) Nature 450: 1043-5<br />
2. Shinoda T et al (2009) Nature 459: 446-50<br />
3. Khalid et al (2010) Biophys J 98: 2290-8<br />
4. Myers et al (2011) Biophys J 100: 70-9<br />
P13<br />
Cardiac troponin: a paramagnetic<br />
relaxation enhancement NMR study<br />
Nicole M Cordina1 , C K Liew2 , D A Gell2 , J P<br />
Mackay2 , T M Logan3 , L J Brown1 1 Department <strong>of</strong> Chemistry and Biomolecular Sciences,<br />
Macquarie <strong>University</strong>, NSW 2109, Australia<br />
2 School <strong>of</strong> Molecular and Microbial Biosciences, <strong>University</strong> <strong>of</strong><br />
Sydney, NSW 2006, Australia<br />
3 Institute <strong>of</strong> Molecular Biophysics, Florida State <strong>University</strong>,<br />
Tallahassee, FL 32306, USA<br />
Abstract<br />
Cardiac Troponin is a trimeric thin filament protein<br />
complex that regulates muscle contraction.<br />
Calcium (Ca2+) binding to the Troponin C (TnC)<br />
subunit triggers the complex to undergo a large<br />
conformational switch from the OFF state, where<br />
acto-myosin interaction is sterically blocked, to the<br />
ON state, where acto-myosin interaction, and thus<br />
muscle contraction can occur. The 18 kDa<br />
Ca2+-binding TnC subunit, which constitutes the<br />
structural core <strong>of</strong> the troponin complex, has been<br />
well characterized, however much less is known
about the 24 kDa inhibitory subunit, TnI. The<br />
structure <strong>of</strong> troponin is <strong>of</strong> medical significance<br />
since several mutations, which occur in all<br />
subunits <strong>of</strong> the troponin complex, have been<br />
linked to hypertrophic cardiomyopathy (HCM). We<br />
have employed site-directed spin-labeling and<br />
paramagnetic relaxation enhancement (PRE) NMR<br />
spectroscopy to position key functional regions <strong>of</strong><br />
the TnI subunit with respect to the TnC subunit in<br />
the binary TnC-TnI complex. The positioning <strong>of</strong> TnI<br />
with respect to TnC was performed in both the<br />
absence and presence <strong>of</strong> Ca2+to understand the<br />
switch mechanism. Our strategy involves attaching<br />
a nitroxide spin label to single cysteine mutants <strong>of</strong><br />
14N-TnI and reconstituting the spin-labeled TnI<br />
with 15N-TnC. TROSY experiments were<br />
performed in the paramagnetic and diamagnetic<br />
states which enabled us to measure distances (10<br />
- 25 Å) between the TnI spin label and assignable<br />
TnC residues. We have spin labeled several key<br />
structural and functional regions <strong>of</strong> TnI, including<br />
the cardiac specific N-extension (TnI-28), the<br />
N-terminal region (TnI-57), the inhibitory region<br />
(TnI-137 and TnI-143), and the switch peptide<br />
(TnI-151 and TnI-159). The two switch peptide<br />
probe sites clearly show release <strong>of</strong> the switch<br />
peptide from the hydrophobic pocket in the TnC<br />
N-domain in the absence <strong>of</strong> Ca2+. The effect <strong>of</strong><br />
TnI phosphorylation on the conformation <strong>of</strong> the<br />
binary troponin complex was also examined.<br />
Phosphorylation <strong>of</strong> the TnI N-extension (S23, S24)<br />
clearly perturbs interactions between the TnC<br />
C-domain and the TnI N-extension. We have also<br />
performed experiments to investigate the effect <strong>of</strong><br />
a recently identified TnC HCM mutation, A8V<br />
(Landstrom et al, 2008, J Mol Cell Cardiol).<br />
Structural consequences <strong>of</strong> this mutation were<br />
observed in isolated TnC.<br />
Poster Presentations<br />
Sunday 4 December - Session 1<br />
P14<br />
Unimolecular Reaction Chemistry <strong>of</strong><br />
Atmospheric Peroxyl Radicals<br />
Gabriel da Silva<br />
Chemical and Biomolecular Engineering, The <strong>University</strong> <strong>of</strong><br />
Melbourne, Parkville 3010, Australia<br />
Abstract<br />
Volatile organic compounds (VOCs) and<br />
oxygenated VOCs (OVOCs) play a key role in<br />
determining the oxidative chemistry <strong>of</strong> the<br />
atmosphere, particularly in the planetary boundary<br />
layer in remote forested regions, where well-known<br />
HOx (OH + HO2) radical cycles are involving ozone<br />
and NOx are relatively inefficient. Numerous field<br />
studies conducted over the last decade have<br />
identified that OH radical levels are consistently<br />
elevated over model predictions in forested areas,<br />
particularly those with high levels <strong>of</strong> the biogenic<br />
VOC isoprene and low levels <strong>of</strong> NOx. Unimolecular<br />
decomposition reactions <strong>of</strong> peroxyl radicals<br />
formed in the OH-initiated oxidation <strong>of</strong> isoprene<br />
have been suggested as a mechanism for hydroxyl<br />
radical regeneration in these environments. Using<br />
computational chemistry and reaction rate<br />
modelling, rate constants have been estimated for<br />
isoprene peroxyl radical decomposition.<br />
Furthermore, the potential for OH radical recycling<br />
from the isoprene oxidation products<br />
methacrolein, methyl vinyl ketone, and glyoxal is<br />
also examined.<br />
111
Poster Presentations<br />
Sunday 4 December - Session 1<br />
P15<br />
Structural Analysis <strong>of</strong> Missense<br />
Mutations in the CLIC2 Chloride<br />
Intracellular Ion Channel Protein<br />
E L Daniel1 , J E Hare2 , S C Goodchild3 , N M<br />
Cordina4 , L J Brown5 1 Department <strong>of</strong> Chemistry and Biomolecular Science, Macquarie<br />
<strong>University</strong>, Sydney, New South Wales, 2109, elizabeth.daniel@<br />
students.mq.edu.au<br />
Abstract<br />
Chloride Intracellular Channels (CLIC proteins) are<br />
GST homologues that are part <strong>of</strong> a conserved<br />
protein family with dynamic properties. Members<br />
<strong>of</strong> the CLIC protein family have been shown to<br />
auto-insert into membranes, directly from the<br />
cytosol, to create active ion channels. Recently,<br />
five missense mutations (H101Q, S109C, P160A,<br />
D161H & D161Y) in the CLIC2 protein have been<br />
found in a large-scale next generation<br />
resequencing <strong>of</strong> X chromosome genes. One <strong>of</strong><br />
these mutations, H101Q, was linked to X-linked<br />
intellectual disability (XLID) with two male patients<br />
exhibiting traits and features <strong>of</strong> XLID including<br />
seizures, thumb position abnormality, large ears,<br />
and large testes. The other four mutations were<br />
also found in healthy individuals, suggesting that<br />
the CLIC2 protein is fully functional despite the<br />
mutations. An in silico modelling study proposed<br />
that the structural stability, inherent flexibility and<br />
membrane binding properties are affected by<br />
these mutations (Witham et al., 2011). In particular,<br />
the XLID linked H101Q mutation was suggested to<br />
be more stable in solution but to exhibit reduced<br />
plasticity necessary for binding and insertion into<br />
the membrane bilayer. To confirm these modelled<br />
observations, we will perform in vitro experiments<br />
for each <strong>of</strong> these mutations to determine the<br />
thermal stability and membrane binding properties<br />
<strong>of</strong> the CLIC2 proteins. We have cloned CLIC2 and<br />
introduced each <strong>of</strong> the five missense mutations.<br />
We will express and purify the proteins for analysis<br />
<strong>of</strong> their conformational stability by Circular<br />
Dichroism spectroscopy. Additionally, we will use a<br />
sucrose loaded vesicle sedimentation assay to<br />
determine if membrane-binding properties are<br />
impaired (Goodchild et al., 2009). The data will be<br />
discussed in the context <strong>of</strong> the predicted effects<br />
for each mutation.<br />
112<br />
References<br />
Witham et al (2011) Proteins: Structure, Function, and<br />
Bioinformatics 79: 2444-54.<br />
Goodchild et al (2009) European Biophysics Journal 39: 129-38.<br />
P16<br />
The Role <strong>of</strong> Spin in Triplet-Triplet<br />
Upconversion<br />
A. Danos1 , Y. Y. Cheng1 , D. R. McCamey2 , T. W.<br />
Schmidt1 1 School <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> Sydney, NSW 2006,<br />
Australia<br />
2 School <strong>of</strong> Physics, The <strong>University</strong> <strong>of</strong> Sydney, NSW 2006,<br />
Australia<br />
Abstract<br />
Triplet-Triplet Annihilation (TTA) Upconversion is a<br />
class <strong>of</strong> photochemical reactions that combine the<br />
energies <strong>of</strong> two or more photons into a single,<br />
higher energy photon. Sunlight with energy below<br />
the bandgap <strong>of</strong> a semiconductor photovoltaic<br />
device, which normally is not absorbed, is thus<br />
reclaimed.<br />
Upconversion occurs when two molecules absorb<br />
low energy photons and Inter-System Cross into<br />
triplet excited states. Conservation <strong>of</strong> the<br />
electrons’ spin allows for the molecules’ energies<br />
to disproportionate, producing two singlet states<br />
as a result. One <strong>of</strong> these is in the ground state, and<br />
the other in a highly excited state. Fluorescence<br />
from the excited singlet generates the upconverted<br />
photon, which can then power a photovoltaic<br />
device. Jablonski diagrams for these processes<br />
are shown in Figure 1.<br />
Figure 1: Jablonski diagrams <strong>of</strong> triplet formation (right), and TTA<br />
(left) [1].<br />
Although TTA upconversion has been<br />
demonstrated within the School <strong>of</strong> Chemistry and<br />
elsewhere, the spin mixing processes that result in<br />
annihilation remain poorly understood [2]
Furthermore, no theoretical treatment has yet<br />
accounted for the spin precession phase, nor<br />
satisfactorily explained the non-uniform effects <strong>of</strong><br />
magnetic alignment upon upconversion rates [3].<br />
Electron Paramagnetic Resonance (EPR) is an<br />
ideal tool for examining the role <strong>of</strong> triplet spin in<br />
TTA upconversion [4]. Our experiments involve<br />
using microwaves to excite molecules between the<br />
three degenerate triplet states, which correspond<br />
to the three possible spin orientations. By altering<br />
the populations <strong>of</strong> the triplet states and observing<br />
the changes in upconversion intensity, we<br />
investigate: the natural occupancy <strong>of</strong> the three spin<br />
states following intersystem crossing, which <strong>of</strong> the<br />
nine triplet pair configurations contribute to<br />
upconversion, and which <strong>of</strong> these pair states<br />
quench the reserve <strong>of</strong> excited molecules.<br />
Key Words<br />
Triplet, spin, upconversion, EPR, ESR, photovoltaics<br />
1. Fückel, B., et al., Singlet Oxygen Mediated Photochemical<br />
Upconversion <strong>of</strong> NIR Light. The Journal <strong>of</strong> Physical Chemistry<br />
Letters, 2011. 2(9): p. 966-971.<br />
2. Cheng, Y.Y., et al., Kinetic Analysis <strong>of</strong> Photochemical<br />
Upconversion by Triplet−Triplet Annihilation: Beyond Any Spin<br />
Statistical Limit. The Journal <strong>of</strong> Physical Chemistry Letters,<br />
2010. 1(12): p. 1795-1799.<br />
3. Mezyk, J., et al., Effect <strong>of</strong> an External Magnetic Field on the<br />
Up-Conversion Photoluminescence <strong>of</strong> Organic Films: The Role<br />
<strong>of</strong> Disorder in Triplet-Triplet Annihilation. Physical Review<br />
Letters, 2009. 102(8): p. 087404.<br />
4. Atherton, N.M., et al., Electron spin resonance. Vol. 14, A Review<br />
<strong>of</strong> recent literature to 19931994, Cambridge: Royal Society <strong>of</strong><br />
Chemistry.<br />
P18<br />
Bonding between the uracil<br />
monomers <strong>of</strong> the cyclobutane<br />
pyrimidine dimer radical anion by<br />
means <strong>of</strong> quantum chemical<br />
calculations<br />
Linda Feketeová 2 , Andreas<br />
Mauracher1 ,David Gschliesser 1 , <strong>Peter</strong> Bartl1 ,<br />
Violaine Vizcaino1 , Lukas An der Lan1 , Catrin<br />
Goeschen2 , Stephan Denifl1 , Richard A.J. O’Hair2 ,<br />
T. D. Märk1 , P. Scheier1 , U. Wille2 1 Institut für Ionenphysik und Angewandte Physik, Leopold-<br />
Franzens-Universität, Technikerstr. 25, Innsbruck, 6020, Austria,<br />
andreas.mauracher@uibk.ac.at<br />
2 ARC Centre <strong>of</strong> Excellence for Free Radical Chemistry and<br />
Biotechnology, School <strong>of</strong> Chemistry and Bio21 Institute <strong>of</strong><br />
Molecular Science and Biotechnology, The <strong>University</strong> <strong>of</strong><br />
Poster Presentations<br />
Sunday 4 December - Session 1<br />
Melbourne, 30 Flemington Road, Victoria, 3010, Australia, lfe@<br />
unimelb.edu.au<br />
Abstract<br />
The major lesion in DNA caused by exposure to<br />
ultraviolet radiation is formation <strong>of</strong> cyclobutane<br />
pyrimidine dimers (CPD), namely cis,syn-thyminethymine<br />
cyclobutane dimers (c,s-TT), resulting<br />
from [2π+2π] cycloaddition <strong>of</strong> two adjacent bases<br />
in the same oligonucleotide strand.1 This lesion<br />
has serious biological consequences, such as<br />
incorrect DNA replication, which could lead to<br />
mutation, cancer or cell death.2 Whereas<br />
placental mammals, including humans, remove<br />
CPD lesions through nucleotide excision repair, in<br />
prokaryotes, plants, and a variety <strong>of</strong> animals<br />
dimerized pyrimidines are efficiently converted to<br />
their monomeric form by the enzyme DNA<br />
photolyase in a light-driven catalytic reductive<br />
electron transfer cycle.3<br />
A recent study4 revealed that the attachment <strong>of</strong> a<br />
free electron to the cyclobutane pyrimidine dimers,<br />
c,s-DMTDMT and c,a-DMTDMT, leads to the<br />
formation <strong>of</strong> dimer radical anions, very likely<br />
through dipole bound states.5 These radical<br />
anions exhibit a lifetime <strong>of</strong> at least 80 μs, showing<br />
that they are much more stable than previously<br />
believed and that the splitting <strong>of</strong> the CPD radical<br />
anion into the respective pyrimidine monomers is<br />
associated with an activation barrier. This study<br />
was extended to investigate the attachment <strong>of</strong> free<br />
electrons to uracil cyclobutane pyrimidine dimers<br />
(DMUDMU) with different geometry at the<br />
cyclobutane ring. Along with these experiments,<br />
we undertake theoretical approach to understand,<br />
how the stereochemistry at the cyclobutane ring<br />
affects the stability <strong>of</strong> the CPD radical anion, and<br />
its dissociation. By means <strong>of</strong> quantum chemical<br />
calculations we investigate the bonding between<br />
the dimers <strong>of</strong> c,s-DMUDMU as well as<br />
t,s-DMUDMU. We compare the results <strong>of</strong><br />
density functional theory to wave function based<br />
approaches and post Hartree-Fock calculations.<br />
In addition we look at the potential energy surface<br />
for an approximate dissociation process <strong>of</strong> the<br />
dimers.<br />
[1] J. Cadet and P. Vigny, in Bioorganic Photochemistry:<br />
Photochemistry and the Nucleic Acids, ed. H. Morrison, John<br />
Wiley, New York, pp. 1–272 (1990)<br />
[2] A. Dussy, E. Meggers and B. Giese, J. Am. Chem. Soc. 120<br />
113
Poster Presentations<br />
Sunday 4 December - Session 1<br />
7399 (1998)<br />
[3] A. Sancar, Chem. Rev. 103 2203 (2003)<br />
[4] A. Edtbauer, K. Russell, L. Feketeová, J. Taubitz, C.<br />
Mitterdorfer, S. Denifl, R. A. J. O’Hair, T. D. Märk, P. Scheier and<br />
U. Wille, Chem. Commun. 7291 (2009).<br />
[5] A. Edtbauer, S. Denifl, V. Vizcaino, L. An der Lan, K. Russell, J.<br />
Taubitz, U. Wille, L. Feketeová, R. A. J. O’Hair, T. D. Märk, E.<br />
Illenberger and P. Scheier, Chem. Phys. Chem. 11 561 (2009)<br />
P19<br />
Structure and binding energies <strong>of</strong><br />
the doubly charged zwitterionic<br />
betaine clusters<br />
Linda Feketeová 2 , Emilie Cauët1 , William A.<br />
Donald2 , Richard A.J. O’Hair2 ,<br />
1 Service de Chimie Quantique et Photophysique, Université Libre<br />
de Bruxelles, avenue F.D. Roosevelt 50 CPi 160/09, Brussels,<br />
1050, Belgium, ecauet@ulb.ac.be<br />
2 ARC Centre <strong>of</strong> Excellence for Free Radical Chemistry and<br />
Biotechnology, School <strong>of</strong> Chemistry and Bio21 Institute <strong>of</strong><br />
Molecular Science and Biotechnology, The <strong>University</strong> <strong>of</strong><br />
Melbourne, 30 Flemington Road, Victoria, 3010, Australia, lfe@<br />
unimelb.edu.au<br />
Abstract<br />
Ionic clusters are important species in a wide<br />
variety <strong>of</strong> areas, none more obvious than biological<br />
processes. The advent <strong>of</strong> electrospray ionisation<br />
(ESI) has provided a new method for generating a<br />
wide range <strong>of</strong> cluster ions, including ionic clusters<br />
and biomolecular clusters. One <strong>of</strong> the fundamental<br />
differences between amino acids in solution and in<br />
the gas phase is the fact that they exist<br />
predominantly in the zwitterionic form<br />
(H3N+CH2CO2-) in solution but adopt the<br />
nonzwitterionic form (H2NCH2CO2H) in the<br />
gas-phase. Betaines (e.g. Glycine Betaine (GB) 1,<br />
Dimethylsulfonioacetate (DMSA) 2) are particularly<br />
interesting as a model <strong>of</strong> the fundamental gas<br />
phase chemistry <strong>of</strong> the zwitterionic form <strong>of</strong> amino<br />
acids, due to the presence <strong>of</strong> their permanent<br />
fixed charges.<br />
114<br />
Since the discovery that the ESI <strong>of</strong> solution <strong>of</strong><br />
zwitterionic GB (1) leads to the formation <strong>of</strong> a<br />
number <strong>of</strong> multiply protonated clusters [Mn+mH]<br />
m+, where m = 1,...,4, with relatively high<br />
abundance,1 we have studied the collisioninduced<br />
dissociation (CID) and electron-induced<br />
dissociation (EID) reactions <strong>of</strong> these clusters in<br />
detail for one <strong>of</strong> the larger size [M21+2H]2+<br />
clusters <strong>of</strong> GB,2 as well as clusters [M15+2H]2+ <strong>of</strong><br />
zwitterionic GB, and DMSA, that are close to the<br />
stability limit, i.e. Coulomb repulsion <strong>of</strong> the charge<br />
within the cluster competes with attractive forces<br />
such as hydrogen bonding and charge-dipole<br />
interactions.3 Multiply protonated clusters<br />
fragment via competitive neutral loss and charge<br />
separation.<br />
Density functional theory (DFT) calculations on the<br />
doubly charged dimer suggest that the doubly<br />
charged clusters consist <strong>of</strong> a [M2+2H]2+ core<br />
based on the hydrogen-bonded carboxylic acid<br />
dimer.1 In the present study we investigate the<br />
structure and stability <strong>of</strong> the larger clusters using<br />
DFT calculations in order to gain additional insight.<br />
[1] Feketeová, L.; O’Hair, R. A. J. Chem. Commun. 2008,<br />
4942-4944.<br />
[2] Feketeová, L.; O’Hair, R. A. J. Rapid Commun. Mass Spectrom.<br />
2009, 23, 3259-3263.<br />
[3] Yoo, E. J.-H.; Feketeová, L.; Khairallah, G. N.; O’Hair, R. A. J. J.<br />
Phys. Chem. A. 2011, 115, 4179-4185.<br />
P20<br />
Parametric Sensitivity Analyses <strong>of</strong><br />
the Insulin Signalling Pathway<br />
Catheryn Gray 1 , Adelle C. F. Coster 1,2<br />
1 School <strong>of</strong> Mathematics & Statistics, <strong>University</strong> <strong>of</strong> New South<br />
Wales<br />
2 Garvan Institute <strong>of</strong> Medical Research, Darlinghurst NSW<br />
Abstract<br />
There is no comprehensive mathematical model <strong>of</strong><br />
the complex biological dynamics <strong>of</strong> insulin<br />
signalling. The identification <strong>of</strong> the main nodes <strong>of</strong><br />
activity and the underlying mechanisms<br />
connecting signal transduction to physical<br />
translocation <strong>of</strong> glucose transporters is a<br />
significant biological outcome as the metabolic<br />
effects <strong>of</strong> insulin on glucose uptake are<br />
fundamental to life. Given the ubiquitous nature <strong>of</strong>
this feedback and control system, quantitative<br />
modelling is <strong>of</strong> widespread scientific interest. In the<br />
longer term, understanding how this dynamic<br />
system operates under normal conditions will be<br />
vital to understanding its operation under<br />
abnormal conditions.<br />
Here we analyse the behaviour <strong>of</strong> an initial model<br />
for the insulin signalling pathway, and identify the<br />
key nodes controlling the regulation <strong>of</strong> the glucose<br />
transporter protein GLUT4 at the cell surface. It is<br />
known that this model has limitations and the<br />
ramifications <strong>of</strong> these are explored, with a view to<br />
modifying this to create a comprehensive,<br />
validated model <strong>of</strong> insulin-stimulated metabolic<br />
responses for fibroblasts, adipocytes and muscle<br />
cells.<br />
As in the cases <strong>of</strong> coupled mitogen-activated<br />
protein kinase and phosphoinositide 3-kinase<br />
(MAPK–PI3K) pathways related to cancers, we<br />
make use <strong>of</strong> integrated and other sensitivity<br />
measures to rank the key nodes in the insulin<br />
network. The sensitivity <strong>of</strong> membrane GLUT4 with<br />
respect to all parameters (rate constants and<br />
non-zero initial concentrations) is explored.<br />
P21<br />
Energy and Charge Transfer<br />
Interactions in a Mixed Porphyrin<br />
Co-sensitized TiO2 Electrode: A<br />
sub-ns Transient Absorption<br />
Spectroscopy Study<br />
M. J. Griffith, 1 A. J. Mozer, 1 P.Wagner, 1 G. G.<br />
Wallace, 1 D. L. Officer, 1 and R. Katoh 2,3<br />
1. ARC Centre <strong>of</strong> Excellence for Electromaterials Science and<br />
Intelligent Polymer Research Institute, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>,<br />
Innovation Campus, Squires Way, North <strong>Wollongong</strong>, NSW,<br />
2500, Australia.<br />
Emails: mjg48@uow.edu, attila@uow.edu.au, pawel@uow.edu.au,<br />
gwallace@uow.edu.au, davido@uowmail.edu.au<br />
2. National Institute <strong>of</strong> Advanced Industrial Science and<br />
Technology, Tsukuba Central 5, Tsukuba, Ibaraki, 305-8565,<br />
Japan.<br />
3. Department <strong>of</strong> Chemical Biology and Applied Chemistry,<br />
College <strong>of</strong> Engineering, Nihon <strong>University</strong>, Tamura, Koriyama,<br />
Fukushima, 963-8642, Japan.<br />
Email: rkatoh@chem.ce.nihon-u.ac.jp<br />
Abstract<br />
Poster Presentations<br />
Sunday 4 December - Session 1<br />
Sensitisation <strong>of</strong> TiO2 with a wide variety <strong>of</strong> organic<br />
and inorganic dyes has been extensively studied in<br />
an attempt to synthesise highly efficient<br />
photovoltaic devices. Given their efficacy in<br />
photosynthesis, porphyrin molecules <strong>of</strong>fer<br />
immense potential in this regard.[1] However, there<br />
have been very few reports <strong>of</strong> artificial porphyrin<br />
devices which reproduce the constructive<br />
intermolecular energy or charge transfer<br />
processes so prevalent in photosynthetic systems.<br />
[2]<br />
In this study a mixed dye system with a potentially<br />
high quantum yield for energy and charge transfer<br />
has been created using two porphyrin<br />
components; one a zinc porphyrin and the other a<br />
free base porphyrin (Figure 1). The mixed system<br />
shows superior solar cell performance when<br />
compared to either <strong>of</strong> the individual<br />
components,[3] a phenomenon which is<br />
remarkable considering the similarity in the<br />
absorption spectra <strong>of</strong> the two dyes.<br />
Fig. 1 Chemical structures <strong>of</strong> porphyrin dyes employed in this<br />
study.<br />
The current study employs transient absorption<br />
spectroscopy with 500 ps resolution to analyse<br />
energy and charge transfer mechanisms by<br />
monitoring formation <strong>of</strong> the dye cations formed<br />
after excitation in the mixed dye system. Results<br />
indicate that the electron injection yield<br />
(proportional to the dye cation concentration) is<br />
only amplified with respect to the individual dyes at<br />
high dye surface coverages and small<br />
intermolecular spacings (Fig 2a). This indicates<br />
Forster type energy transfer is responsible for the<br />
amplified injection yield. Furthermore, the free<br />
base cation signal disappears on very fast time<br />
scales in the mixture (Fig 2b), indicative <strong>of</strong> a charge<br />
transfer mechanism which limits recombination.<br />
115
Poster Presentations<br />
Sunday 4 December - Session 1<br />
Fig. 2 (a) Kinetics <strong>of</strong> dye cation creation in the mixed dye system<br />
for different dye surface coverages. (b) Transient absorption<br />
spectra <strong>of</strong> the mixed dye system at various time delays after<br />
excitation<br />
Keywords<br />
Porphyrin, energy and charge transfer, sub-ns transient absorption<br />
References<br />
[1]. W.M. <strong>Campbell</strong>, A.K. Burrell, D.L. Officer, K.W Jolley; Coord.<br />
Chem. Rev. 2004, 248, 1363.<br />
[2]. A.J. Mozer, M.J. Griffith, G. Tsekouras, P. Wagner, G.G.<br />
Wallace, S. Mori, K. Sunahara, M. Miyashita, J.C. Earles, K.C.<br />
Gordon, L. Du, R. Katoh, A. Furube, D.L. Officer; J. Am. Chem.<br />
Soc., 2009, 131, 15621.<br />
[3]. M.J. Griffith, A.J. Mozer, G. Tsekouras, Y. Dong, P. Wagner, K.<br />
Wagner, G.G. Wallace, S. Mori, D.L. Officer; App. Phys. Lett.,<br />
2011, 98, 163502.<br />
116<br />
P22<br />
ACE I/D genotypes and their impact<br />
on heart rate variability: a limited<br />
meta-analysis<br />
Brett Hambly6 , Ethan Ng1 , Yaxin Lu 2 , Slade<br />
Matthews3 , Herbert Jelinek4 , Craig McLachlan 5<br />
1 Pathology, Univ <strong>of</strong> Sydney, NSW 2006, etng1324@uni.sydney.<br />
edu.au<br />
2 Pathology, Univ <strong>of</strong> Sydney, NSW 2006, yalu4496@uni.sydney.<br />
edu.au<br />
3 Pharmacology, Univ <strong>of</strong> Sydney, NSW 2006, sladem@med.usyd.<br />
edu.au<br />
4 School <strong>of</strong> Community Health, Charles Sturt Univ, NSW 2640,<br />
HJelinek@csu.edu.au<br />
5 Rural Clinical School, Univ <strong>of</strong> NSW, NSW, 2650, reperfusion@<br />
hotmail.com<br />
6 Pathology, Univ <strong>of</strong> Sydney, NSW 2006, bretth@pathology.usyd.<br />
edu.au<br />
Abstract<br />
Heart rate variability (HRV) is a measure <strong>of</strong><br />
fluctuations in response to different stressors and<br />
environmental changes. High HRV indicates the<br />
ability to adapt to situations, while lower HRV<br />
implies dysfunction and therefore a less healthy<br />
individual. Genetic factors are capable <strong>of</strong><br />
substantial influence over HRV. A potential locus<br />
for this is the angiotensin converting enzyme<br />
(ACE), where polymorphisms may have an effect<br />
on HRV. The ACE Insertion/Deletion (I/D)<br />
genotypes are thought to influence HRV. The ACE<br />
I/D polymorphism is the insertion <strong>of</strong> 287 bases into<br />
intron 16 <strong>of</strong> the ACE gene. The D allele is<br />
associated with higher levels <strong>of</strong> circulating ACE<br />
and also with increased cardiovascular risk in a<br />
number <strong>of</strong> studies <strong>of</strong> CVS disease states. Studies<br />
investigating an association between HRV and<br />
ACE I/D have been conducted in various normal<br />
and diseased patient groups. These have<br />
exhibited a lack <strong>of</strong> standardization in both<br />
measurement and analysis <strong>of</strong> HRV, resulting in<br />
apparently contradictory results. Consequently,<br />
there remains much controversy surrounding the<br />
ACE I/D polymorphism. We aimed to consolidate<br />
and analyse these data using a meta-analysis. We<br />
have reviewed published data to clarify some <strong>of</strong><br />
the uncertainty<br />
regarding how ACE I/D polymorphisms affect HRV<br />
and autonomic activity.
Secondly, this study has examined whether the<br />
relevant genetic cohort studies are guided by the<br />
HRV task force recommendations. Our initial<br />
analysis shows that the ACE I/D polymorphism<br />
correlates with HRV.<br />
P23<br />
UV-Vis Action Spectroscopy <strong>of</strong> Room<br />
Temperature Protonated Aromatics<br />
Christopher S. Hansen1 , Ben B. Kirk1 ,<br />
Richard A.J. O’hair 2 , Stephen J. Blanksby1, Adam<br />
J. Trevitt 1<br />
1 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>, NSW 2522<br />
Australia<br />
2 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Melbourne, VIC 3010<br />
Australia<br />
Abstract<br />
We have developed an instrumental setup for<br />
investigating the UV-Vis photodissociation<br />
efficiency <strong>of</strong> ions bringing together a mid-band<br />
tunable OPO laser (220 nm – 2.5 μm) and a<br />
modified, commercial stretched linear quadrupole<br />
ion trap. Here we report preliminary<br />
photodissociation studies <strong>of</strong> various protonated<br />
aromatic compounds.<br />
Briefly, ions are generated using electrospray<br />
ionisation and focused into the linear ion trap<br />
where the ion <strong>of</strong> interest is isolated. This trapped<br />
ion population is then irradiated with a single OPO<br />
laser pulse and scanned out <strong>of</strong> the trap for mass<br />
spectrometric analysis. This allows us to examine<br />
multiple fragmentation channels and infer their<br />
relative photodissociation in a single experiment.<br />
Our preliminary studies have focused on<br />
protonated aromatic compounds, a significant<br />
number <strong>of</strong> important compounds are aromatic or<br />
contain aromatic units. An example <strong>of</strong> a<br />
compound we have studied is the meta and para<br />
isomers <strong>of</strong> aminophenol. Aminophenol exhibits<br />
many fragmentation channels, mainly amine<br />
hydrogen loss, alcohol hydrogen loss, ammonia<br />
(NH3) loss and OH loss. This makes it a good<br />
model compound for studying larger molecules<br />
containing aromatic moieties with amine or alcohol<br />
functionalisations.<br />
Poster Presentations<br />
Sunday 4 December - Session 1<br />
Although the direct photoexcitation cross-sections<br />
are typically weak, upon the population <strong>of</strong> a σ*<br />
orbital aromatic moieties exhibit interesting<br />
phot<strong>of</strong>ragmentation dynamics that are <strong>of</strong>ten<br />
masked by optically bright π*π transitions. Insight<br />
into these σ* excited states is necessary for<br />
developing a thorough understanding <strong>of</strong> a<br />
system’s electronic excited states and overall<br />
photochemical behaviour. We present an effective<br />
method for exploring such excited states and<br />
compare our results to excited state calculations.<br />
In this poster the experimental setup will be<br />
explained in further detail and action spectra <strong>of</strong><br />
various protonated aromatics will be presented<br />
and discussed.<br />
P24<br />
Stabilisation and Delivery <strong>of</strong><br />
Medicinal Pigment Curcumin by<br />
g-Cyclodextrin Dimers<br />
Takaaki Harada1 , Duc-Truc Pham1 , Huy Tien<br />
Ngo1 , Tiffany Harris2 , Eleanor Need2 , Grant<br />
Buchanan2 ,Mandy Leung1 , Stephen F. Lincoln1 ,<br />
Tak W. Kee1 1 School <strong>of</strong> Chemistry and Physics, The <strong>University</strong> <strong>of</strong> Adelaide,<br />
North Terrace Campus, Adelaide, SA, 5005, Australia, takaaki.<br />
harada@adelaide.edu.au<br />
2 Molecular Ageing Laboratory, School <strong>of</strong> Medicine, The<br />
<strong>University</strong> <strong>of</strong> Adelaide, Basil Hetzel Institute, The Queen<br />
Elizabeth Hospital, 28 Woodville Road, Woodville South, SA,<br />
5011, Australia<br />
Abstract<br />
Curcumin is a polyphenol extracted from turmeric<br />
which is known not only as a curry spice but also<br />
as herbal medicine in India and other Asian<br />
117
Poster Presentations<br />
Sunday 4 December - Session 1<br />
countries. This yellow molecule possesses variety<br />
<strong>of</strong> therapeutic effects such as anti-cancer,<br />
antioxidant and wound healing activities. However,<br />
the poor aqueous solubility and stability <strong>of</strong><br />
curcumin limits its availability in biologically relevant<br />
environment.<br />
It is, therefore, important to encapsulate curcumin<br />
with suitable delivery agents such as<br />
g-cyclodextrin (g-CD). We have demonstrated that<br />
this molecular self-assembly stabilises curcumin<br />
moderately and it may potentially improve the<br />
bioavailability <strong>of</strong> curcumin. In this poster, we<br />
present the stabilisation and molecular delivery <strong>of</strong><br />
curcumin by diamide linked g-CD dimers, which<br />
consist <strong>of</strong> two g-CDs that are connected with<br />
either a succinamide or urea linker. They stabilize<br />
curcumin highly effectively by strong cooperative<br />
binding to form a 1:1 complex with curcumin under<br />
physiological conditions. The half-life <strong>of</strong> curcumin<br />
is extended by 180-780 times. In addition, we<br />
demonstrate that curcumin delivered by both the<br />
diamide linked g-CD dimers exhibits antiproliferative<br />
effect on a metastatic prostate cancer<br />
cell line (PC-3). Therefore, the diamide linked g-CD<br />
dimers have great potential for delivery <strong>of</strong><br />
curcumin in cancer therapy.<br />
P25<br />
Effective Core Potential<br />
Benchmarking for Cerium Oxide<br />
Clusters Using Density Functional<br />
Theory<br />
Robert A. Hardy1 , Birte Reichers2 and<br />
Gregory F. Metha3 1 Department <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> Adelaide, South<br />
Australia 5005, robert.hardy@adelaide.edu.au<br />
2 Department <strong>of</strong> Chemistry, Bielefeld <strong>University</strong>, 33615 Bielefeld,<br />
Germany, birte.reichers@uni-bielefeld.de<br />
3 Department <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> Adelaide, South<br />
Australia 5005, gregory.metha@adelaide.edu.au<br />
Abstract<br />
Benchmark Density Functional Theory calculations<br />
were performed on small cerium oxide clusters<br />
(CemOn, m=2,3; n=0,1,…,m) to determine an<br />
effective core potential (ECP) suitable for<br />
calculations <strong>of</strong> cluster geometries and ionisation<br />
118<br />
energies. The ECPs selected for testing included<br />
the 28 electron core Stuttgart Relativistic Small<br />
Core (SRSC) with a valence shell configuration <strong>of</strong><br />
6s25d14f1, and the 47 electron core (4fn/Q11) and<br />
48 electron core (4fn/Q10) MWB ECPs with<br />
valence shell configurations <strong>of</strong> 5s25p66s25d1 and<br />
5s25p66s2, respectively.<br />
Our calculations show the 4fn/Q11 ECP performs<br />
well with regard to providing similar ionisation<br />
energies and cluster geometries to those<br />
calculated using the well established SRSC<br />
ECP1,2. This result, in consideration with the<br />
greatly reduced computational expense<br />
associated with the 4fn/Q11 ECP compared to the<br />
SRSC ECP, makes the 4fn/Q11 ECP appropriate<br />
for future calculations <strong>of</strong> our larger cluster systems.<br />
The 4fn/Q10 ECP shows a poor comparison <strong>of</strong><br />
cluster geometries and ionisation energies to<br />
those calculated using the SRSC ECP, particularly<br />
in the case <strong>of</strong> the Cem bare metal clusters where<br />
Ce-Ce bond lengths were calculated to be in<br />
excess <strong>of</strong> 4 Å. The 4fn/Q10 ECP is therefore not<br />
considered suitable for future calculations <strong>of</strong><br />
CemOn clusters. The comparison <strong>of</strong> the Ce<br />
valence shell configurations for each <strong>of</strong> the ECPs<br />
suggests that the Ce 4f electron is not imperative<br />
in the formation <strong>of</strong> chemical bonds, whereas the<br />
single 5d electron is crucial to bonding.<br />
This poster will present comparisons <strong>of</strong> cluster<br />
geometries and ionisation energies for structures<br />
optimised with the SRSC, 4fn/Q11 and 4fn/Q10<br />
ECPs. These results will be supported with a<br />
comparison <strong>of</strong> vibrational frequencies calculated<br />
for CeO and Ce2 dimers with published<br />
experimental results3 to show the effectiveness <strong>of</strong><br />
the ECPs to model Ce-Ce and Ce-O bonding.<br />
(1) Dolg, M.; Stoll, H.; Preuss, H. J. Chem. Phys. 1988, 90.<br />
(2) Wu, X.-N.; Ding, X.-L.; Bai, S.-M.; Xu, B.; He, S.-G.; Shi, Q. The<br />
Journal <strong>of</strong> Physical Chemistry C 2011, 115, 13329.<br />
(3) Shen, X.; Fang, L.; Chen, X.; Lombardi, J. R. J. Chem. Phys.<br />
2000, 113.
P26<br />
Probing the Integral Membrane<br />
Form <strong>of</strong> Clic1 Using Fluorescence<br />
Resonance Energy Transfer<br />
Joanna E Hare1 , Sophia C Goodchild2,<br />
Louise J Brown3 1 Department <strong>of</strong> Chemistry and Biomolecular Science, Macquarie<br />
<strong>University</strong>, Sydney, New South Wales, 2109, joanna.hare@<br />
students.mq.edu.au<br />
2 Department <strong>of</strong> Chemistry and Biomolecular Science, Macquarie<br />
<strong>University</strong>, Sydney, New South Wales, 2109, sophia.goodchild@<br />
mq.edu.au<br />
3 Department <strong>of</strong> Chemistry and Biomolecular Science, Macquarie<br />
<strong>University</strong>, Sydney, New South Wales, 2109, louise.brown@mq.<br />
edu.au<br />
Abstract<br />
Chloride Intracellular Channel proteins (CLICs) are<br />
controversial because they can adopt two<br />
drastically different, yet stable conformations.<br />
CLICs are expressed either as soluble, globular<br />
proteins or integral membrane proteins.<br />
Electrophysiological studies have shown that the<br />
integral membrane form <strong>of</strong> the CLIC can function<br />
as an ion channel. The controversy is raised as the<br />
classic protein structure paradigm poses that the<br />
unique amino acid sequence gives a protein a<br />
single well defined three dimensional structure.<br />
However, it is becoming increasingly apparent that<br />
some proteins can adopt more than one stable<br />
conformation. These proteins are referred to as<br />
‘metamorphic’ proteins (Murzin, 2008).<br />
Crystal structures have been solved for the soluble<br />
form <strong>of</strong> several members <strong>of</strong> the CLIC family but<br />
attempts to solve the integral membrane form<br />
using traditional methods, such as NMR and<br />
crystallography, have been largely unsuccessful.<br />
Fluorescence Resonance Energy Transfer (FRET)<br />
was therefore used to probe the transition <strong>of</strong><br />
CLIC1 as it interacts with the membrane bilayer.<br />
FRET is a spectroscopic technique that is used to<br />
measure distances and changes in distances that<br />
may occur in a dynamic macromolecule. Two<br />
chemical probes are attached to the<br />
macromolecule(s) <strong>of</strong> interest. If the two probes are<br />
in close proximity and the fluorescence emission<br />
<strong>of</strong> the donor overlaps with the absorption pr<strong>of</strong>ile <strong>of</strong><br />
the acceptor, then energy can be transferred via a<br />
dipole-to-dipole interaction. The amount <strong>of</strong> energy<br />
Poster Presentations<br />
Sunday 4 December - Session 1<br />
transfer is proportional to the distance between<br />
the probes.<br />
FRET is especially useful for investigation <strong>of</strong> the<br />
CLIC structure as both the soluble form and the<br />
integral membrane form can be probed<br />
simultaneously under physiological conditions. In<br />
this study, FRET was used to provide the first<br />
structural evidence that CLIC1 interacts with the<br />
bilayer. Two sets <strong>of</strong> probe pairs were used so that<br />
a range <strong>of</strong> distances could be obtained. FRET was<br />
measured from CLIC1 to a probe attached to the<br />
head group <strong>of</strong> lipids incorporated into liposomes.<br />
A strong FRET signal was detected between<br />
CLIC1 and the bilayer. Environmental parameters<br />
shown to affect channel activity, including pH and<br />
redox, are now under investigation using the<br />
established FRET assay to understand whether<br />
they also promote membrane insertion <strong>of</strong> CLIC1.<br />
Murzin, A., ‘Metamorphic Proteins’, Science, 2008. 320, p.<br />
1725-1726<br />
P27<br />
Single Molecule Widefield<br />
Fluorescence Studies <strong>of</strong> Conjugated<br />
Polymers<br />
Emma N. Hooley1 , Toby D. M. Bell2 , Kenneth<br />
P. Ghiggino3 1 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Melbourne, Victoria 3010,<br />
e.hooley@student.unimelb.edu.au<br />
2 School <strong>of</strong> Chemistry, Monash <strong>University</strong>, Victoria, 3800, toby.<br />
bell@monash.edu<br />
3 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Melbourne, Victoria 3010,<br />
ghiggino@unimelb.edu.au<br />
Abstract<br />
Conjugated polymers are finding increasing<br />
application as materials for organic light emitting<br />
diodes, photovoltaic devices and lasers. However,<br />
polymers are inherently heterogeneous (e.g.<br />
molecular weight distribution, conformation) and<br />
understanding the light induced processes in<br />
these materials remains a challenge.<br />
Single molecule fluorescence spectroscopy (SMS)<br />
allows isolated polymer chains to be studied,<br />
removing the averaging inherent to bulk<br />
fluorescence measurements. Intrachain relaxation<br />
processes otherwise obscured can be identified<br />
119
Poster Presentations<br />
Sunday 4 December - Session 1<br />
and characterised. SMS has been previously<br />
applied to investigate the photophysics <strong>of</strong><br />
conjugated polymers and has yielded information<br />
on triplet state dynamics, photo-oxidation and<br />
charge-transfer mechanisms.<br />
Widefield fluorescence microscopy is emerging as<br />
a powerful single molecule spectroscopy<br />
technique, as it allows a wide area to be<br />
illuminated and the behaviour <strong>of</strong> the molecules to<br />
be observed in real time. In addition, defocused<br />
widefield images can provide information on the<br />
properties and dynamics <strong>of</strong> the emission transition<br />
dipole, thus allowing an insight into the molecular<br />
orientation and diffusion properties <strong>of</strong> the polymer<br />
chromophores.<br />
Figure 1: a) Alt-co-MEH-PPV; b) F8BT<br />
Derivatives <strong>of</strong> phenylene vinylene (PPV), especially<br />
2-methoxy-5-(2’-ethyl-hexaloxy)-1,4-phenylene<br />
vinylene (MEH-PPV) have been widely studied. We<br />
have synthesised and investigated an alternating<br />
co-polymer <strong>of</strong> PPV and MEH-PPV, (alt-co-MEH-<br />
PPV) (Figure 1 a) and poly(9,9’-dioctylfluorenecobenzothiadiazole)<br />
(F8BT) (Figure 1 b) and<br />
applied SMS and defocused widefield techniques<br />
to investigate the effects <strong>of</strong> structure on the<br />
photophysical processes occurring in single<br />
chains.<br />
Single chains <strong>of</strong> alt-co-MEH-PPV and F8BT exhibit<br />
fluorescence blinking on both short (submillisecond)<br />
and long (seconds) time scales and<br />
these results are interpreted in terms <strong>of</strong><br />
contributions by triplet state excursions, and<br />
charge and energy transport processes. Widefield<br />
fluorescence microscopy has provided additional<br />
information on the nature and dynamics <strong>of</strong> the<br />
emitting chromophores present in individual<br />
polymer chains. Although the polymers consist <strong>of</strong><br />
a distribution <strong>of</strong> absorbing chromophores, in most<br />
cases they behave as single fluorophore emitters.<br />
120<br />
P28<br />
Potential role <strong>of</strong> forbidden disulfide<br />
motifs in Zn fingers.<br />
Hulugalle D V K 1,3 , Haworth N L 2 , Ballouz S<br />
1,4 2 1,3 2,3<br />
, Jason Y. Liu , Fan S W , Wouters M A<br />
1 Structural and Computational Biology Program, Victor Chang<br />
Cardiac Research Institute, Sydney, Australia.<br />
2 School <strong>of</strong> Life and Environmental Sciences, Deakin <strong>University</strong>,<br />
Geelong, Australia<br />
3 School <strong>of</strong> Medical Sciences, <strong>University</strong> <strong>of</strong> New South Wales,<br />
Sydney, Australia<br />
4 School <strong>of</strong> Computer Science & Engineering, <strong>University</strong> <strong>of</strong> New<br />
South Wales, Sydney, Australia<br />
Abstract<br />
Expulsion <strong>of</strong> Zn2+ from proteins following oxidation<br />
<strong>of</strong> ligating Cysteine residues is an emerging area <strong>of</strong><br />
the oxidative stress response. During a recent data<br />
mining survey <strong>of</strong> protein structures with pairs <strong>of</strong><br />
thiols in both reduced and oxidized (disulfide<br />
bonded) states, we found two structural motifs<br />
repeatedly associated with Zn2+ binding (1).<br />
Forbidden disulfides are a canonical set <strong>of</strong><br />
disulfides with abnormal stereochemistry<br />
associated with redox-activity. Here we show<br />
through systematic analysis <strong>of</strong> Zinc finger<br />
structures and sequences, that one <strong>of</strong> these<br />
motifs is extremely prevalent in Zinc fingers. We<br />
show that in around 50% <strong>of</strong> Zinc finger structures<br />
two <strong>of</strong> the Zn2+-ligating thiols are embedded in a<br />
secondary structure similar to an anti-parallel<br />
β-diagonal disulfide-like motif (aBDD), located on<br />
the β-hairpin structure known as a Zinc knuckle.<br />
Formation <strong>of</strong> a disulfide by thiols <strong>of</strong> this motif has<br />
recently been characterized in the molecular<br />
chaperone Hsp33 and also demonstrated in<br />
several other transcription factors (2). Although<br />
other forbidden disulfide motifs are occasionally<br />
present in Zinc fingers, none are as ubiquitous as<br />
this aBDD-like motif. We show that the presence<br />
<strong>of</strong> this motif and its position in the structure is<br />
characteristic <strong>of</strong> different types <strong>of</strong> Zinc fingers,<br />
suggesting a functional relationship. As Zinc<br />
fingers comprise more than 17% <strong>of</strong> the human<br />
genome, this motif is likely important in Zn2+<br />
signalling.<br />
1. Fan SW, George RA, Haworth NL, Feng LL, Liu JY, Wouters<br />
MA. Conformational changes in redox pairs <strong>of</strong> protein<br />
structures. Prot Sci 18: 1745-1765, 2009.
2. Ilbert M, Horst J, Ahrens S, Winter J, Graf PCF, Lilie H, Jakob U.<br />
The redox-switch domain <strong>of</strong> Hsp33 functions as dual stress<br />
sensor. Nat Struct Mol Biol 14: 556-563, 2007.<br />
P29<br />
Cryopreservation – The Beginning <strong>of</strong><br />
a DSC (Differential Scanning<br />
Calorimetric) Understanding.<br />
Taavi Hunt 1 , Anja Kaczmarczyk 2, 3 , Bryn<br />
Funnekotter 2, 3, Shane Turner 3, 4 , Eric Bunn 3, 4 ,<br />
Gary Bryant1 , Ricardo L. Mancera2 1 Applied Physics, School <strong>of</strong> Applied Sciences, RMIT <strong>University</strong>,<br />
Melbourne, VIC, Australia.<br />
2 Curtin Health Innovation Research Institute, Western Australian<br />
Biomedical Research Institute, Curtin <strong>University</strong>, Perth WA,<br />
Australia.<br />
3 Botanic Gardens and Parks Authority, Fraser Avenue, West<br />
Perth WA , Australia.<br />
4 School <strong>of</strong> Plant Biology, Faculty <strong>of</strong> Natural and Agricultural<br />
Sciences, <strong>University</strong> <strong>of</strong> Western Australia, Crawley WA,<br />
Australia.<br />
Abstract<br />
A collaborative project between: RMIT <strong>University</strong>,<br />
Curtin <strong>University</strong>, Botanical Gardens and Parks<br />
Authority (WA) and Alcoa Australia. The major goal<br />
<strong>of</strong> this collaboration is to understand the major<br />
factors that determine the ability <strong>of</strong> various<br />
recalcitrant plant species to survive cryogenic<br />
storage, with particular relevance to post mining<br />
restoration <strong>of</strong> bauxite mining activities in South<br />
Western Australia. The Western Australian<br />
endemic species Loxocarya Cinerea is a key<br />
understory component <strong>of</strong> the Jarrah Forests <strong>of</strong><br />
South-West Western Australia, and hence is very<br />
important in post-mining restoration. However it<br />
rarely produces seeds and cannot be propagated<br />
conventionally, therefore is produced via tissue<br />
culture.<br />
Cryopreservation refers to the storage <strong>of</strong> living<br />
biological organisms at ultra low temperatures,<br />
(Liquid Nitrogen, -196 °C), in such a way that they<br />
can be revived and restored to their original living<br />
state. This can be used as a long-term<br />
conservation method and has been mainly applied<br />
to agricultural plants requiring specific genotype<br />
conservation [1], but this approach can also be<br />
applied to endangered or recalcitrant plant<br />
species.<br />
Poster Presentations<br />
Sunday 4 December - Session 1<br />
From plant tissue culture, L. Cinerea shoot tips are<br />
isolated, precultured, partial dehydrated then<br />
cryoprotected. This process aims to avoid<br />
intracellular ice formation that would otherwise<br />
disrupt the cell membranes integrity, leading to<br />
cellular death [2]. Dehydration and cryoprotection<br />
are both critical steps; too much desiccation<br />
results in over-exposure <strong>of</strong> the isolated shoot tips<br />
to cryoprotective agents, this too can be<br />
detrimental. Plant tissues can be successfully<br />
cryogenically stored when intracellular water is<br />
transformed into a vitrified glass during fast<br />
cooling. [3]<br />
Differential Scanning Calorimetry (DSC) measures<br />
the energy released during a phase transition, i.e.<br />
the formation <strong>of</strong> ice. DSC was performed on L.<br />
Cinerea shoot tips to determine whether ice<br />
formation takes place during LN cooling. DSC<br />
results revealed zero or very little ice formation in<br />
shoot tips treated with plant vitrification solution 2<br />
(PVS2) for 30 min, indicating that L. cinerea shoot<br />
tips are adequately cryoprotected with current<br />
protocols. Therefore unsuccessful re-growth <strong>of</strong><br />
shoot tips after cryopreservation cannot be<br />
explained by intracellular ice formation. Further<br />
research on membrane components and oxidative<br />
stress analysis during cryopreservation will help to<br />
develop a successful protocol for this endemic<br />
species.<br />
[1] Kaczmarczyk A., (2008), CryoLetters 29(2), 145-156.<br />
[2] Wolfe J., Bryant G., (1999), Cryobiology 39, 103-129.<br />
[3] Turner S., Touchell D., (2001), CryoLetters 22(3), 163-174.<br />
P30<br />
A DNA-Based Assay for Chemical<br />
Toxicity in Wastewater and Drinking<br />
Water<br />
Vangelis George Kanellis1 , Amy<br />
Foreman1 , Leo Phillips1 , Cris dos Remedios1 , David<br />
Hibbert2 , John Chapman3 , Moreno Julli3 , Ronald<br />
Patra3 1 Anatomy and Histology, The <strong>University</strong> <strong>of</strong> Sydney, Sydney,<br />
Anderson Stuart Building (F13), Sydney <strong>University</strong>, NSW, 2006,<br />
Australia, cris.dosremedios@sydney.edu.au<br />
2 School Chemistry , <strong>University</strong> <strong>of</strong> New South Wales, Sydney,<br />
Dalton Building, <strong>University</strong> <strong>of</strong> New South Wales, Kensington,<br />
2052, b.hibbert@unsw.edu.au<br />
3 New South Wales Government,<br />
121
Poster Presentations<br />
Sunday 4 December - Session 1<br />
Abstract<br />
We describe a monitoring system employing DNA<br />
as a biosensor <strong>of</strong> toxic chemicals. Metal ions and<br />
other toxicants bind to DNA causing structural<br />
changes that are detected by a fluorescent<br />
reporter dye. Statistical analyses <strong>of</strong> test data using<br />
48 wastewater samples indicate that the DNA-dye<br />
assay compares favourably with C. dubia and<br />
Microtox® bioassays as used by the Department<br />
<strong>of</strong> the Environment Climate Change and Water<br />
(DECCW). The DNA-dye system is simple,<br />
operates within a sample pH range <strong>of</strong> 4-10, is<br />
minute-long, has a 12-month use-by date, and<br />
appears well suited as a field screening assay for<br />
wastewater and drinking water. We found there is<br />
a good correlation between the sample toxicity<br />
and the heavy metal ion content as determined by<br />
mass spectrometry (ICP-MS or ICP-OES).<br />
However, it was also clear that some samples<br />
were toxic despite the absence <strong>of</strong> heavy metals.<br />
This suggests the DNA-dye system must have<br />
sensed toxic organic compounds contained within<br />
the samples. One <strong>of</strong> the main differences between<br />
bioassays and mass spectroscopy is the former’s<br />
synergistic or antagonistic toxic behaviours to<br />
mixtures <strong>of</strong> metal ions in aqueous solutions.<br />
Because environmental water samples rarely<br />
contain only one types <strong>of</strong> metal ion, we examined<br />
pairwise combinations <strong>of</strong> 16 metal ions (results<br />
shown below). Similar to other studies, we also<br />
noticed that some binary combinations <strong>of</strong> heavy<br />
metal ions were more toxic than other binary<br />
combinations. Two sources <strong>of</strong> genomic DNA were<br />
investigated. One seems to be more suited to<br />
testing the toxicity <strong>of</strong> wastewater, whilst the other<br />
more sensitive DNA-dye was more suitable for<br />
testing drinking water. Finally, we demonstrate<br />
using high resolution NMR that Hg binding causes<br />
a shift in the NMR spectrum consistent with it<br />
binding to the aromatic base pairs <strong>of</strong> the genomic<br />
DNA. We conclude the DNA-dye test is a<br />
surrogate bioassay suitable for screening chemical<br />
toxicity, particularly for toxic metal ions.<br />
122<br />
Observed toxicities for binary combinations <strong>of</strong><br />
metals ions using HAZ-1 DNA-dye. Open boxes<br />
indicate metals that have additive toxicities (44%).<br />
Boxes with vertical bars represent metals that,<br />
when combined, are less toxic than their separate<br />
additive effects; i.e., their effects are antagonistic<br />
(35%). Lightly shaded boxes represent metals with<br />
observed toxicities that are 50% greater than their<br />
separate additive effects, that is, synergistic (21%).<br />
The more darkly shaded boxes indicate metal<br />
combinations for which the observed toxicities are<br />
more than twice the toxicity predicted from their<br />
separate (independent) effects, that is, very<br />
synergistic (9%).<br />
P31<br />
Experimental and Computational<br />
Studies on Gas Phase Bimetallic<br />
Holmium-Rhodium Clusters<br />
Aidan M. Karayilan 1 , Gregory F. Metha1 1 <strong>University</strong> <strong>of</strong> Adelaide, North Terrace Campus Adelaide, SA,<br />
5005, aidan.karayilan@adelaide.edu.au<br />
2 <strong>University</strong> <strong>of</strong> Adelaide, North Terrace Campus Adelaide, SA,<br />
5005, greg.metha@adelaide.edu.au<br />
Abstract<br />
The adiabatic ionisation energies <strong>of</strong> six metal oxide<br />
cluster series (Ho2Rh3On (n = 1,2), Ho3On (n =<br />
1,2), Ho3RhOn (n = 0-4), Ho3Rh2On (n = 1-3),<br />
Ho4On (n = 1-6) and Ho4RhOn (n=1-3)) have been<br />
determined via photoionization efficiency<br />
spectroscopy. Computational studies were<br />
performed on three <strong>of</strong> the six metal oxide series<br />
(Ho3On (n = 1,2), Ho3RhOn (n = 0-4), Ho4On (n =<br />
1-6)) utilising DFT at the B3P86/SDD level <strong>of</strong> theory<br />
to determine theoretical ionisation energies to<br />
compare with experimental data. The<br />
computational data was found to be complicated,<br />
with many low lying isomers for each oxygen<br />
containing cluster found to have ionisation<br />
transitions correlating satisfactorily with<br />
experiment. The incorporation <strong>of</strong> rhodium into the<br />
holmium dominated oxide clusters was found to<br />
induce a large charge separation, with the rhodium<br />
atom attaining a δ- charge. The δ- charge on the<br />
rhodium atom was found to decrease linearly with<br />
oxidation level <strong>of</strong> the cluster.
P32<br />
Neutron membrane diffraction<br />
measurements <strong>of</strong> dehydrated DOPC<br />
bilayers with introduced sugars<br />
Ben Kent1 , Gary Bryant2 , Taavi Hunt2 and<br />
Christopher J. Garvey1 1 Bragg Institute, Australian Nuclear Science and Technology<br />
Organisation, Lucas Heights, Australia<br />
2 Applied Physics, School <strong>of</strong> Applied Sciences, RMIT <strong>University</strong>,<br />
Melbourne, Australia<br />
Abstract<br />
Sugars play a key role in the prevention <strong>of</strong> damage<br />
to natural cell membranes during desiccation or<br />
slow freezing. Their accumulation is a known<br />
natural defence against destabilisation <strong>of</strong> lipid<br />
bilayer membranes during low water availability.<br />
Dehydration causes the distance between bilayers<br />
to be reduced, resulting in the onset <strong>of</strong> the<br />
hydration force, which induces a compressive<br />
stress in the plane <strong>of</strong> the membrane, reducing the<br />
area per lipid headgroup and causing phase<br />
transitions to occur. While sugars are known to<br />
delay or prevent these transitions occurring, the<br />
mechanisms <strong>of</strong> membrane stabilisation by sugars<br />
are still under debate1 . Central to this debate is<br />
whether the observed effects are attributable to<br />
insertion <strong>of</strong> sugars between lipid headgroups<br />
preventing reduction in the area per lipid during<br />
compressive stress2 , or whether non-specific<br />
effects <strong>of</strong> sugars – volumetric and osmotic,<br />
combine to prevent close approach <strong>of</strong> membranes<br />
during dehydration, thereby delaying the onset <strong>of</strong><br />
hydration forces3 .<br />
Membrane diffraction is an established technique<br />
for the study <strong>of</strong> the structure <strong>of</strong> lipid bilayers and<br />
interaction with different molecules. Here we<br />
report on the use <strong>of</strong> the technique to study the<br />
interaction <strong>of</strong> a bilayer with a small solute<br />
molecule, glucose. Scattering density pr<strong>of</strong>iles<br />
obtained provide structural information across the<br />
bilayers and information on the location <strong>of</strong><br />
introduced solutes. Due to the differences in<br />
mechanisms between the competing membrane<br />
preservation theories, the concentration pr<strong>of</strong>ile <strong>of</strong><br />
sugars across the unit cell <strong>of</strong> the bilayer membrane<br />
should differ according to the dominant effect. We<br />
Poster Presentations<br />
Sunday 4 December - Session 1<br />
present results <strong>of</strong> neutron membrane diffraction<br />
measurements designed to study the interaction <strong>of</strong><br />
glucose with partially dehydrated DOPC bilayers.<br />
Concentration pr<strong>of</strong>iles <strong>of</strong> the sugars across the<br />
unit cell are extracted, and the resulting<br />
information on the interaction and location <strong>of</strong> the<br />
sugars is discussed in terms <strong>of</strong> mechanisms <strong>of</strong><br />
membrane protection.<br />
1. H. D. Andersen, C. Wang, L. Arleth, G. H. <strong>Peter</strong>s and P. Westh,<br />
Proceedings <strong>of</strong> the National Academy <strong>of</strong> Sciences, 2011, 108,<br />
1874-1878.<br />
2. L. M. Crowe, Comparative Biochemistry and Physiology - Part<br />
A: Molecular & Integrative Physiology, 2002, 131, 505-513.<br />
3. J. Wolfe and G. Bryant, Cryobiology, 1999, 39, 103-129.<br />
P33<br />
•NO ejection from R• + •NO2. Does<br />
•NO2 add through N or O?<br />
Benjamin B. Kirk1 , Adam J. Trevitt 1 , Stephen<br />
J. Blanksby1 1 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>, NSW, 2522<br />
Abstract<br />
Nitrogen dioxide (•NO2) is a free radical<br />
tropospheric pollutant which contributes to the<br />
brown colouration characteristic <strong>of</strong> photochemical<br />
smog. •NO2 undergoes a variety <strong>of</strong> reactions in<br />
the troposphere including radical-radical<br />
combination with alkyl and alkoxyl radicals [1].<br />
Recently, •NO2 was used as a radical trap to<br />
probe carbon-centred radical intermediates<br />
present during radical directed dissociation <strong>of</strong><br />
peptides in the gas phase [2]. Collision induced<br />
dissociation (CID) <strong>of</strong> these reaction products in a<br />
mass spectrometer resulted in ejection <strong>of</strong> •NO<br />
suggesting that •NO2 may have added through an<br />
oxygen. The ground state (2A1) <strong>of</strong> •NO2 formally<br />
places the unpaired electron on the nitrogen, thus,<br />
in order to add through oxygen, •NO2 must be<br />
excited to its first excited state (2B2) where the<br />
unpaired electron resides on oxygen. This<br />
excitation requires considerable energy at 27.8<br />
kcal mol-1 [3], suggesting that addition <strong>of</strong> •NO2<br />
should not occur through nitrogen under ambient<br />
conditions.<br />
123
Poster Presentations<br />
Sunday 4 December - Session 1<br />
In this study we attempted to distinguish between<br />
charge-tagged nitrobenzene and<br />
nitrosoxybenzene analogues using CID. These<br />
experiments were undertaken using an ion-trap<br />
mass spectrometer modified to allow a fixed flow<br />
<strong>of</strong> a neutral reagent (such as O2, •NO or •NO2) to<br />
be seeded into the helium buffer gas <strong>of</strong> the mass<br />
spectrometer. N,N,N-trimethylammonium<br />
charge-tagged analogues <strong>of</strong> nitrobenzene (A)<br />
generated by addition <strong>of</strong> •NO2 to a phenyl radical<br />
(C6H5• + •NO2 → C6H5NO2) and<br />
nitrosoxybenzene (B) generated by addition <strong>of</strong><br />
•NO to a phenoxyl radical (C6H5O• + •NO →<br />
C6H5ONO), were subjected to CID and a<br />
comparison made to the CID spectrum <strong>of</strong><br />
authentic N,N,N-trimethyl-4-nitrobenzaminium<br />
cation (C). CID <strong>of</strong> A and C resulted in a major ion<br />
due to neutral loss <strong>of</strong> •CH3 (Figure 1a). In contrast,<br />
the major product generated during CID <strong>of</strong> B<br />
occurred due to loss <strong>of</strong> •NO (Figure 1b). These<br />
results suggest that •NO2 adds to phenyl radicals<br />
via nitrogen and that loss <strong>of</strong> •NO observed during<br />
CID <strong>of</strong> •NO2 adducts most likely occurs due to<br />
isomerisation at the nitro substituent prior to<br />
ejection <strong>of</strong> •NO.<br />
[1] Atkinson, R.; Arey, J.; Chem. Rev., 2003, 103, 4605-4638<br />
[2] Barlow, C.K.; Wright, A.; Easton, C.J.; O’Hair, R.A.J.; Org.<br />
Biomol. Chem., 2011, 9, 3733–3745<br />
[3] Weaver, A.; Metz, R.B.; Bradforth, S.E.; Neumark, D.M.; J.<br />
Chem. Phys., 1989, 90, 2070–2071<br />
124<br />
P34<br />
Non-Markovian Memory from<br />
Time-Local Stochastic Trajectories<br />
Werner Koch1 , Frank Grossmann1 1 Research School <strong>of</strong> Chemistry, ANU, Canberra, ACT, 0200,<br />
Australia<br />
2 Institut für Theoretische Physik, TU Dresden, 01062 Dresden,<br />
Germany<br />
Abstract<br />
Markovian approximations for open quantum<br />
systems have seen an impressive range <strong>of</strong><br />
applications for describing the dynamics <strong>of</strong><br />
molecules under the disturbing influence <strong>of</strong> an<br />
environment. While in some special cases a<br />
Markovian treatment is exact, it remains an<br />
approximation for most others. In cases where the<br />
strict ranges <strong>of</strong> applicability <strong>of</strong> a certain<br />
approximation are transgressed, this does not<br />
necessarily void the results obtained from such a<br />
scheme. A comparison <strong>of</strong> such approximate<br />
results with the full non-Markovian treatment can<br />
identify the true limits for the (usually less arduous)<br />
Markovian method and highlight the details <strong>of</strong> how<br />
it fails. We present such an investigation for<br />
trajectory based implementations <strong>of</strong> the non-<br />
Markovian Stochastic Liouville-von-Neumann<br />
method [1] and a finite difference implementation<br />
<strong>of</strong> the Markovian Caldeira-Leggett master<br />
equation [2].<br />
[1] J. T. Stockburger, H. Grabert, Chem. Phys. 268 (2001) 249-256.<br />
[2] F. Grossmann, W. Koch, J. Chem. Phys. 130 (3) (2009) 034105.<br />
P35<br />
Quantum Effects in H2-Li+-Benzene:<br />
A Model For Hydrogen Storage<br />
Materials<br />
Stephen J. Kolmann 1 , Meredith J. T.<br />
Jordan2 1 School <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> Sydney, NSW, 2006.<br />
email: s.kolmann@chem.usyd.edu.au<br />
2 School <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> Sydney, NSW, 2006.<br />
email: m.jordan@chem.usyd.edu.au
Abstract<br />
The interaction <strong>of</strong> H2 with a Li+-benzene<br />
complex—a model for Li-doped MOF-5 and<br />
Li-doped carbon based materials—is<br />
characterised using Quantum Diffusion Monte<br />
Carlo (QDMC) and Path Integral Monte Carlo<br />
(PIMC) calculations on reduced- and fulldimensional<br />
modified Shepard interpolated<br />
potential energy surfaces, constructed using DFT.<br />
The importance <strong>of</strong> quantum effects and<br />
anharmonicity on the adsorption <strong>of</strong> H2 is<br />
quantified by comparing these QDMC and PIMC<br />
calculations to thermodynamic quantities obtained<br />
from a harmonic oscillator normal mode analysis.<br />
QDMC calculations suggest that the H2 ground<br />
state nuclear wavefunction is completely<br />
delocalised above the Li+-benzene complex, and<br />
the PIMC calculations allow us to examine the<br />
mobility <strong>of</strong> H2 with changing temperature. These<br />
calculations together suggest simplifications that<br />
can be used to model larger hydrogen storage<br />
systems.<br />
Recent modifications to the modified Shepard<br />
interpolation scheme <strong>of</strong> Collins and co-workers1 to<br />
interpolate systems <strong>of</strong> the size presented here will<br />
also be discussed.<br />
Figure 1. The H2-Li+-benzene minimum energy structure,<br />
optimised at the M05-2X/6-311+G(2df,p) level <strong>of</strong> theory. The Li+<br />
ion is sandwiched between the benzene ring and the H2 molecule.<br />
The Li+ to H2 centre-<strong>of</strong>-mass distance is indicated.<br />
1 M. A. Collins, Theor. Chem. Acc. 108, 313 (2002).<br />
Poster Presentations<br />
Sunday 4 December - Session 1<br />
P36<br />
Anion photoelectron spectra <strong>of</strong> the<br />
halide-(N2)n and -(N2O)n clusters<br />
Kim M. Lapere1 , Allan J. McKinley1 & Duncan<br />
A. Wild1 1 Chemistry, <strong>University</strong> <strong>of</strong> Western Australia, M313 35 Stirling<br />
Hwy Crawley, WA 6009, 20169424@student.uwa.edu.au<br />
Abstract<br />
We present recent work focusing on the anion<br />
photoelectron spectra and ab initio calculations <strong>of</strong><br />
the X-···(N2)n and X-···(N2O)n clusters (X = Cl, Br<br />
and I). Spectra <strong>of</strong> these species are recorded<br />
using our newly-constructed Time-Of-Flight mass<br />
spectrometer coupled to a PhotoElectron<br />
Spectrometer (TOF-PES)[1].<br />
We explore the radical X•···(N2)n and X•···(N2O)n<br />
clusters via photodetachment <strong>of</strong> an electron from<br />
the anion complexes. The work samples the<br />
reaction potential energy surface for the dimer<br />
complex, and follows the solvation upon increased<br />
cluster size.<br />
Fundamental data such as the electron affinities<br />
(EA) and cluster stabilisation energies (Estab) <strong>of</strong><br />
the clusters are derived from experimental spectra<br />
and compared with ab initio calculations for the<br />
anion and neutral radical monomer species<br />
employing MP2 and CCSD(T) methodologies. We<br />
present geometries, vibrational frequencies and<br />
energies for the Cl•···N2 and Br•···N2 clusters.<br />
References:<br />
Lapere, K. M.; LaMacchia, R. J.; Quak, L. H.; McKinley, A. J.; Wild,<br />
D. A. Chemical Physics Letters 2011, 504, 13-19.<br />
P37<br />
Transient Absorption Spectroscopy<br />
<strong>of</strong> Curcumin-Copper Complex<br />
Hei Man Mandy Leung ,1 , Dr Tak W. Kee2 1 School <strong>of</strong> Chemistry and Physics, <strong>University</strong> <strong>of</strong> Adelaide,<br />
Adelaide, South Australia, 5005, Australia,<br />
hei.leung@adelaide.edu.au<br />
2 School <strong>of</strong> Chemistry and Physics, <strong>University</strong> <strong>of</strong> Adelaide,<br />
Adelaide, South Australia, 5005, Australia, tak.kee@adelaide.<br />
edu.au<br />
125
Poster Presentations<br />
Sunday 4 December - Session 1<br />
Abstract<br />
Curcumin is the yellow pigment from turmeric<br />
which exhibits anti-cancer and anti-amyloid<br />
properties.<br />
Studies have suggested that the anticancer and<br />
anti-amyloid action <strong>of</strong> curcumin can be related to<br />
its interaction with copper ions because there is an<br />
elevated concentration <strong>of</strong> copper ions in tumours<br />
and amyloid aggregates. In this work, ultrafast<br />
transient absorption spectroscopy is used to study<br />
the excited state <strong>of</strong> curcumin and curcumincopper<br />
complex to elucidate the interactions<br />
between curcumin and copper. Our results show<br />
that the excited state lifetime <strong>of</strong> curcumin has<br />
shortened when it is complexed in both the SDS<br />
micellar solution and methanol. T his result<br />
suggests that ligand-metal charge transfer (LMCT)<br />
as a pathway for excited state relaxation.<br />
P38<br />
Optimising the responsive behaviour<br />
<strong>of</strong> polymer surfaces using Molecular<br />
Dynamics<br />
Kamron Ley 1 , George Yiapanis 1 , Irene<br />
Yarovsky 1 , Evan Evans 2<br />
1 Applied Sciences, RMIT <strong>University</strong>, GPO BOX 2476V, Victoria,<br />
3001, Australia<br />
2 BlueScope Steel Research, Port Kembla, NSW, Australia<br />
Abstract<br />
Recent advancements made in polymer<br />
modification have lead to a large demand for<br />
responsive surfaces, particularly in the paintcoatings<br />
industry. This is because responsive<br />
systems have the potential to shed dirt by<br />
reversibly switching between hydrophobic and<br />
hydrophilic states with changing humidity.<br />
In this study, we looked at utilising responsive<br />
hydrophilic polymer brushes in the form <strong>of</strong><br />
polyethylene glycol (PEG) oligomers grafted on an<br />
organic polyester based substrate1-4, with varying<br />
levels <strong>of</strong> surface hydroxylation. By employing<br />
force-field based molecular mechanics and<br />
dynamics we were able to gain insight into the<br />
responsive behaviour <strong>of</strong> these PEG modified<br />
substrates under a variety <strong>of</strong> conditions and<br />
126<br />
environments, with particular interest in the<br />
property <strong>of</strong> PEG expansion driven by<br />
intermolecular hydrogen bonding with both the<br />
substrate and other PEG chains.<br />
Our initial research demonstrates that increasing<br />
the grafting density results in interactions between<br />
the grafted polymer segments. This in turn leads<br />
different configurations being adopted by the PEG<br />
chains, where they transition from a flat<br />
configuration to a mushroom like configuration<br />
(see figure below), which in turn leads to an<br />
increased response displayed by the surfaces.<br />
Typical MD configurations adopted by the PEG<br />
chains; at lower grafting densities (flat), and higher<br />
grafting densities (mushroom).<br />
(1) Yarovsky, I.; Evans, E.; Polymer 2002, 43, 963<br />
(2) Yiapanis, G.; Evans, E.; Henry, D. J.; Yarovsky, I. J Phys Chem C<br />
2010, 114, 478.<br />
(3) Yiapanis, G.; Henry, D. J.; Evans, E.; Yarovsky, I. J Phys Chem<br />
C 2008, 112, 18141.<br />
(4) Yiapanis, G.; Henry, D. J.; Evans, E.; Yarovsky, I. In<br />
Nanotechnology in Australia: Showcase <strong>of</strong> Early Career<br />
Research in Australia; Kane, D. M., Micolich, A. P., Rabeau, J.<br />
R., Eds.; Pan Stanford Publishing: 2011.<br />
P39<br />
Laser Spectroscopic Studies <strong>of</strong><br />
Conformation in Neurotransmitters<br />
Isabella Antony Lobo1 , David Wilson2 ,<br />
Evan Bieske3 , Evan G Robertson 4<br />
1 Department <strong>of</strong> Chemistry, La Trobe <strong>University</strong>, Bundoora,<br />
Victoria, 3086, ialobo@students.latrobe.edu.au<br />
2 Department <strong>of</strong> Chemistry, La Trobe <strong>University</strong>, Bundoora,<br />
Victoria, 3086, david.wilson@latrobe.edu.au<br />
3 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Melbourne, Victoria, 3010,<br />
evanjb@unimelb.edu.au<br />
4 Department <strong>of</strong> Chemistry, La Trobe <strong>University</strong>, Bundoora,<br />
Victoria, 3086, e.robertson@latrobe.edu.au<br />
Abstract
Neurotransmitters are special class <strong>of</strong> biological<br />
compounds controlling physical and mental<br />
well-being. Probing the preferred conformational<br />
structures <strong>of</strong> these biomolecules paves the way for<br />
the development <strong>of</strong> drugs. The conformers <strong>of</strong><br />
amino-p-phenethylamine (APEA) were studied in a<br />
jet-cooled environment by Resonance Enhanced<br />
Two Photon Ionisation (R2PI) and IR-UV ion<br />
depletion techniques. In this way conformerspecific<br />
IR spectra were measured for four distinct<br />
conformers <strong>of</strong> APEA. The experimental studies are<br />
complimented by ab initio calculations conducted<br />
at various levels <strong>of</strong> theory including g MP2/6-<br />
311G+(d,p) and B3LYP/6-311G+(d,p). Comparison<br />
<strong>of</strong> experimental and computed spectra allows the<br />
two most populated conformers to be<br />
unambiguously identified as those having a<br />
gauche arrangement <strong>of</strong> the side chain which<br />
facilitates an NH… type hydrogen bond. The other<br />
two observed conformers are assigned to<br />
structures with an anti side chain. The fifth gauche<br />
conformer, predicted to be least stable, is not<br />
observed. The above findings are in clear contrast<br />
to a recent study on APEA, relying mainly on<br />
ionisation energies as well as computational<br />
methods, that identified an anti conformer as<br />
being the most stable and heavily populated one.<br />
Tranylcypromine (Parnate) is another subject for<br />
these studies. It is a drug <strong>of</strong> the substituted<br />
phenethylamine and amphetamine classes and<br />
used in the treatment <strong>of</strong> mood and anxiety<br />
disorders. Results for this compound will also be<br />
presented.<br />
Poster Presentations<br />
Sunday 4 December - Session 1<br />
Notes<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
127
Poster Presentations<br />
Monday 5 December - Session 2<br />
P40<br />
Single Cell Membrane Analysis by<br />
TERS is Reaching Nanometer Scale<br />
Christian Löbbe1 , Marc Richter2 , Heiko<br />
Haschke2 , Martin Hedegaard 3 , Tanja Deckert-<br />
Gaudig4 , <strong>Peter</strong> Lampen5 , Volker Deckert 4,6<br />
1 Scitech P/L, 4/72-74 Chifley Dr, Preston 3072, Australia<br />
2 JPK Instruments AG, Bouchestr. 12, 12435 Berlin, Germany<br />
3 Technical Faculty <strong>University</strong> <strong>of</strong> Southern Denmark, Institute <strong>of</strong><br />
Sensors, Signals and Electrotechnics (SENSE), Campusvej 55,<br />
5230 Odense M, Denmark<br />
4 Institute <strong>of</strong> Photonic Technology (IPHT), Albert-Einstein-Straße<br />
9, 07745 Jena, Germany<br />
5 Leibnitz-Institut für Analytische Wissenschaften – ISAS e.V.,<br />
Bunsen-Kirchh<strong>of</strong>f-Str. 11, Dortmund 44139, Germany<br />
6 Friedrich-Schiller-Universität Jena, Institute <strong>of</strong> Physical<br />
Chemistry, Helmholzweg 4, 07743 Jena, Germany<br />
Abstract<br />
It is known that cell surface glycoproteins are<br />
acting as cell specific identifiers for cell-cell<br />
interactions. Those macromolecules are <strong>of</strong>ten<br />
important integral membrane proteins, where they<br />
play a role as a receptor for active ingredients and<br />
second messengers.<br />
A common method to identify membrane proteins<br />
is antibody labeling. Depending on the nature <strong>of</strong><br />
the markers it is possible to use fluorescence or<br />
Raman spectroscopy as an analytical method.<br />
Especially silver and gold-labeled antibodies<br />
turned out to be very interesting as they can be<br />
used to increase the sensitivity <strong>of</strong> Raman labels via<br />
a plasmon enhancement1. However, the lateral<br />
resolution capability with respect to the location <strong>of</strong><br />
specific protein arrangements <strong>of</strong> this method is<br />
limited. Another disadvantage <strong>of</strong> labeling with<br />
antibodies is the selectivity <strong>of</strong> the marker. Different<br />
and specific markers must be chosen for each<br />
protein <strong>of</strong> interest.<br />
To provide spectroscopic information with high<br />
spatial resolution tip-enhanced Raman scattering<br />
(TERS) is the technique <strong>of</strong> choice2. The<br />
combination <strong>of</strong> an atomic force microscope (AFM)<br />
with a Raman microscope provides information on<br />
the topography and the molecular structure <strong>of</strong> a<br />
sample with high sensitivity.<br />
We present results <strong>of</strong> TERS mapping<br />
measurements on colon cancer cells. In particular,<br />
128<br />
an area <strong>of</strong> 90x90 nm was analyzed. Within this<br />
area spectra were recorded on a square grid with<br />
a spacing <strong>of</strong> 10 nm.<br />
Based on hyperspectral unmixing algorithm<br />
(N-FINDR), a band assignment <strong>of</strong> the spectrum <strong>of</strong><br />
each endmember was done. As expected, all the<br />
TERS bands can be attributed to proteins or lipids,<br />
the known components <strong>of</strong> the cell membrane.<br />
Correlating the band assignment and the N-FINDR<br />
results . a label-free localization <strong>of</strong> membrane<br />
components could be achieved. We demonstrate<br />
that the combination <strong>of</strong> high lateral resolution and<br />
specificity <strong>of</strong> TERS potentially allows a direct<br />
differentiation <strong>of</strong> membrane protein and lipid<br />
regions3.<br />
References<br />
[1] J.Kneipp, H. Kneipp, K. Kneipp, Chem. Soc. Rev. 27 (2008),<br />
1052–1060.<br />
[2]E. Bailo, V. Deckert, Chem. Soc. Rev. 37 (2008), 921-930.<br />
[3]M. Richter et al, Small 7 (2011), 209-214.<br />
P41<br />
Reinvestigating the 308 nm<br />
Photodissociation Dynamics <strong>of</strong><br />
Acetaldehyde: A Velocity Map<br />
Imaging Study Examining Two<br />
Distinct Pathways Producing CO +<br />
CH4<br />
Alan T. Maccarone 1 , Mitchell S. Quinn2 ,<br />
Gabi de Wit3 , Scott A. Reid4 , B. Klaas Nauta5 ,<br />
Scott H. Kable6 1 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney, New South Wales<br />
2006, alanmac@uow.edu.au<br />
2 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney, New South Wales<br />
2006, mqui5334@uni.sydney.edu.au<br />
3 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney, New South Wales<br />
2006, g.dewit@chem.usyd.edu.au<br />
4 Department <strong>of</strong> Chemistry, Marquette <strong>University</strong>, Milwaukee, WI,<br />
USA, scott.reid@marquette.edu<br />
5 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney, New South Wales<br />
2006, k.nauta@chem.usyd.edu.au<br />
6 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney, New South Wales<br />
2006, s.kable@chem.usyd.edu.au<br />
Abstract<br />
Since the discovery <strong>of</strong> the non-classical roaming<br />
mechanism to form carbon monoxide and
molecular hydrogen in the photodissociation <strong>of</strong><br />
formaldehyde [1], similar pathways have been<br />
discovered and postulated to exist in other<br />
systems [2,3]. Notably acetaldehyde (CH3CHO)<br />
has received much attention, with several studies<br />
examining the dynamics and energy dependence<br />
<strong>of</strong> the channels producing methane (CH4) and<br />
carbon monoxide (CO) [4,5,6]. Despite many<br />
thorough investigations, there are still<br />
discrepancies in the branching ratio <strong>of</strong> the roaming<br />
to transition state pathway.<br />
In an experimental study, Velocity Map Images <strong>of</strong><br />
CO(v=0,J) phot<strong>of</strong>ragments from the 308 nm<br />
photodissociation <strong>of</strong> acetaldehyde were BASEX<br />
transformed to obtain CO speed distributions. The<br />
distributions for J between 10 and 30 are clearly<br />
bimodal and are well fit by two characteristic<br />
temperatures. This bimodality reaffirms the<br />
presence <strong>of</strong> two dynamically different mechanisms<br />
to form CO + CH4, first proposed by Houston and<br />
Kable using LIF and Doppler analysis [2]: one<br />
produces CO with relatively little speed and<br />
angular momentum, while the second makes fast<br />
CO in high angular momentum states. When CH4<br />
internal energy distributions are calculated directly<br />
from the total kinetic energy distributions through<br />
conservation <strong>of</strong> energy, the result is in good<br />
agreement with the FT-IR results <strong>of</strong> Heazlewood<br />
et. al. [5]. The branching ratio between the two<br />
different pathways is seen to heavily favor the<br />
mechanism which produces slow CO partnered<br />
with highly internally excited CH4 fragments.<br />
Theoretical work to describe these two different<br />
mechanisms via direct dynamics calculations has<br />
recently uncovered a few caveats. Experiments to<br />
examine the competition between the two<br />
pathways as a function <strong>of</strong> available energy could<br />
help towards a more complete evaluation <strong>of</strong> the<br />
dynamics in acetaldehyde photodissociation.<br />
References:<br />
[1] D. Townsend, S.A. Lahankar, S.K. Lee, S.D. Chambreau, A.G.<br />
Suits, X. Zhang, J. Rheinecker, L.B. Harding, J.M. Bowman,<br />
Science, 306, 1158 (2004).<br />
[2] P.L. Houston, S.H. Kable, PNAS, 103, 16079 (2006).<br />
[3] V. Goncharov, N. Herath, A.G. Suits, J. Phys. Chem. A, 112,<br />
9423 (2008).<br />
[4] B.F. Gherman, R.A. Friesner, T. Wong, Z. Min, R. Bersohn, J.<br />
Chem. Phys., 114, 6128 (2001).<br />
[5] B.R. Heazlewood, M.J.T. Jordan, S.H. Kable, T.M. Selby, D.L.<br />
Osborn, B.C. Shepler, B.J. Braams, J.M. Bowman, PNAS, 105,<br />
12719 (2008).<br />
[6] L.B. Harding, Y. Georgievskii, S.J. Klippenstein, J. Phys. Chem.<br />
A, 114, 765 (2010).<br />
Poster Presentations<br />
Monday 5 December - Session 2<br />
P42<br />
Understanding the immune<br />
interactions <strong>of</strong> an unstructured<br />
protein antigen: the malaria surface<br />
protein MSP2<br />
Christopher A. MacRaild1 , Marie Ø.<br />
Pedersen1 , Christopher G. Adda2 , Robin F. Anders2 and Raymond S. Norton1 1 Department <strong>of</strong> Medicinal Chemistry and Drug Action, Monash<br />
<strong>University</strong>, Melbourne, Australia.<br />
2 Department <strong>of</strong> Biochemistry, La Trobe <strong>University</strong>, Melbourne,<br />
Australia<br />
Abstract<br />
Merozoite surface protein 2 (MSP2) is one <strong>of</strong> the<br />
most abundant proteins on the surface <strong>of</strong> the<br />
merozoite stage <strong>of</strong> Plasmodium falciparum, and is<br />
a clinically validated candidate for inclusion in a<br />
malaria vaccine1. MSP2 is a GPI-anchored protein<br />
consisting <strong>of</strong> conserved N- and C-terminal regions<br />
and a variable central region <strong>of</strong> low sequence<br />
complexity. Like many malaria antigens, MSP2 is<br />
intrinsically unstructured, with few regions <strong>of</strong><br />
limited conformational restriction in solution2. The<br />
central variable region appears to dominate natural<br />
and protective immune responses; the mechanism<br />
by which the conserved regions apparently evade<br />
immune surveillance is unclear. More generally, the<br />
structural basis and functional significance <strong>of</strong><br />
immune interactions with unstructured proteins is<br />
poorly understood. We address these questions<br />
by characterising the conformational behavior <strong>of</strong><br />
MSP2 under a range <strong>of</strong> solution conditions, and by<br />
probing the interactions <strong>of</strong> a panel <strong>of</strong> monoclonal<br />
antibodies against recombinant MSP2 in solution<br />
and native MSP2 on the merozoite surface. These<br />
antibodies recognise recombinant MSP2 but<br />
possess variable abilities to recognise MSP2 on<br />
the merozoite surface, consistent with patterns <strong>of</strong><br />
natural human immune responses. Understanding<br />
the conformational basis <strong>of</strong> these cryptic epitopes<br />
may aid the design <strong>of</strong> more effective vaccines<br />
based on MSP2.<br />
1. Genton, B., Betuela, I., Felger, I., Al-Yaman, F., Anders, R. F.,<br />
Saul, A. et al. (2002). J. Infect. Dis. 185: 820–827.<br />
2. Zhang, X., Perugini, M. A., Yao, S., Adda, C. G., Murphy, V. J.,<br />
Low, A., Anders, R. F. and Norton, R. S. (2008). J. Mol. Biol.<br />
379: 105-121.<br />
129
Poster Presentations<br />
Monday 5 December - Session 2<br />
P43<br />
Computational investigation <strong>of</strong><br />
binding <strong>of</strong> kappa conotoxin to<br />
voltage-gated potassium channels<br />
S. Mahdavi 1 and S. Kuyucak1 1-School <strong>of</strong> Physics, <strong>University</strong> <strong>of</strong> Sydney, NSW2006, Australia<br />
Abstract<br />
PVIIA is a member <strong>of</strong> the kappa conotoxin family<br />
that inhibits the drosophila Shaker potassium<br />
channel (Shaker). While Shaker is sensitive to this<br />
toxin, the rat Kv1.1 channel is resistant to it. Here,<br />
we study the interactions <strong>of</strong> PVIIA with the Shaker<br />
and Kv1.1 channel to determine the molecular<br />
interactions that give rise to this selectivity.<br />
The 3D structure <strong>of</strong> Shaker and Kv1.1 are created<br />
by structural homologies with Kv1.2 structure<br />
(3LUT). We have docked PVIIA (ID:1AV3) on both<br />
channels using HADDOCK. The best complex for<br />
each channel is selected and embedded in a lipid<br />
bilayer. They are equilibrated and ran for up to<br />
20ns in MD simulations. From the trajectory data,<br />
we have determined pair-residue interactions in<br />
each channel-toxin complex.<br />
In the Shaker-PVIIA complex, K7 enters the filter<br />
and makes contact with the Y445 carbonyls. The<br />
R2, K25, R22, N5 and N24 residues are also<br />
responsible for electrostatic interactions and<br />
hydrogen bonds through their contact with N423<br />
and D447. There are several hydrophobic<br />
interactions between I3, F9, L12, F23 and F425,<br />
V451, which are stable throughout the simulation.<br />
The Shaker-PVIIA structure yields results that are<br />
in good agreement with experiments. We are now<br />
performing free energy perturbation (FEP)<br />
calculations to determine the changes in the<br />
binding affinities when the toxin residues are<br />
mutated. This will provide a more quantitative test<br />
for the proposed complex and increase the<br />
confidence in the computational model.<br />
130<br />
Figure1: Side view <strong>of</strong> two monomers <strong>of</strong> Shaker and PVIIA (in pink)<br />
in Shaker-PVIIA complex . R2, K7 and N24 residues in toxin are<br />
explicitly shown.<br />
For Kv1.1-PVIIA complex, most charged and polar<br />
residues <strong>of</strong> the toxin are in contact with E353 and<br />
H355 . Consequently, electrostatic interactions<br />
between Kv1.1and toxin are more probable.<br />
However, these interactions are near the turret<br />
region <strong>of</strong> the pore and fluctuate frequently. There<br />
are no significant hydrophobic interactions as they<br />
mostly mismatch.<br />
In conclusion, in Shaker there are several stable<br />
interactions <strong>of</strong> the toxin residues with the residues<br />
near the selectivity filter and the turret region,<br />
which generate a stable complex. In contrast, for<br />
Kv1.1 most interactions are in the turret region<br />
which are not very stable, and there is<br />
considerable mismatch among the hydrophobic<br />
residues.<br />
P44<br />
Ab initio and classical molecular<br />
dynamics study <strong>of</strong> the aggregation<br />
propensities <strong>of</strong> amyloidogenic<br />
peptides in the presence <strong>of</strong><br />
nanomaterials<br />
A.J. Makarucha 1 , N. Todorova1 , A. Most<strong>of</strong>i 2 ,<br />
I. Yarovsky 1<br />
1 Health Innovations Research Institute, School <strong>of</strong> Applied<br />
Sciences, RMIT <strong>University</strong>, GPO Box 2476 V, Melbourne, VIC,<br />
Australia.<br />
2 Department <strong>of</strong> Material s & Physics, Imperial College London,<br />
London, U.K., SW7 2AZ
Abstract<br />
As nanotechnology becomes more prevalent in<br />
industrial, consumer and medical industries the<br />
rate <strong>of</strong> exposure to nanomaterials increases and<br />
the need to understand the effects that these<br />
materials have on biological systems is paramount.<br />
Computational studies have been conducted to<br />
investigate the effects <strong>of</strong> nanomaterials on many <strong>of</strong><br />
these biological systems however few studies have<br />
considered how surfaces and nanomaterials could<br />
promote or inhibit peptide self-assembly <strong>of</strong> fibril<br />
forming proteins 1.<br />
Amyloidogenic proteins, which form insoluble<br />
protein aggregates are involved in many<br />
degenerative diseases, such as Parkinson’s,<br />
Alzhiemer’s, Creutzfeld-Jacob disease, and<br />
dialysis-related amyloidosis. It has been shown<br />
that due to the large surface area <strong>of</strong> nanoparticles<br />
the rate <strong>of</strong> protein fibrillation and thus aggregation<br />
can be enhanced by decreasing the lag time for<br />
nucleation 2. Apolipoprotein C-II (apoC-II) is a type<br />
<strong>of</strong> plasma apolipoprotein known to aggregate in<br />
lipid-depleted conditions, and its fibrils are a major<br />
component in human atherosclerotic plaques 3. In<br />
addition to the full-length protein, the peptide<br />
fragments composed <strong>of</strong> residues 60 to 70<br />
possess an inherent propensity for amyloid fibril<br />
formation in solution. Comprehensive investigation<br />
has been performed on the effects <strong>of</strong> mutation, pH<br />
and lipid environment on the amyloidogenic<br />
propensities <strong>of</strong> apoC-II(60-70)4-7.<br />
Explicit solvent molecular dynamics was utilised to<br />
investigate the aggregation propensities <strong>of</strong><br />
apoC-II(60-70) peptide in the presence <strong>of</strong><br />
carbonaceous nanomaterials, such as fullerene<br />
(C60), single walled carbon nanotube and a<br />
graphene sheet. The different shape <strong>of</strong> these<br />
nanomaterials allowed for the effects <strong>of</strong> curvature<br />
to be investigated on the fibril formation<br />
propensities <strong>of</strong> the peptide. Simulations <strong>of</strong><br />
monomeric and dimeric apoC-II(60-70) were<br />
performed. The results were compared with our<br />
previous studies <strong>of</strong> apoC-II(60-70) peptides in<br />
ambient conditions 5. We identified the<br />
conformational changes in the peptide due to<br />
curvature effects and surface-peptide interactions<br />
that could affect the fibrillation propensities <strong>of</strong><br />
apoC-II(60-70).<br />
Poster Presentations<br />
Monday 5 December - Session 2<br />
The linear-scaling DFT package, ONETEP, was<br />
utilised to characterise the electronic structure and<br />
binding energies <strong>of</strong> the previously identified<br />
peptide – nanomaterial complexes using classical<br />
MD. Geometry optimisation and binding energy<br />
calculations were performed to characterise the<br />
affinity <strong>of</strong> the peptide for individual nanomaterial.<br />
We also identified the key residues in apoC-II(60-<br />
70) that are responsible for association with<br />
carbonaceous nanomaterials.<br />
Chemistry B 113, 9447-53 (2009).<br />
ApoC-II (60-70) adsorbed to carbon nanotube and<br />
fullerene<br />
1. Makarucha, A.J., Todorova, N. & Yarovsky, I. Nanomaterials in<br />
biological environment: a review <strong>of</strong> computer modelling studies.<br />
European Biophysics Journal 40, 103-115 (2010).<br />
2. Linse, S. et al. Nucleation <strong>of</strong> protein fibrillation by nanoparticles.<br />
Proc Natl Acad Sci U S A 104, 8691-8696 (2007).<br />
3. Hatters, D.M., MacPhee, C.E., Lawrence, L.J., Sawyer, W.H. &<br />
Howlett, G.J. Human apolipoprotein C-II forms twisted amyloid<br />
ribbons and closed loops. Biochemistry 39, 8276-83 (2000).<br />
4. Todorova, N. et al. Effects <strong>of</strong> mutation on the amyloidogenic<br />
propensity <strong>of</strong> apolipoprotein C-II60–70 peptide. Physical<br />
Chemistry Chemical Physics 12, 14762 (2010).<br />
5. Hung, A., Griffin, M.D.W., Howlett, G.J. & Yarovsky, I. Effects <strong>of</strong><br />
oxidation, pH and lipids on amyloidogenic peptide structure:<br />
implications for fibril formation? European Biophysics Journal<br />
38, 99-110 (2008).<br />
6. Todorova, N., Hung, A. & Yarovsky, I. Lipid concentration effects<br />
on the amyloidogenic apoC-II(60-70) peptide: a computational<br />
study. Journal <strong>of</strong> Physical Chemistry B 114, 7974-82 (2010).<br />
7. Hung, A., Griffin, M.D., Howlett, G.J. & Yarovsky, I. Lipids<br />
enhance apolipoprotein C-II-derived amyloidogenic peptide<br />
oligomerization but inhibit fibril formation. Journal <strong>of</strong> Physical<br />
131
Poster Presentations<br />
Monday 5 December - Session 2<br />
P45<br />
Competitive N-O and O-C homolysis<br />
in TEMPO-based alkoxyamines<br />
David L. Marshall1 , Martin R. L. Paine1 ,<br />
Ganna Gryn’ova2 , Philip J. Barker3, Michelle L.<br />
Coote2 , Stephen J. Blanksby1 1 ARC Centre <strong>of</strong> Excellence for Free Radical Chemistry and<br />
Biotechnology & School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>,<br />
NSW, 2522<br />
2 ARC Centre <strong>of</strong> Excellence for Free Radical Chemistry and<br />
Biotechnology & Research School <strong>of</strong> Chemistry, Australian<br />
National <strong>University</strong>, Canberra ACT, 0200<br />
3 BlueScope Steel Research, P.O Box 202, Port Kembla, NSW,<br />
2502<br />
Abstract<br />
2,2’,6,6’-tetramethylpiperidine-N-oxyl (TEMPO)<br />
free radicals are highly stable radical scavengers.<br />
As such, they find wide usage as the active form <strong>of</strong><br />
antioxidant hindered amine light stabilizers (HALS)<br />
in surface coatings, including COLORBOND®<br />
steel. The mechanism <strong>of</strong> action involves reversible<br />
cycling between the nitroxyl radical and<br />
alkoxyamine, the net result being the conversion <strong>of</strong><br />
deleterious alkyl and peroxyl radicals into inert<br />
products, with the continual regeneration <strong>of</strong> the<br />
active nitroxyl, as described by the “Denisov<br />
Cycle”.<br />
However, from empirical evidence it is clear that<br />
HALS are inevitably chemically deactivated or<br />
physically removed from the polymer; leading to<br />
the eventual fading or discolouration <strong>of</strong> the<br />
coating. We have previously demonstrated that<br />
hydroxyl radicals decompose HALS into volatile<br />
by-products, which would be readily lost by<br />
evaporation during polymer cure, or in-service. An<br />
increased abundance <strong>of</strong> secondary amines were<br />
also detected upon exposure <strong>of</strong> TEMPO<br />
derivatives to hydroxyl radicals, implying that N-H<br />
amines (and by extension, aminyl radicals) may<br />
play a greater role in the stabilization mechanisms<br />
<strong>of</strong> HALS than previously considered. 1<br />
Analysis <strong>of</strong> 4-carboxy-TEMPO based<br />
alkoxyamines by negative ion electrosprayionization<br />
mass spectrometry (ESI-MS) was<br />
undertaken, with either collision induced<br />
dissociation (CID) or photodissociation (PD)<br />
tandem mass spectrometry (MS/MS). Herein, we<br />
132<br />
demonstrate a competition between nitrogenoxygen<br />
and oxygen-carbon bond homolysis,<br />
dependent on the structure <strong>of</strong> the captured radical<br />
that forms the alkoxyamine. Moreover, for certain<br />
alkoxyamine structures, the nitrogen-oxygen bond<br />
is cleaved preferentially over the oxygen-carbon<br />
bond.<br />
The observed structure-dependent trends<br />
correlate well with previous high level ab initio<br />
calculations into relative free energies <strong>of</strong> homolysis<br />
between the N-O and O-C bonds. For example,<br />
heteroatoms in the α-position to the nitroxyl<br />
oxygen stabilize the oxygen-carbon bond through<br />
anomeric interactions, therefore promoting<br />
nitrogen-oxygen homolysis. Conversely, a<br />
neighbouring aromatic system greatly enhances<br />
O-C homolysis through stabilization <strong>of</strong> the formed<br />
benzyl radical. 2<br />
The primary products <strong>of</strong> the undesirable N-O<br />
cleavage are aminyl and alkoxyl radicals. If<br />
repeated within surface coatings, the antioxidant<br />
activity <strong>of</strong> the nitroxyl radical is lost, and moreover<br />
the formed alkoxyl radicals may further propagate<br />
polymer degradation. Indeed, an<br />
oxidation product attributed to N-O homolysis has<br />
already been observed after accelerated<br />
weathering <strong>of</strong> a polymer coating containing a<br />
commercially available alkoxyamine HALS. 3<br />
Understanding the structure-activity relationship<br />
between these two competing homolysis<br />
processes will prove extremely useful, particularly<br />
for assisting the selection <strong>of</strong> systems that promote<br />
the desired O-C homolysis and/or resist the<br />
unwanted N-O homolysis pathway.<br />
1. Marshall DL, Christian ML, Gryn’ova G, Coote ML, Barker PJ,<br />
Blanksby SJ. Organic & Biomolecular Chemistry 2011; 9: 4936.<br />
2. Hodgson JL, Roskop LB, Gordon MS, Lin CY, Coote ML. J.<br />
Phys. Chem. A 2010; 114: 10458.<br />
3. Paine MRL, Barker PJ, Blanksby SJ. Analyst 2011; 136: 904.
P46<br />
Thermodynamics and Metabolomics<br />
integration into metabolic Genome<br />
Scale Model to resolve metabolic<br />
flux directions<br />
Veronica Martinez1 , Stefanie Dietmair1 ,<br />
Lake-Ee Quek1 , Lars Keld Nielsen1 1 Australian Institute for Bioengineering and Nanotechnology, The<br />
<strong>University</strong> <strong>of</strong> Queensland Australia , Corner College and Cooper<br />
Rds (Bldg 75) Brisbane, Qld, 4072<br />
Abstract<br />
The incorporation <strong>of</strong> the second law <strong>of</strong><br />
thermodynamics and the standard Gibbs energy<br />
<strong>of</strong> formation with metabolomics data into<br />
metabolic models can reveal the flux direction for a<br />
subset <strong>of</strong> reversible reactions under a given<br />
physiological condition. The flux solution space is<br />
reduced when the direction <strong>of</strong> a reversible reaction<br />
is fixed. This approach allows us to apply<br />
metabolomics to better quantify cell metabolism.<br />
For example, for parallel pathways that are found<br />
in more than one compartment, it is possible to<br />
distinguish between the degradation and the<br />
synthesis routes if the reaction directions are<br />
known. This will lead to a better understanding <strong>of</strong><br />
the possible functional states <strong>of</strong> the cell.<br />
A s<strong>of</strong>tware based on NET optimization was<br />
developed to incorporate thermodynamic<br />
constraints with metabolite concentrations. A<br />
Human Genome Scale Model (GeM) was modified<br />
to work with two compartments (cytoplasm and<br />
mitochondria). Compartment properties (pH, ionic<br />
strength, etc ) for mammalian cells were compiled<br />
from literature together with thermodynamic data.<br />
By applying only thermodynamic constraints to a<br />
Human GeM 15 new irreversible reactions were<br />
found. Nonetheless, there are still 1188 reversible<br />
reactions that are not constrained. Accurate<br />
metabolite concentrations are required to resolve<br />
the direction <strong>of</strong> these reactions, but it is at present<br />
not possible to determine the concentration <strong>of</strong> all<br />
metabolites. Preliminary studies on HEK293 cells<br />
revealed that no new reaction directions could be<br />
determined despite measuring concentrations <strong>of</strong><br />
13 nucleotides. Therefore, targeted metabolomics<br />
must be applied. In silico simulations based on the<br />
Poster Presentations<br />
Monday 5 December - Session 2<br />
network topology and thermodynamic constraints<br />
were realized to screen for potential metabolite<br />
candidates. Briefly, the approach attempts to find<br />
a set <strong>of</strong> reactions that, if each <strong>of</strong> their direction is<br />
known, will fix the direction <strong>of</strong> other reversible<br />
reactions; metabolites linked to these reactions are<br />
therefore considered most useful to unravel the<br />
directions <strong>of</strong> the initially reversible reactions.<br />
In conclusion, thermodynamics can be<br />
incorporated with a multi-compartment metabolic<br />
network to identify new irreversibilities that are<br />
essential to further constrain the metabolic model.<br />
However, our simulations revealed that a large<br />
number <strong>of</strong> metabolites still need to be measured,<br />
possibly attributed to the fact that most metabolic<br />
reactions can operate independently and are<br />
highly redundant.<br />
P47<br />
Diagnostics for Orbital and<br />
Wavefunction Quality<br />
Laura K McKemmish 1 *, Jia Deng1 and<br />
<strong>Peter</strong> Gill1 1 Research School <strong>of</strong> Chemistry, Australian National <strong>University</strong>,<br />
Science Road, Acton 2601 ACT Australia<br />
* laura.mckemmish@gmail.com<br />
Abstract<br />
As computer power increases, more chemistry is<br />
done on computers, rather than in a laboratory,<br />
because it is safer, cheaper and faster. Yet black<br />
box approaches for computational chemistry are<br />
limited by the lack <strong>of</strong> a universal method <strong>of</strong><br />
quantifying a wavefunction’s accuracy. Here, we<br />
propose a paradigm for a diagnostic that<br />
quantifies how well an approximate wavefunction<br />
satisfies its Schrodinger equation (SE) independent<br />
<strong>of</strong> any external input. This diagnostic is the angle in<br />
Hilbert space, using some inner product metric,<br />
between and ; an angle <strong>of</strong> zero implies that the<br />
wavefunction satisfies its SE exactly, while higher<br />
angles imply increasingly low quality<br />
wavefunctions. A threshold, say 0.05, can be<br />
selected for desired accuracy based on the<br />
calculation’s purpose.<br />
A useful functional intermediate step is to quantify<br />
how well an orbital satisfies its Hartree-Fock<br />
133
Poster Presentations<br />
Monday 5 December - Session 2<br />
equation[1]. We investigate the effect <strong>of</strong> different<br />
inner product metrics (overlap, Coulomb and<br />
Gaussian). We conclude that the inner product<br />
metric is unsuitable due to its high sensitivity to<br />
electron-nuclear cusp inaccuracies. Furthermore,<br />
despite the relative ease <strong>of</strong> the required molecular<br />
integrals for the Gaussian inner product metric,<br />
this metric suffers from fatal errors in giving very<br />
good scores to extremely bad orbitals for the<br />
hydrogen-like atoms. Therefore, the most suitable<br />
metric currently is the Coulomb metric. However,<br />
some <strong>of</strong> the required molecular integrals have not<br />
been found analytically due to the complicating<br />
presence <strong>of</strong> two Coulomb operators; however,<br />
one- and two-dimensional numeric integrals can<br />
be found through a slight alteration <strong>of</strong> the method<br />
presented in King[2].<br />
In defining the wavefunction diagnostic, we make<br />
slight alterations to the most obvious definition to<br />
introduce size-intensivity to the diagnostic based<br />
on a toy model. We then look at finding this<br />
wavefunction quality diagnostic for the most<br />
straightforward wavefunctions consisting <strong>of</strong> a sum<br />
<strong>of</strong> Slater determinants in a Gaussian basis<br />
function; this will allow Hartree-Fock (HF) and<br />
CI-based wavefunctions to be analysed. In doing<br />
so, we note that a four-dimensional numeric<br />
integration is currently required. We present results<br />
for a variety <strong>of</strong> small test cases and compare the<br />
orbital and wavefunction diagnostic for HF results.<br />
[1] Deng, J.; Gilbert, A. T. B.; Gill, P. M. W., Can. J. Chem., 2010,<br />
88, 754-758<br />
[2] Komornicki, A.; King, H. F., J. Chem. Phys., 2011, 134, 244115<br />
P48<br />
Far-Infrared spectroscopy <strong>of</strong> water<br />
aerosols using synchrotron radiation<br />
Chris Medcraft1,2 , Evan G. Robertson 3 ,<br />
Dominque R.T. Appadoo2 , Sigurd Bauerecker 4 and<br />
Don McNaughton1 1 School <strong>of</strong> Chemistry, Monash <strong>University</strong>, Victoria 3800<br />
Australia, chris.medcraft@monash.edu, don.mcnaughton@<br />
monash.edu<br />
2 Australian Synchrotron, 800 Blackburn Rd, Clayton, 3168,<br />
Victoria. Australia, dominque.appadoo@synchrotron.org.au<br />
3 Department <strong>of</strong> Chemistry, La Trobe <strong>University</strong>, Victoria 3083<br />
Australia, E.Robertson@latrobe.edu.au<br />
4 Institut für Physikalische und Theoretische Chemie, Technische<br />
Universität Braunschweig, Hans-Sommer-Strasse 10, D-38106<br />
Braunschweig, Germany<br />
134<br />
Abstract<br />
An Enclosive Flow Cooling (EFC) Cell has been<br />
coupled to the Bruker IFS125HR spectrometer on<br />
the High Resolution Infrared Beamline at the<br />
Australian Synchrotron. This cell has been<br />
modified from the design <strong>of</strong> Bauerecker[1] to<br />
extend its optical range into the far-infrared where<br />
the synchrotron source has the greatest<br />
advantage over thermal sources. The coupling <strong>of</strong><br />
the EFC cell to a synchrotron source provides us<br />
with a unique ability to perform a range <strong>of</strong> different<br />
experiments in the gas and condensed phases<br />
over a wide range <strong>of</strong> frequencies. We have used<br />
this capability to probe the intermolecular<br />
vibrations <strong>of</strong> water aerosols centred around 240<br />
cm-1 over a wide range <strong>of</strong> particle formation<br />
temperatures (see figure 1) and pressures (10-500<br />
mbar). These bands are <strong>of</strong> great interest to<br />
astronomers, providing an indication <strong>of</strong> both the<br />
temperature <strong>of</strong> an observed ice feature, and<br />
whether it arises from amorphous or crystalline ice<br />
(which in turn has implications for solar system<br />
formation). Significantly, we notice differences<br />
between the band positions our aerosol spectra<br />
and the previously published spectra <strong>of</strong> based on<br />
thin films. We attribute these differences to the<br />
absence <strong>of</strong> scattering (from the real part <strong>of</strong> the<br />
refractive index <strong>of</strong> water ice) in our spectra.<br />
Experiments were also performed in the midinfrared<br />
as a means <strong>of</strong> comparison with previous<br />
experiments.<br />
References<br />
[1] S. Bauerecker, M. Taraschewski, C. Weitkamp and H.K.<br />
Cammenga, Rev. Sci. Instr., 2001, 72, 3946.<br />
[2] C. Medcraft, E.G. Robertson, C.D. Thompson, S. Bauerecker<br />
and D. McNaughton, PCCP, 2009, 11, 7848-7852<br />
[3] E.G. Robertson, C. Medcraft, L. Puskar, R. Tuckermann, C.D.<br />
Thompson, S. Bauerecker and D. McNaughton, PCCP, 2009,<br />
11, 7853-7860
P49<br />
Quantum Chemical Studies <strong>of</strong> the<br />
Catalytic Mechanism <strong>of</strong> E3<br />
Hydrolysis <strong>of</strong> Organophosphates in<br />
the Australian Sheep Blowfly L.<br />
cuprina<br />
Tamara Meirelles 1 , Junming Ho1 , Michelle<br />
Coote1 , Colin Jackson1 *<br />
1 Research School <strong>of</strong> Chemistry, Australian National <strong>University</strong><br />
ACT 0200<br />
*cjackson@rsc.anu.edu.au<br />
Abstract<br />
Inhibition <strong>of</strong> acetylcholinesterase-catalyzed<br />
acetylcholine hydrolysis at nerve synapses by<br />
organophosphates has led to their use as<br />
insecticides and chemical warfare agents.<br />
Through a single point mutation, the sheep blowfly<br />
Lucilia cuprina has evolved a new<br />
organophosphate hydrolase, E3, from an existing<br />
carboxylesterase. However, the exact mechanism<br />
by which this active site mutation can confer this<br />
change in activity is currently unknown. To<br />
investigate the basis for the altered catalytic<br />
mechanism <strong>of</strong> E3 in detail, we have performed<br />
high-level quantum chemistry calculations on the<br />
reaction mechanism as catalyzed by the original,<br />
and mutant, enzymes. Our results suggest that a<br />
Gly-Asp mutation within the active site introduces<br />
a general base that can activate a water molecule<br />
as part <strong>of</strong> a new catalytic mechanism. These<br />
results provide a framework that can be used as<br />
the basis for future work that will attempt to<br />
identify further mutations to generate a more<br />
efficient catalyst that can be used to detoxify<br />
organophosphate in agricultural and medical<br />
applications.<br />
Poster Presentations<br />
Monday 5 December - Session 2<br />
P50<br />
Ab Initio Folding <strong>of</strong> Helical Peptides<br />
Using Adaptive Hydrogen Bond-<br />
Specific Charge Scheme<br />
Siti Raudah Mohamed Lazim1 , Dawei<br />
Zhang2 Division <strong>of</strong> Chemistry and Biological Chemistry, School <strong>of</strong> Physical<br />
and Mathematical Sciences, Nanyang Technological <strong>University</strong>, 21<br />
Nanyang Link, Singapore, 637371,<br />
1 SITI13@e.ntu.edu.sg<br />
2 zhangdw@ntu.edu.sg<br />
Abstract<br />
Adaptive hydrogen bond-specific charge (HBC)<br />
scheme is a newly developed on-the-fly charge<br />
fitting that relays a relatively accurate electrostatic<br />
polarization effect for hydrogen bonds established<br />
during the theoretical folding <strong>of</strong> proteins.<br />
Considering the inherent nature <strong>of</strong> protein folding<br />
which necessitate continuous formation and<br />
interruption <strong>of</strong> hydrogen bonds, on-the-fly charge<br />
fitting <strong>of</strong> amino acids involved in hydrogen bonding<br />
through fragment quantum mechanical<br />
calculations <strong>of</strong>fers an accurate description <strong>of</strong> the<br />
electronic distribution <strong>of</strong> the protein during the<br />
folding process. Here, we report the ab initio<br />
folding <strong>of</strong> four different helical peptides viz. b30-82<br />
domain <strong>of</strong> Subunit b <strong>of</strong> Escherichia Coli F1Fo<br />
adenosine triphosphate synthase (2KHK),<br />
transmembrane helix 6 <strong>of</strong> the human cannabinoid<br />
receptor-2 (2KI9), N36 and C34 domain <strong>of</strong> gp41<br />
from HIV-1 envelope glycoprotein through<br />
conventional molecular dynamics simulation at<br />
room temperature using implicit continuum<br />
solvation model. Employing adaptive HBC<br />
scheme, the effective folding <strong>of</strong> the four peptides<br />
were observed with best Cα-RMSD with reference<br />
to experimental structures determined to be 0.69<br />
Å for 2KHK, 2.97 Å for 2KI9, 0.52 Å for N36 and<br />
0.66 Å for C34. The folding pathways <strong>of</strong> the<br />
proteins folded in this work were also elaborated<br />
through detailed analyses encompassing<br />
clustering, secondary structure assignment and<br />
folding landscapes. These analyses showcased<br />
the continuous growth <strong>of</strong> α-helix from the<br />
nucleation sites formed along the peptide which<br />
concurs with helix-coil transition model which<br />
describes the first turn <strong>of</strong> helix as the nucleus<br />
135
Poster Presentations<br />
Monday 5 December - Session 2<br />
acting as a stable infrastructure obliging the<br />
development <strong>of</strong> consecutive turns to form a fully<br />
compact α-helical protein.<br />
P51<br />
Amyloid-like fibrillization and the<br />
structure <strong>of</strong> glucagon in the<br />
presence <strong>of</strong> anionic bicelles<br />
Ayano Momose 1 , Izumi Yamane 1 , Hideki<br />
Fujita 1 , Eri Yoshimoto 1 , Izuru Kawamura1 ,<br />
Marc-Antoine Sani2 , Frances Separovic2 , Akira<br />
Naito1 1 Graduate School <strong>of</strong> Engineering, Yokohama National <strong>University</strong>,<br />
Hodogaya-ku, Yokohama 240-8501, Japan naito@ynu.ac.jp<br />
2 School <strong>of</strong> Chemistry, Bio21 Institute, <strong>University</strong> <strong>of</strong> Melbourne,<br />
VIC 3010<br />
Abstract<br />
Glucagon is a 29-residue peptide hormone<br />
secreted by α-cells <strong>of</strong> pancreatic islet cells when<br />
blood glucose level is low and promotes resolution<br />
<strong>of</strong> glycogen in liver to increase the level. Glucagon<br />
easily dissolves in acidic solvent and forms<br />
amyloid-like fibrils. In the case <strong>of</strong> amyloidogenic<br />
proteins, when the monomers change to fibrils, the<br />
secondary structure changes mainly from α-helix<br />
to β-sheet. After glucagon changes to β-sheet<br />
structure, it becomes cytotoxic. The detailed<br />
structures and mechanism <strong>of</strong> fibrillization is not<br />
well understood. In this study, the structure and<br />
fibril formation <strong>of</strong> glucagon in acetic acid solution<br />
(pH 3.3) and the effect <strong>of</strong> neutral and anionic<br />
phospholipids (bicelles) was observed using high<br />
resolution solid-state 13C NMR and TEM.<br />
The fibrillization kinetics was revealed using a<br />
two-step autocatalytic reaction model composed<br />
<strong>of</strong> fibril nucleation (rate constant <strong>of</strong> k1) and fibril<br />
extension reaction (rate constant <strong>of</strong> k2) by means<br />
<strong>of</strong> 13C solid-state NMR. We observed signal<br />
intensity change <strong>of</strong> glucagon fibrils under 5<br />
different conditions: in solution, in presence <strong>of</strong><br />
neutral, 10%, 15% and 25% anionic bicelles. In the<br />
presence <strong>of</strong> bicelles, the first reaction was faster<br />
but the second reaction was slower than in<br />
solution. It is conceivable that in the presence <strong>of</strong><br />
bicelles, peptides interacted and concentrated on<br />
the surface and, therefore, the first reaction<br />
136<br />
(nucleation) was accelerated but the second<br />
reaction (fibril elongation) was delayed. In addition,<br />
the higher the concentration <strong>of</strong> negatively charged<br />
lipids in the bicelles, the faster was the nucleation<br />
and the slower the fibril elongation. This is due to<br />
the electrostatic interaction between glucagon and<br />
the negatively charged lipids. Glucagon has a<br />
charge <strong>of</strong> +1 for the entire peptide and, under low<br />
pH conditions, the positive charge is emphasized.<br />
Thus we conclude that the bicelles accelerated the<br />
nucleation and decelerated the fibril elongation <strong>of</strong><br />
glucagon. This tendency was strengthened by<br />
increasing the ratio <strong>of</strong> negatively charged lipids in<br />
the bicelles.<br />
P52<br />
Modelling Molecular Response in<br />
Anisotropic Electric Fields<br />
Michael Morris1 , Meredith Jordan2 1 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney, NSW, 2006,<br />
morris_m@chem.usyd.edu.au<br />
2 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney, NSW, 2006, mjtj@<br />
chem.usyd.edu.au<br />
Abstract<br />
It has long been established that long-range<br />
interactions play a key role in molecular processes.<br />
For example, protein-ligand association is strongly<br />
influenced by long-range interactions. Despite their<br />
importance, it is <strong>of</strong>ten difficult to determine how<br />
molecular structure is affected by changes in<br />
these long-range interactions. In such<br />
circumstances, it is useful to model the long-range<br />
interaction using a classical power series<br />
expanded in the field, F, and field gradient, ÑF, <strong>of</strong><br />
an interacting species, with coefficients<br />
determined at zero-field. The accuracy and limits<br />
<strong>of</strong> applicability <strong>of</strong> this expansion are investigated<br />
for a small model system, ClH:NH3, which<br />
undergoes significant spectroscopic and structural<br />
change in typical atomic-scale fields. The ClH:NH3<br />
structure and vibrational frequencies are<br />
determined at first and second order in F and ÑF<br />
for a variety <strong>of</strong> uniform and anisotropic external<br />
fields. Spectroscopic and structural characteristics<br />
are predicted, at first order in F and ÑF, to within<br />
experimental error, with lower errors being<br />
observed at second order. The accuracy <strong>of</strong> the
expansion is found to reduce as the magnitude <strong>of</strong><br />
F and ÑF increases or as the energy level<br />
considered increases, although even at field<br />
strengths large enough to cause dramatic<br />
structural change in the complex, both the<br />
structure and vibrational frequencies are effectively<br />
quantitatively predicted using only terms linear in F<br />
and ÑF. These results suggest that knowledge <strong>of</strong><br />
the zero-field molecular potential energy and<br />
dipole and quadrupole moment surfaces may be<br />
sufficient to accurately model long-range<br />
interactions in a wide range <strong>of</strong> circumstances.<br />
P53<br />
Examining the Electronic<br />
Interactions <strong>of</strong> Ligated Copper<br />
Nanoparticles Generated by Laser<br />
Ablation Synthesis in Solution<br />
(LASiS)<br />
Ashley Mulder1 , Mark Buntine1 , Aidon<br />
Slaney1 , Sean Long1 , Max Massi1 , Franca Jones1 ,<br />
Mark Ogden1 1 Department <strong>of</strong> Chemistry, Curtin <strong>University</strong>, GPO BOX U1987,<br />
Perth WA 6845<br />
Abstract<br />
We have successfully created copper<br />
nanoparticles (CuNP’s) in aqueous solutions using<br />
the LASiS method (Laser Ablation Synthesis in<br />
Solution). The fundamental frequency (1064 nm) <strong>of</strong><br />
a Nd:YAG laser was focused onto a copper plate in<br />
a glass vial with approximately 15 mL <strong>of</strong> solution.<br />
The copper plate was ablated for 30 minutes<br />
during which time the solution took on a green/<br />
yellow colour, characteristic <strong>of</strong> the CuNP surface<br />
plasmon resonance (SPR) transition. Various<br />
nitrogen containing ligands were added to the<br />
solutions prior to laser ablation, to act as capping<br />
ligands to protect the copper nanoparticles from<br />
oxidation. All solutions were made up to 10-4<br />
molar concentration and the ligands used were: (1)<br />
1,10-phenanthroline, (2) 2,2’-bipyridine, (3)<br />
4,4’-bipyridine, (4) 5-(2-pyridyl)-1H-tetrazole, (5)<br />
5-(4-pyridyl)-1H-tetrazole, (6) 5-(2-pyridyl)-2-(tbutyl)-1H-tetrazole,<br />
(7) 5-(4-pyridyl)-2-(t-butyl)-1Htetrazole,<br />
(8) 5-phenyl-1H-tetrazole and (9)<br />
5-methyl-1H-tetrazole. These solutions post<br />
Poster Presentations<br />
Monday 5 December - Session 2<br />
ablation all took on a blue/green through to green/<br />
yellow colour. These solutions were analysed by<br />
TEM imaging to determine the particle size<br />
distributions and by UV-Vis spectrometry to<br />
ascertain the position <strong>of</strong> each solution’s SPR and<br />
to study the kinetics <strong>of</strong> laser-based CuNP<br />
formation and stability.<br />
P54<br />
Patch Fluorimetry to Measure the<br />
Membrane Tension Required to Gate<br />
Mechanosensitive Ion Channels<br />
Takeshi Nomura1 , Charles Cranfield1,2 , Boris<br />
Martinac1,2 1 Victor Chang Cardiac Research Institute, Lowy-Packer Building,<br />
405 Liverpool St, Darlinghurst, 2010.<br />
2 St Vincent’s Clinical School, The <strong>University</strong> <strong>of</strong> New South<br />
Wales, de Lacy, Victoria St, Darlinghurst, 2010<br />
T.nomura@victorchang.edu.au; C.cranfield@victorchang.edu.au;<br />
B.martinac@victorchang.edu.au<br />
Abstract<br />
We report here the ability to measure the<br />
membrane bilayer tension required to gate<br />
mechanosensitive ion channels in a patch pipette<br />
by using patch fluorimetry. By reconstituting<br />
mechanosensitive ion channels in azolectin<br />
liposomes that incorporate 0.1-1% 1,2-Dioleoyl-sn-<br />
Glycero-3-Phosphoethanolamine-N-Lissamine<br />
Rhodamine B Sulfonyl (PE-Rhod) it is possible to<br />
visualise the patch in the pipette in situ using<br />
fluorescence confocal microscopy. To open<br />
mechanosensitive ion channels, such as the<br />
bacterial mechanosensitive channels <strong>of</strong> large and<br />
small conductance (MscL and MscS), a<br />
measurable negative hydrostatic pressure is<br />
applied to a patch. By a simple derivation <strong>of</strong><br />
Laplace’s law, the tension <strong>of</strong> the membrane at the<br />
threshold pressure for channel opening can be<br />
calculated according to the formula T=PR/2,<br />
where T is the tension <strong>of</strong> the membrane (measured<br />
in N/m), P is the pressure applied to stretch the<br />
patch membrane (Pa) and R is the radius <strong>of</strong> the<br />
curvature <strong>of</strong> the patch (m)[1]. The use <strong>of</strong><br />
fluorescence confocal microscopy enables a clear<br />
visualisation and measurement <strong>of</strong> the radius <strong>of</strong><br />
curvature inside the patch when stretched. Here<br />
we demonstrate how this patch fluorimetry has<br />
137
Poster Presentations<br />
Monday 5 December - Session 2<br />
provided an accurate and quantifiable method to<br />
measure the influence <strong>of</strong> amphipaths and other<br />
lipophilic compounds on lipid-ion channel<br />
interactions in situ.<br />
[1] Sokabe, M., Sachs, F., and Jing, Z. Q., Quantitative video<br />
microscopy <strong>of</strong> patch clamped membranes stress, strain,<br />
capacitance, and stretch channel activation. Biophysical<br />
Journal, 1991. 59(3): p. 722-728.<br />
Supported by the NHMRC Grant 635525.<br />
P55<br />
Structural characterization <strong>of</strong><br />
triacylglycerols by radical directed<br />
dissociation<br />
Huong T. Pham 1 , Stephen J. Blanksby 2 ,<br />
Gavin E. Reid 3<br />
1 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>, Australia,<br />
thp658@uow.edu.au<br />
2 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>, Australia,<br />
blanksby@uow.edu.au<br />
3 Department <strong>of</strong> Chemistry, Michigan State <strong>University</strong>, United<br />
States, reid@chemistry.msu.edu<br />
Abstract<br />
Deep-fat frying is one <strong>of</strong> the most common uses<br />
for edible vegetable oils in the food industry. Oil<br />
used for deep-fat frying is subjected to high<br />
temperatures, moisture from foods, and migration<br />
<strong>of</strong> other compounds from foods that can act as<br />
pro-oxidants.[1] Unsaturated triacylglycerols are<br />
the major components in such oils are likely<br />
participating in dimerization and higher order<br />
polymerization during this thermal procedure.<br />
Such bio-polymerization processes are proposed<br />
to occur via free radical mechanisms although<br />
these are well understood due to the molecular<br />
complexity <strong>of</strong> these species. Radical directed<br />
dissociation (RDD) [2] is a mass spectrometric<br />
technique that can be used to selectively initiate<br />
and study radical reactions within isolated<br />
molecules or simple ensembles within the gas<br />
phase. In this approach a free radical initiator is<br />
appended to an adducting agent (e.g.,<br />
4-iodoaniline or 4-iodobenzoic acid) and<br />
complexed with a lipid(s) by electrospray<br />
ionisation. Such an approach was employed to<br />
generate adduct ions <strong>of</strong> triacylglycerols (TAGs) in a<br />
linear ion-trap mass spectrometer with<br />
subsequent photoactivation <strong>of</strong> the mass-selected<br />
138<br />
complex at 266 nm (Nd:YAG, Continuum Minilite II)<br />
leading to homolysis <strong>of</strong> the carbon-iodine bond.<br />
Subsequent collision induced dissociation (CID) <strong>of</strong><br />
the nascent radical ions gives rise to a radical<br />
directed dissociation mass spectrum that provides<br />
a wealth <strong>of</strong> structurally informative fragmentation.<br />
This methodology is complementary to<br />
conventional approaches to structure elucidation<br />
<strong>of</strong> complex lipids in mass spectrometry and will<br />
assist in the characterisation <strong>of</strong> lipid dimers (and<br />
higher polymers) arising from thermolysis <strong>of</strong><br />
vegetable oils. In addition, non-covalent<br />
complexes <strong>of</strong> a single adducting agent with<br />
multiple lipids have been generated with the<br />
resulting RDD spectra providing evidence for<br />
cross-linking <strong>of</strong> the lipids. Such spectra may<br />
provide clues as to the chemical pathways<br />
involved in free radical polymerisation <strong>of</strong> lipids.<br />
[1] Winkler, J. K.; Warner, K. Eur. J. Lipid Sci. Technol. 2008, 110,<br />
1068.<br />
[2] Ly, T.; Julian, R. R. J. Am. Chem. Soc. 2008, 130, 351.<br />
P56<br />
Organic Monolayers for Water<br />
Evaporation Suppression: A<br />
Molecular Dynamic Study<br />
Michael Plazzer, George Yiapanis, Irene<br />
Yarovsky 1<br />
1 Applied Physics, School <strong>of</strong> Applied Sciences, RMIT <strong>University</strong>,<br />
Melbourne 3000 irene.yarovsky@rmit.edu.au<br />
Abstract<br />
Self assembled monolayers (SAM) have been <strong>of</strong><br />
interest to industry and medical science for their<br />
novel properties <strong>of</strong> self-organisation, wetting and<br />
selective permeation. The latter, is particularly<br />
important for applications pertaining to chemical<br />
and biological sensors1 , and to the facilitation <strong>of</strong><br />
gas transport2 and evaporation resistance.<br />
However, when monolayers are used for external<br />
applications, issues <strong>of</strong> stability and anchoring to<br />
water have been shown to arise3 , affecting the<br />
long-term functionality <strong>of</strong> the monolayers. To<br />
elucidate the precise properties required for<br />
long-term efficacy, one requires a thorough<br />
understanding <strong>of</strong> the nature and strength <strong>of</strong>
interactions occurring at the monolayer / fluid<br />
interface.<br />
In this research, we use classical molecular<br />
dynamics to investigate a range <strong>of</strong> amphiphilic<br />
molecules forming monolayers on the surface <strong>of</strong><br />
water. The molecules comprise <strong>of</strong> alkyl chains <strong>of</strong><br />
18 carbons with different headgroups including<br />
hydroxyl, glycol, methyl and ether groups. We<br />
demonstrate how surface pressure isotherms can<br />
be an indicator <strong>of</strong> the robustness <strong>of</strong> the<br />
monolayers, that is, their ability to maintain stability<br />
over a range <strong>of</strong> surface pressures. We show that<br />
hydrogen bonding between the monolayer<br />
headgroups and the interfacial water layer, as well<br />
as between the headgroups themselves, play a<br />
primary role in the anchoring and stability <strong>of</strong> the<br />
monolayers, and that a balance between the two<br />
leads to optimum water evaporation suppressing<br />
performance. We conclude that the choice <strong>of</strong><br />
headgroup is the primary determining factor <strong>of</strong> the<br />
monolayer’s performance.<br />
1. N. Hambdi, J. Wang, H.G. Monbouquette, Journal <strong>of</strong><br />
Electroanalytical Chemistry 2005, 581, 2.<br />
2. J.H. Kim, S.Y. Ha, S.Y. Nam, J.W. Rhim, K.H. Paek and Y.M.<br />
Lee, J. Membr. Sci. 2001, 186<br />
3 Solomon et al. Environmental Health. 2007, 7, 3.<br />
Poster Presentations<br />
Monday 5 December - Session 2<br />
P57<br />
Langevin dynamics modelling <strong>of</strong><br />
water diffusion in anisotropic<br />
biophysical structures.<br />
Sean Powell1 , Konstantin Momot1 1 Discipline <strong>of</strong> Physics, Queensland <strong>University</strong> <strong>of</strong> Technology, GPO<br />
Box 2434, Brisbane, QLD, 4001, sean.powell@qut.edu.au<br />
Abstract<br />
Diffusion Tensor Imaging (DTI) is a form <strong>of</strong><br />
Magnetic Resonance Imaging (MRI) that measures<br />
the anisotropic diffusion <strong>of</strong> water molecules. This<br />
non-invasive imaging technique enjoys success in<br />
medical research fields such as brain research [1,<br />
2], studies <strong>of</strong> articular cartilage structure and<br />
biomechanics [3], and clinical diagnosis <strong>of</strong> medical<br />
conditions such as stroke, diffuse axonal injury,<br />
multiple sclerosis, Alzheimer’s disease and<br />
tumours. The utility <strong>of</strong> DTI for investigating the<br />
microstructure <strong>of</strong> some ordered tissues is<br />
presently constrained by the lack <strong>of</strong> models for<br />
quantitative morphological interpretation <strong>of</strong> the<br />
diffusion tensor.<br />
In this study, we developed course-grained<br />
molecular models <strong>of</strong> water dynamics in articular<br />
cartilage. The aim was to relate diffusion<br />
measurements to microstructural characteristics<br />
such as collagen fibre volume fraction and fibre<br />
alignment. In articular cartilage, the displacement<br />
<strong>of</strong> diffusing water molecules is restricted in some<br />
directions more than others by the collagen fibres.<br />
DTI measures this RMS displacement anisotropy<br />
to generate three-dimensional maps representing<br />
the anisotropy <strong>of</strong> the average collagen alignment<br />
on the length scale 50-200μm.<br />
A c++ computer model was developed and<br />
simulations <strong>of</strong> water diffusion in various articular<br />
cartilage structures were performed. The degree<br />
and direction <strong>of</strong> diffusion anisotropy was then<br />
calculated for a range <strong>of</strong> collagen volume fractions<br />
and fibre alignment angles. The simulations were<br />
run on a 600 CPU SGI Altix supercomputer and<br />
took around 3-5 days per structure to process.<br />
Quantitative models were then built to relate the<br />
diffusion tensor to articular cartilage<br />
microstructure. This study is the first to quantify<br />
the effects <strong>of</strong> different collagen fibre alignment<br />
139
Poster Presentations<br />
Monday 5 December - Session 2<br />
angles on the diffusion tensor. The use <strong>of</strong> Langevin<br />
dynamics enabled chemically realistic modelling <strong>of</strong><br />
the water-fibre interactions and allows for the<br />
inclusion <strong>of</strong> proteoglycans.<br />
The use <strong>of</strong> these models as an interpretive tool will<br />
improve the utility <strong>of</strong> diffusion tensor imaging for<br />
clinical diagnosis <strong>of</strong> diseases <strong>of</strong> articular cartilage<br />
and assisting in the development <strong>of</strong> an improved<br />
understanding <strong>of</strong> its biomechanical properties.<br />
This research can be expanded to develop<br />
quantitative models describing the diffusion <strong>of</strong><br />
water in a larger range <strong>of</strong> ordered biophysical<br />
tissue such as tendons and muscle.<br />
References<br />
[1] Behrens TEJ, Berg HJ, Jbabdi S, Rushworth MFS, Woolrich<br />
MW. Neuroimage 2007; 34:144-155<br />
[2] Tournier JD, Yeh CH, Calamante F, Cho KH, Connelly A, Lin CP.<br />
Neuroimage 2008; 42:617-625.<br />
[3] de Visser SK, Bowden JC, Wentrup-Byrne E, Rintoul L,<br />
Bostrom T, Pope JM, Momot KI. Osteoarthr Cartilage 2008;<br />
16:689-697.<br />
P58<br />
Photodissociation Dynamics <strong>of</strong><br />
Acetaldehyde at 308 nm: A<br />
Comparison <strong>of</strong> Experimental Studies<br />
and a Classical Trajectory Study <strong>of</strong><br />
the Transition State Mechanism<br />
Mitchell S. Quinn1 , Gabi de Wit2 , Scott A.<br />
Reid3 , B. Klaas Nauta4 , Alan T. Maccarone 5 , Scott<br />
H. Kable6 , Meredith J. T. Jordan7 1 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney, New South Wales<br />
2006, m.quinn@chem.usyd.edu.au<br />
2 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney, New South Wales<br />
2006, g.dewit@chem.usyd.edu.au<br />
3 Department <strong>of</strong> Chemistry, Marquette <strong>University</strong>, Milwaukee, WI,<br />
USA, scott.reid@marquette.edu<br />
4 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney, New South Wales<br />
2006, k.nauta@chem.usyd.edu.au<br />
5 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney, New South Wales<br />
2006, alanmac@uow.edu.au<br />
6 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney, New South Wales<br />
2006, s.kable@chem.usyd.edu.au<br />
7 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Sydney, New South Wales<br />
2006, mjtj@chem.usyd.edu.au<br />
Abstract<br />
The recent discovery <strong>of</strong> an alternate pathway to<br />
molecular products, in a phenomenon known as<br />
‘roaming’, has led to investigation <strong>of</strong> several<br />
140<br />
organic molecules that are thought to exhibit these<br />
characteristics. The first two experimental systems<br />
where the roaming mechanism was identified were<br />
formaldehyde (H2CO) [1] and acetaldehyde<br />
(CH3CHO) [2]. In these systems, the H atom or<br />
CH3 group “roams” in the radical dissociation<br />
channel and, in a self-abstraction process,<br />
abstracts an H atom from the other moiety. The<br />
products <strong>of</strong> a roaming mechanism are typically<br />
rotationally and translationally cold, with high<br />
vibrational excitation <strong>of</strong> the molecule that contains<br />
the roaming fragment (i.e. H2 for H2CO and CH4<br />
for CH3CHO). Molecular products formed from<br />
more traditional “transition state” (TS) mechanisms<br />
have a large amount <strong>of</strong> kinetic energy; that is, the<br />
product molecules are translationally and<br />
rotationally hot. Armed with this knowledge<br />
chemists have since moved on to investigate<br />
roaming in about a dozen other reactions.<br />
This poster uses trajectories obtained on a<br />
reaction path potential energy surface (PES) to<br />
investigate the photodissociation <strong>of</strong> CH3CHO.<br />
These trajectories were restricted to a section <strong>of</strong><br />
the PES consisting <strong>of</strong> only the traditional TS<br />
mechanism. The trajectory results were compared<br />
to recent experiments performed by the authors<br />
(see the poster presented by Alan T. Maccarone)<br />
as well as detailed trajectory calculations on a full<br />
PES [3] and a reanalysis <strong>of</strong> previous experimental<br />
results [2].<br />
Furthermore the poster will outline the construction<br />
<strong>of</strong> a zero point energy (ZPE) corrected PES for<br />
H2CO. Due to the difficulty in including the ZPE in<br />
classical and quasi-classical trajectories, and the<br />
current impracticality <strong>of</strong> running full quantum<br />
mechanical trajectories, we propose a method that<br />
accounts for the ZPE when running classical<br />
trajectories. A first order interpolated ZPE surface<br />
is added to a second order modified Shepard<br />
interpolated PES to create the corrected surface.<br />
The surface allows for full classical trajectories to<br />
include this important quantum mechanical<br />
property through a well-defined and systematic<br />
method.<br />
References:<br />
[1] D. Townsend, S. S. Lahankar, S. K. Lee, S. D. Chambreau, A.<br />
G. Suits, X. Zang, J. Rheinecker, L. B. Harding, J. Bowman,<br />
Science 306 1158 (2004)<br />
[2] P. L. Houston, S. H. Kable, Proc. Natl. Acad. Sci. USA 103<br />
16079 (2006)<br />
[3] B. R. Heazlewood, M. J. T. Jordan, S. H. Kable, T. M. Selby, D.<br />
L. Osborn, et al., Proc. Natl. Acad. Sci. USA 105 12719 (2008)
P59<br />
Alanine scan <strong>of</strong> an<br />
immunosuppressive peptide:<br />
Surface plasmon resonance analysis<br />
and structure-function relationships<br />
Laura Raguine1 , Marina Ali1 , Veronika<br />
Bender1 , Eve Diefenbach2 , Nicholas Manolios1 1 Department <strong>of</strong> Rheumatology, Westmead Hospital, Westmead,<br />
NSW 2145, Australia.<br />
2 Protein Production Facility, Westmead Millennium Institute,<br />
Westmead, NSW 2145, Australia.<br />
Abstract<br />
T-cell mediated autoimmune diseases arise due to<br />
the immune system’s inability to differentiate<br />
between “self” and “non-self” antigens. This leads<br />
to destruction and loss <strong>of</strong> function <strong>of</strong> the target<br />
organ or tissue. T-cell mediated diseases include<br />
rheumatoid arthritis and type I diabetes.<br />
Immunosuppression is required to prevent such<br />
diseases. Core peptide (CP) is an<br />
immunosuppressive peptide that has the potential<br />
to treat T-cell mediated diseases by inhibiting<br />
autoreactive T-cell activation. By using an alanine<br />
scan, the aim <strong>of</strong> this study was to examine the<br />
structure versus function relationship <strong>of</strong> the<br />
constituent amino acids within CP.<br />
CP and CP analogues were synthesised using<br />
solid phase peptide synthesis. CP analogues were<br />
produced by substituting single amino acids with<br />
alanine. Surface Plasmon Resonance (SPR) was<br />
used to assess binding <strong>of</strong> CP and its analogues to<br />
lipid membrane models. Antigen Presentation<br />
Assays (APA) were used to assess CP and its<br />
analogues ability to alter/inhibit cytokine<br />
production. Briefly, CP was introduced to an<br />
antigen specific T-cell culture (2B4 cells) in the<br />
presence <strong>of</strong> antigen. Supernatant from these<br />
activated T-cell cultures were collected after 24<br />
hours and cytokine levels in the supernatant were<br />
measured using a cytokine-dependent<br />
proliferation assay and flow cytometry. Cytokine<br />
levels were compared to a positive control.<br />
CP and 10 <strong>of</strong> its analogues have been<br />
synthesised, purified by high performance liquid<br />
chromatography and characterised by mass<br />
spectroscopy. Preliminary data using dimyristoyl<br />
Poster Presentations<br />
Monday 5 December - Session 2<br />
phosphatidylglycerol (DMPG) liposomes suggest<br />
the hydrophobic face between the two charged<br />
amino acids (arginine and lysine) are important in<br />
affecting binding affinity. A more complete<br />
description <strong>of</strong> the biological and biophysical data<br />
will be discussed at a later stage. Any correlations<br />
between binding in SPR and cytokine production<br />
in the APA will also be discussed.<br />
P60<br />
Affinity and selectivity <strong>of</strong> sea<br />
anemone toxin ShK for the Kv1.1,<br />
Kv1.2 and Kv1.3 channels from free<br />
energy simulations.<br />
M.H. Rashid and S. Kuyucak<br />
School <strong>of</strong> Physics, <strong>University</strong> <strong>of</strong> Sydney, NSW 2006, Australia,<br />
harun@physics.usyd.edu.au, serdar@physics.usyd.edu.au.<br />
Abstract<br />
The voltage gated potassium channel (Kv1.x) is an<br />
attractive target for autoimmune diseases[1].<br />
Potent and selective blockers <strong>of</strong> potassium<br />
channels are potential therapeutics for treating<br />
these diseases. Here we present molecular<br />
dynamics (MD) studies for binding <strong>of</strong> the ShK toxin<br />
from the sea anemone to the Kv1 channels, which<br />
inhibits Kv1.1 and Kv1.3 potently and blocks Kv1.2<br />
with a much lower affinity.<br />
Based on crystal structure <strong>of</strong> voltage-gated<br />
potassium channel Kv1.2 we have constructed a<br />
homology model <strong>of</strong> Kv1.1 and Kv1.3. Haddock has<br />
been used for the exploration <strong>of</strong> the<br />
conformational space and the determination <strong>of</strong><br />
ligand/protein contacts. We have run the MD<br />
simulation up to 10 ns for the channel-toxin<br />
complex in a solvated lipid bilayer environment.<br />
Several recognition and contact residues have<br />
been identified from the MD simulations <strong>of</strong> the<br />
complex. The most important is the K22 side<br />
chain, which enters the filter and forms contacts<br />
with the Y carbonyls in all Kv1 channels. Shk<br />
interact with Kv1.1 channel by Lys18/Glu353(C),<br />
Phe27/Gly376(A), Gln16/Glu353(B), Arg11/<br />
Asp361(B), Arg29/Glu353(D), Ser20/Tyr379(D)<br />
residue pairs, Kv1.2 channel by R29/E353(A), S19/<br />
T383(C), M21/Gln357(D) residue pairs and Arg11/<br />
141
Poster Presentations<br />
Monday 5 December - Session 2<br />
Asp402(A), Phe27/Gly401(C), Thr6/Asp402(D),<br />
Arg1/Glu373(C), His19/Ser378(B), Ser20/Gly401(B)<br />
Met21/Met403(C) residue pairs in Kv1.3 channel.<br />
We calculate the potential <strong>of</strong> mean force (PMF) for<br />
unbinding <strong>of</strong> ShK from the Kv1 channels using<br />
umbrella sampling MD simulation. The absolute<br />
binding energies determined form the PMF’s differ<br />
by about 1 kcal/mol from the experimental<br />
measurements [2].<br />
P61<br />
Large-scale fully ab initio<br />
calculations <strong>of</strong> ionic liquids using the<br />
Fragment Molecular Orbital<br />
approach<br />
Jason D. Rigby, Douglas R. MacFarlane,<br />
Ekaterina I. Izgorodina<br />
School <strong>of</strong> Chemistry, Monash <strong>University</strong>, Wellington Road, Victoria,<br />
3800, E-Mail: jason.rigby@monash.edu<br />
Abstract<br />
Ionic liquids are a new class <strong>of</strong> liquids and solvent<br />
based entirely on organic salts. They are finding a<br />
wide range <strong>of</strong> applications, including in a number<br />
<strong>of</strong> bio-science and biotechnology contexts. It has<br />
been shown in previous studies1 that the interplay<br />
between electrostatic and dispersion forces in<br />
ionic liquids is an important factor when predicting<br />
thermodynamic and transport properties.<br />
However, the extent to which these components<br />
impact on physical properties have not yet been<br />
studied in detail using a fully ab initio method. The<br />
main issue is that correlated levels <strong>of</strong> theory in<br />
general scale poorly with molecular size, thereby<br />
making the study <strong>of</strong> large clusters challenging.<br />
Demands on disk and RAM become excessive<br />
and typically extend beyond the capabilities <strong>of</strong><br />
most High Performance Computing clusters. The<br />
Fragment Molecular Orbital (FMO) approach2,3 is<br />
an attractive alternative, in which the system is<br />
divided into molecular fragments, which are<br />
relatively inexpensive to compute even at a<br />
correlated level <strong>of</strong> theory. We explored the FMO<br />
approach combined with the MP2 level <strong>of</strong> theory<br />
for a series <strong>of</strong> four archetypical ionic liquids,<br />
[NMe4][BF4], [C1mim][BF4], [C3mim][BF4] and<br />
[C4mim][BF4], built into clusters <strong>of</strong> 1, 2, 4 and 8<br />
142<br />
ion-pairs. In these calculations, individual ions<br />
were selected as fragments. The results were<br />
compared with full MP2 and it was found that the<br />
FMO-MP2 method produced excellent accuracy<br />
within 0.2 kJ mol-1 when a three-body correction<br />
was included. Significant improvements in<br />
computational time were observed, thus paving<br />
the way for fully ab initio large-scale calculations <strong>of</strong><br />
novel ionic liquids.<br />
References<br />
1. Bernard, U. L.; Izgorodina, E. I.; MacFarlane, D. R. The Journal<br />
<strong>of</strong> Physical Chemistry C 2010, 114, 20472<br />
2. Kitaura, L.; Ikeo, I.; Asada, T.; Nakano, T.; Uebayasi, M.<br />
Chemical Physics Letters 1999, 313, 701<br />
3. Fedorov, D.; Kitaura, K. The Journal <strong>of</strong> Chemical Physics 2004,<br />
120, 6832<br />
P62<br />
Structural studies <strong>of</strong> osmoregulatory<br />
ABC transporters<br />
Stephanie J. Ruiz12 , , Maaike Jansen, and<br />
Bert Poolman<br />
1 Department <strong>of</strong> Biochemistry, <strong>University</strong> <strong>of</strong> Groningen, The<br />
Netherlands<br />
2 s.j.ruiz@rug.nl<br />
Abstract<br />
Osmoregulatory ABC transporters protect<br />
bacterial cells against hyperosmotic stress by<br />
accumulating small, osmotically active compounds<br />
known as compatible solutes. Individual<br />
transporters have been shown to influence<br />
virulence in Staphylococcus aureus and Listeria<br />
monocytogenes through their ability to specifically<br />
import compatible solutes that are present at<br />
higher concentrations in tissue.<br />
OpuA from Lactococcus lactis is an<br />
osmoregulatory transporter that has been<br />
extensively studied due to its unusual architecture<br />
(1,2). All ABC transporters contain two intracellular<br />
ATP-binding domains (NBDs) and two multitransmembrane<br />
domains (TMDs). In prokaryotic<br />
ABC importers this core translocation complex, a<br />
homodimer comprised <strong>of</strong> four separate<br />
polypeptide chains, is joined by a substrate<br />
binding protein (SBP) that provides the main<br />
determinant <strong>of</strong> the transporter specificity. In most<br />
studied systems, this SBP is either free in the
periplasm or anchored to the cell membrane via a<br />
lipid moiety. In OpuA the TMD and SBP are fused,<br />
resulting in a homodimeric complex that contains<br />
two SBPs (or substrate-binding domains, SBDs)<br />
per transporter. This has interesting implications<br />
for the interactions between the various<br />
subdomains <strong>of</strong> the transporter complex.<br />
Lmo1421 and Lmo1422 from L. monocytogenes<br />
are homologous to L. lactis OpuAA (NBD) and<br />
OpuABC (TMD-SBD), respectively. They express<br />
well and form a stable homodimeric complex in<br />
various detergent solutions. Interestingly, despite<br />
high levels <strong>of</strong> sequence similarity to OpuA and<br />
other osmoregulatory ABC transporters,<br />
Lmo1421/1422 has previously been identified as a<br />
bile exclusion system (3).<br />
The C-terminus <strong>of</strong> Lmo1422, containing the<br />
putative substrate-binding domain, has been<br />
expressed and purified in isolation. An X-ray<br />
crystal structure <strong>of</strong> Lmo1422SBD was solved at a<br />
resolution <strong>of</strong> 1.6 Å and reveals an overall fold<br />
typical <strong>of</strong> class II substrate-binding proteins, with<br />
the protein in the open conformation. A<br />
comparison with homologous SBPs shows that<br />
residues involved in binding a range <strong>of</strong> compatible<br />
solutes are structurally conserved. Identification <strong>of</strong><br />
the Lmo1422SBD ligand(s) will aid in further<br />
biochemical characterization <strong>of</strong> this unusual<br />
transporter.<br />
1. Biemans-Oldehinkel, H. et al. (2006) Proc. Natl. Acad. Sci. USA,<br />
103, 10624-10629.<br />
2. Karasawa, A. et al. (2011) J Biol Chem., 286, 37280-37291.<br />
3. Sleator, R.D. et al. (2005) Mol. Microbiol., 55, 1183-1195.<br />
P63<br />
Determination <strong>of</strong> Threshold<br />
Dissociation Energies <strong>of</strong> Li+, Na+, K+<br />
and Cs+ Cationized Dimers <strong>of</strong><br />
3-Hydroxyflavone, 5-Hydroxyflavone<br />
and 5-Methoxyflavone by FTICR<br />
Mass Spectrometry and DFT<br />
Calculations<br />
Chowdhury Hasan Sarowar 1 , Michael<br />
Guilhaus2 , Gary David Willett3 , Grainne Moran4 1 School <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> New South Wales,<br />
Sydney, NSW 2052, Australia, c.h.sarowar@unsw.edu.au<br />
Poster Presentations<br />
Monday 5 December - Session 2<br />
2 Bioanalytical Mass Spectrometry Facility, The <strong>University</strong> <strong>of</strong> New<br />
South Wales, Sydney, NSW 2052, Australia<br />
3 School <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> New South Wales,<br />
Sydney, NSW 2052, Australia, g.willett@unsw.edu.au<br />
4 Mark Wainwright Analytical Centre, The <strong>University</strong> <strong>of</strong> New South<br />
Wales, Sydney, NSW 2052, Australia, g.moran@unsw.edu.au<br />
Abstract<br />
The threshold dissociation energies <strong>of</strong> alkali<br />
metal-flavonoid complexes <strong>of</strong> the type [2L+M]+ are<br />
determined in Fourier transform ion cyclotron<br />
resonance mass spectrometry. The flavonoids (L)<br />
examined are 3-hydroxyflavone, 5-hydroxyflavone<br />
and 5-methoxyflavone and the alkali metal cation<br />
(M) includes Li+, Na+, K+ and Cs+. The ions are<br />
generated in a high pressure external ion source<br />
and allowed to collide with target gas in the ion<br />
cyclotron resonance cell. The threshold<br />
dissociation energies for loss <strong>of</strong> one ligand from<br />
[2L+M]+ <strong>of</strong> 5-methoxyflavone follow the order Li+<br />
> Na+ > K+>>Cs+, and 3-hydroxyflavone and<br />
5-hydroxyflavone follow the order Li+ > Na+. For<br />
the same metal cation experiment, the<br />
5-methoxyflavone complex has the highest<br />
dissociation energy compared to 3-hydroxyflavone<br />
and 5-hydroxyflavone. Density functional theory<br />
calculations with a B3LYP functional and a<br />
6-31G(d,p) basis set were used to obtain the<br />
minimized energy geometric structures, vibrational<br />
frequencies, and rotational constants for [L+M]+<br />
and [2L+M]+ isomeric ions. The theoretical bond<br />
dissociation energies are then calculated and<br />
compared with the experimental bond dissociation<br />
energies. The agreement between theory and<br />
experiment is very good in all cases. This work has<br />
been extended to determine the threshold<br />
dissociation energies <strong>of</strong> Cu2+, Zn2+ and Pb2+<br />
cationized flavone complexes.<br />
143
Poster Presentations<br />
Monday 5 December - Session 2<br />
P64<br />
The impact <strong>of</strong> the ab initio/DFT<br />
method used for geometry<br />
optimisations on reaction enthalpies<br />
D. L. A. Scarborough, C. D. Thompson, E.<br />
I. Izgorodina<br />
School <strong>of</strong> Chemistry, Monash <strong>University</strong>, Victoria, Australia, 3800<br />
Email: David.Scarborough@monash.edu<br />
Abstract<br />
Proton-Transfer-Reaction Mass-Spectrometry<br />
(PTR MS), a technique that employs an ionmolecule<br />
reaction as its ionisation method, is<br />
becoming a common method for the online<br />
detection <strong>of</strong> atmospheric pollutants. However,<br />
molecular ion fragmentation can make the<br />
identification and quantification <strong>of</strong> species difficult.<br />
Quantum chemical calculations performed in the<br />
gas phase, can provide great insight into the<br />
reaction mechanisms, activation barriers and<br />
reaction enthalpies <strong>of</strong> ion-molecule reactions. A<br />
previous study by our group examined the<br />
fragmentation pathways <strong>of</strong> small, atmospheric<br />
pollutants such as ethanol and oxalic acid. As an<br />
example a fragmentation pathway <strong>of</strong> ethanol in a<br />
PTR MS system is shown below. It was found that<br />
in some cases the level <strong>of</strong> theory used for<br />
geometry optimisations affected the enthalpy<br />
re-calculated at higher levels <strong>of</strong> theory such as<br />
CCSD(T) and CBS-QB3 by up to 20 kJ mol-1<br />
when compared to experiment. In the current work<br />
the species involved in 30 ion-molecule reactions<br />
were optimised using M06, B2PLYP, MP2 and<br />
CCSD with the aug-cc-pVTZ basis set and B3LYP<br />
with the 6-31+G(d) and 6-311G(d,p) basis sets to<br />
determine which level <strong>of</strong> theory produces<br />
structures that give the most accurate enthalpies<br />
compared to CCSD(T) and CBS-QB3 calculations<br />
and experimental results.<br />
144<br />
P65<br />
Spectroscopy <strong>of</strong> resonancestabilized<br />
hydrocarbon radicals<br />
Tyler P. Troy, Nahid Chalyavi, Zijun Ge, Gerard<br />
D. O’Connor, Masakazi Nakajima, Neil J. Reilly,<br />
Damian L. Kokkin, Klaas Nauta, Scott H. Kable,<br />
Timothy W. Schmidt<br />
School <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> Sydney, NSW 2006,<br />
Australia<br />
Abstract<br />
Resonance-stabilized radicals such as<br />
1-phenylpropargyl [1] and 1-vinylpropargyl [2] have<br />
been identified to be prominent and ubiquitous<br />
products <strong>of</strong> hydrocarbon discharges. The<br />
identification <strong>of</strong> these radicals was made using a<br />
synergy <strong>of</strong> resonant 2-colour 2-photon ionization<br />
and 2-dimensional fluorescence spectroscopies.<br />
We have since exploited the principle <strong>of</strong> resonance<br />
stabilization to obtain the spectra <strong>of</strong> a library <strong>of</strong><br />
such radicals, including:<br />
1-naphthylmethyl, 2-naphthylmethyl,<br />
acenaphthenyl, 4-methylnaphth-1-ylmethyl, 5-<br />
methylnaphth-1-ylmethyl, 4-phenylbenzyl,<br />
4-(4’-methylphenyl)benzyl, 1,4-pentadienyl,<br />
phenalenyl, indanyl, 2-hydroxyindan-1-yl,<br />
2-methylindan-1-yl, 2-hydroxy-2-methylindan-1-yl,<br />
inden-2-ylmethyl, 1-phenylallyl,[3] 2-ethylindan-1-yl,<br />
2-ethyl-2-hydroxyindan-1-yl, 1-inden-2-ylethyl,<br />
4,”,”-trimethylbenzyl and 4, “-dimethylbenzyl<br />
radicals. The above species all display spectra in<br />
the visible region <strong>of</strong> the spectrum, yet show a<br />
variety <strong>of</strong> spectroscopic phenomena including
long Franck- Condon progressions, (pseudo)<br />
Jahn- Teller effects, Fermi resonance and<br />
Duschinsky mixing. The spectra will be discussed<br />
in the context <strong>of</strong> the chemistry <strong>of</strong> combustion,<br />
planetary atmospheres and interstellar space<br />
References:<br />
[1] Neil J. Reilly, Damian L. Kokkin, Masakazu Nakajima, Klaas<br />
Nauta, Scott H. Kable, Timothy W.<br />
Schmidt, J. Am. Chem. Soc. 130, 3137 (2008).<br />
[2] Neil J. Reilly, Masakazu Nakajima, Tyler P. Troy, Nahid Chalyavi,<br />
Kieran A. Duncan, Klaas Nauta,<br />
Scott H. Kable, Timothy W. Schmidt, J. Am. Chem. Soc. 131,<br />
13423-9 (2009).<br />
[3] Tyler P. Troy, Nahid Chalyavi, Ambili S. Menon, Gerard D.<br />
O'Connor, Burkhard Fückel,<br />
Klaas Nauta, Leo Radom, and Timothy W. Schmidt, Chemical<br />
Science, in press.<br />
P66<br />
Surface-enhanced Raman<br />
Spectroscopy <strong>of</strong> Is<strong>of</strong>lavones on<br />
Alternative Substrates<br />
Ryo Sekine1 , Evan G. Robertson, 2 Leone<br />
Spiccia3 , Richard A. Dluhy4 , Don McNaughton 5<br />
1 Centre for Biospectroscopy, Monash <strong>University</strong>, Clayton, VIC,<br />
3800, Australia, ryo.sekine@monash.edu<br />
2 School <strong>of</strong> Molecular Sciences, La Trobe <strong>University</strong>, VIC, 3086,<br />
Australia, e.robertson@latrobe.edu.au<br />
3 School <strong>of</strong> Chemistry, Monash <strong>University</strong>, Clayton, VIC, 3800,<br />
Australia, leone.spiccia@monash.edu<br />
4 Department <strong>of</strong> Chemistry, The <strong>University</strong> <strong>of</strong> Georgia, Athens,<br />
GA, USA, dluhy@uga.edu<br />
5 Centre for Biospectroscopy, Monash <strong>University</strong>, Clayton, VIC,<br />
3800, Australia, don.mcnaughton@monash.edu<br />
Abstract<br />
Is<strong>of</strong>lavones are naturally occurring chemicals with<br />
a breadth <strong>of</strong> potential health effects. As an<br />
anti-oxidant and phytoestrogen, it reduces the risk<br />
<strong>of</strong> cardio-vascular diseases, cancer, osteoporosis<br />
and various postmenopausal symptoms, while<br />
adverse effects have also been found through its<br />
action as an endocrine disruptor. 1 We have<br />
recently investigated a series <strong>of</strong> is<strong>of</strong>lavones using<br />
surface-enhanced Raman spectroscopy (SERS), a<br />
technique capable <strong>of</strong> enormous Raman signal<br />
enhancements and which overcome the low<br />
sensitivity <strong>of</strong> normal Raman spectroscopy. SERS<br />
is achieved by the adsorption <strong>of</strong> the analyte on<br />
roughened metal surfaces or particles, where<br />
Poster Presentations<br />
Monday 5 December - Session 2<br />
signals are enhanced by electromagnetic and<br />
chemical mechanisms. SERS analysis <strong>of</strong><br />
is<strong>of</strong>lavones using citrate-reduced silver colloids<br />
revealed that the deprotonation <strong>of</strong> hydroxyl groups<br />
lead to their interaction with the metal surface, and<br />
as a consequence, it is highly sensitive to the<br />
hydroxyl substitution pattern <strong>of</strong> the is<strong>of</strong>lavones. 2<br />
One <strong>of</strong> the key shortcomings <strong>of</strong> SERS precluding<br />
its widespread use is the significant spectral<br />
variability observed as a result <strong>of</strong> different<br />
analyte-metal interactions. The technique relies<br />
critically on the adsorption <strong>of</strong> the analyte on to the<br />
metal, which is dependent on not only the analyte<br />
and the metal, but also on the surrounding<br />
environment. Furthermore, despite the intense<br />
research in SERS, the preparation <strong>of</strong> reproducibly<br />
effective, robust and reusable surfaces (substrates)<br />
has been met with significant challenge. In the<br />
interests <strong>of</strong> broadening the scope <strong>of</strong> SERS<br />
applications in is<strong>of</strong>lavone research, we have<br />
examined the spectra <strong>of</strong> is<strong>of</strong>lavones on alternative<br />
substrates, selected on the basis <strong>of</strong> ease <strong>of</strong><br />
fabrication and reusability: oblique angle<br />
deposition SERS substrates (OADs) 3,4 and sol gel<br />
SERS substrates. 5 SERS from these substrates<br />
are presented and compared to SERS on the Ag<br />
colloids.<br />
Figure 1. SERS <strong>of</strong> is<strong>of</strong>lavones daidzein and formononetin on Ag<br />
colloids (black) and OADs (blue).<br />
References<br />
(1) Karahalil, B. Benefits and Risks <strong>of</strong> Phytoestrogens. In<br />
Phytoestrogens in Functional Foods; Yildiz, F., Ed.; CRC Press:<br />
Boca Raton, 2006.<br />
(2) Sekine, R.; Vongsvivut, J.; Robertson, E. G.; Spiccia, L.;<br />
McNaughton, D. J. Phys. Chem. B 2010, 114, 7104.<br />
(3) Chaney, S. B.; Shanmukh, S.; Dluhy, R.; Zhao, Y.-P. Appl. Phys.<br />
Lett. 2005, 87, 031908.<br />
(4) Driskell, J. D.; Shanmukh, S.; Liu, Y.-J.; Chaney, S. B.; Tang,<br />
X.-J.; Zhao, Y.-P.; Dluhy, R. A. J. Phys. Chem. C 2008, 112, 895.<br />
(5) Lee, Y.-H.; Dai, S.; Young, J. P. J. Raman Spectrosc. 1997, 28,<br />
635.<br />
145
Poster Presentations<br />
Monday 5 December - Session 2<br />
P67<br />
Dynamic Characterisation <strong>of</strong><br />
Surfaces Using In-Silico Nano-<br />
Indentation<br />
Lachlan Shaw1 , George Yiapanis1 , David<br />
Henry2 , Evan Evans3 , Irene Yarovsky2 1 School <strong>of</strong> Applied Sciences, RMIT <strong>University</strong>, GPO Box 2476,<br />
Vic, 3001<br />
2 School <strong>of</strong> Chemical and Mathematical Sciences, Murdoch<br />
<strong>University</strong>, South Street, WA, 6150<br />
3 BlueScope Steel Research Laboratories, Islands Rd, Port<br />
Kembla, NSW, 2505<br />
Abstract<br />
Physical and chemical aspects <strong>of</strong> polymer<br />
surfaces play an important role in interfacial<br />
phenomena and dominate processes such as<br />
adhesion and wettability. However, polymer<br />
surfaces <strong>of</strong>ten display dynamic characteristics,<br />
which depending on the intended application can<br />
have a pr<strong>of</strong>ound effect on the efficacy <strong>of</strong> the<br />
materials performance. For example, studies have<br />
shown that functionalized polymer surfaces tend<br />
to recover their native state as a result <strong>of</strong><br />
ageing1-2. Here, using force-field Molecular<br />
Dynamics we undertake in-silico nanoindentation3<br />
to dynamically study the physical and<br />
chemical properties <strong>of</strong> polymer surfaces and their<br />
affects on adhesion. The indenter material chosen<br />
represents a primary atmospheric contaminant<br />
and the surface, a modified crosslinked polyester<br />
paint coating. During nano-indentation we<br />
calculated the adhesion between contaminant and<br />
surface, and monitored dynamically, the surface<br />
roughness, skewness and density <strong>of</strong> the polymer<br />
layers (figure) by creation <strong>of</strong> surface contour maps.<br />
This technique may be applied to any deformable<br />
surface. Our results indicate that surface<br />
crosslinking increases the roughness and rigidity<br />
<strong>of</strong> the outer layer. In addition, crosslinking also<br />
provides a stable platform for modifiers that would<br />
otherwise migrate into the bulk material. However,<br />
nano-indentation <strong>of</strong> regions with flexible subsurfaces<br />
results in slip whereby crosslinkers shift<br />
dramatically to the side <strong>of</strong> the incoming<br />
contaminant resulting in both loss <strong>of</strong> rigidity and<br />
envelopment <strong>of</strong> contaminant.<br />
146<br />
Adhesion and density obtained during nanoindentation.<br />
Graph shows that variations in<br />
interfacial adhesion are accompanied with<br />
changes in the density <strong>of</strong> the indented material.<br />
A. Oláh, H. Hillborg, G. J. Vancso, Applied Surface Science 239,<br />
410 (2005).<br />
G. Yiapanis, D. J. Henry, E. Evans, I. Yarovsky, The Journal <strong>of</strong><br />
Physical Chemistry C 111, 6465 (2007).<br />
G. Yiapanis, D. J. Henry, E. Evans, I. Yarovsky, The Journal <strong>of</strong><br />
Physical Chemistry C 114, 478 (2009).<br />
P68<br />
CyDNA: A versatile photoswitchable<br />
biopolymer for advanced<br />
fluorescence microscopy<br />
applications.<br />
Darren A. Smith 1,2 ,*, Philipp Holliger 3 ,<br />
Cristina Flors 1 ,*<br />
1 EaStChem School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Edinburgh,<br />
Edinburgh, EH9 3JJ, United Kingdom.<br />
2 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Melbourne, Melbourne, 3010,<br />
Australia.<br />
3 MRC Laboratory <strong>of</strong> Molecular Biology, Cambridge, CB2 0QH,<br />
United Kingdom.<br />
Abstract<br />
CyDNA is the product <strong>of</strong> the controlled synthesis<br />
<strong>of</strong> a DNA fragment with a high density <strong>of</strong> Cy3 or<br />
Cy5 dyes incorporated into its structure.[1] This<br />
novel material can be up to 1kb and is constructed<br />
by a modified DNA polymerase that allows<br />
substitution <strong>of</strong> dC bases by their fluorescent<br />
dye-labelled analogue, Cy3- or Cy5-dC. The<br />
resulting biopolymer displays hundreds <strong>of</strong><br />
fluorophores and is brightly coloured and<br />
fluorescent. The high dye content can, however,<br />
result in dye-dye interactions that affect the overall
ightness <strong>of</strong> CyDNA. This may hinder the<br />
application <strong>of</strong> this material in microarray and<br />
micr<strong>of</strong>luidic applications. We will present a bulk<br />
and single-molecule photophysical study that<br />
reveals several quenching mechanisms occurring<br />
in CyDNA, namely, the formation <strong>of</strong> nonfluorescent<br />
H-aggregates <strong>of</strong> the dyes as well as<br />
energy hopping and transfer to lower energy,<br />
non-fluorescent traps. We investigate the optimal<br />
substitution patterns to enhance CyDNA<br />
brightness.<br />
Furthermore, it has previously been shown that, in<br />
the presence <strong>of</strong> a thiol and an enzymatic oxygen<br />
scavenging system, proximal Cy3 and Cy5<br />
fluorophores form an optical switch.[2] We have<br />
used this property to transform CyDNA into an<br />
efficient photoswitchable biopolymer by<br />
hybridizing complementary single stranded Cy3and<br />
Cy5-substituted CyDNA. Photoswitching has<br />
been applied in localization-based superresolution<br />
fluorescence microscopy to study<br />
CyDNA topology in nanoscale detail. Moreover, we<br />
have used CyDNA photoswitching in Optical<br />
Lock-in Detection imaging,[3] which is a technique<br />
capable <strong>of</strong> greatly enhancing image contrast in<br />
fluorescence microscopy. The combination <strong>of</strong><br />
CyDNA and the above techniques has enormous<br />
potential in the study <strong>of</strong> the structure <strong>of</strong><br />
chromosomes at the nanoscale.[4]<br />
This work was supported by a Universitas21 PhD<br />
Scholarship and The Royal Society.<br />
References:<br />
[1] N. Ramsay et al., J. Am. Chem. Soc. 132 (2010) 5096.<br />
[2] M. Bates et al., Phys. Rev. Lett. 94 (2005) 108101.<br />
[3] G. Marriott et al., Proc. Natl. Acad. Sci. U.S.A. 105 (2008)<br />
17789.<br />
[4] C. Flors, Biopolymers 95 (2011) 290.<br />
*Corresponding authors: E-mail: d.smith@student.unimelb.edu.au,<br />
cristina.flors@ed.ac.uk<br />
Poster Presentations<br />
Monday 5 December - Session 2<br />
P69<br />
Computational characterisation <strong>of</strong><br />
an unusual metallacalix[4]arene<br />
dinitrogen activator<br />
Richard Terrett1 , Germán E. Cavigliasso1 ,<br />
Rob Stranger1 , B.F. Yates 2<br />
1 Research School <strong>of</strong> Chemistry, Australian National <strong>University</strong>,<br />
ACT, 0200, email: rterrett@rsc.anu.edu.au<br />
2 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Tasmania, Private Bag 75,<br />
Hobart, TAS 7001<br />
Abstract<br />
The calix[4]arene niobium(III) complex ([L]<br />
Nb–N=N–Nb[L] where [L] = p-tert-butylcalix[4]<br />
arene), reported to bind N2 in a μ2–linear dimeric<br />
capacity and to activate the N–N triple bond to<br />
1.39 Å, corresponding to the longest N–N bond<br />
known in the end–on coordination mode, was<br />
subjected to a computational investigation<br />
involving both density functional and wavefunction<br />
based methods to establish the basis for the<br />
unprecedented level <strong>of</strong> activation. Replacement <strong>of</strong><br />
the calix[4]arene ligand with hydroxide or<br />
methoxide ligands reveals that the organic<br />
backbone structure <strong>of</strong> the calix[4]arene ligand<br />
exerts negligible electronic influence over the metal<br />
centre, serving only to geometrically constrain the<br />
coordinating phenoxide groups. A fragment<br />
bonding analysis shows that metal–to–dinitrogen<br />
π* backbonding is the principal Nb–N interaction,<br />
providing a strong electronic basis for analogy with<br />
other well–characterised three– and four–<br />
coordinate complexes which bind N2 end–on.<br />
While the calculated structure <strong>of</strong> the metallacalix[4]<br />
arene unit is reproduced with high accuracy, as is<br />
also the Nb–Nb separation, the calculated<br />
equilibrium geometry <strong>of</strong> the complex under a<br />
variety <strong>of</strong> conditions consistently indicates against<br />
a 1.39 Å activation <strong>of</strong> the N–N bond. Instead, the<br />
calculated N–N distances fall within the range<br />
1.26–1.28 Å, a result concordant with closely<br />
related three– and four-coordinate μ2–N2<br />
complexes as well as predictions derived from<br />
trends in N–N stretching frequency for a number <strong>of</strong><br />
crystallographically characterized linear N2<br />
activators. A number <strong>of</strong> potential causes for this<br />
bond length discrepancy are explored.<br />
147
Poster Presentations<br />
Monday 5 December - Session 2<br />
P70<br />
Recombination <strong>of</strong> photolytically<br />
generated iodine in single<br />
iodoalkane microdroplets<br />
Phillip J. Tracey1 , Bartholomew S. Vaughn1 ,<br />
Adam J. Trevitt 1<br />
1 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>, NSW, 2522<br />
pjt105@uowmail.edu.au<br />
Abstract<br />
Chemistry occurring at the liquid/air interface <strong>of</strong><br />
single microdroplets is important in chemical<br />
systems such as fuel sprays and aerosols. To<br />
explore this chemistry we have developed an<br />
experimental setup probe physical and chemical<br />
changes in single microdroplets by combining<br />
optical cavity enhancement effects with Raman<br />
spectroscopy. As a preliminary demonstration <strong>of</strong><br />
the instrumentations capabilities we have<br />
investigated the recombination <strong>of</strong> iodine radicals<br />
generated within single microdroplets using cavity<br />
enhanced Raman spectroscopy (CERS).<br />
The instrument consists <strong>of</strong> a three-laser setup and<br />
a drop-on-demand droplet generator. Single<br />
microdroplets are formed and free fall, first passing<br />
through a CW He:Ne laser (λ = 632 nm) whose<br />
scatter provides a trigger signal for the other two<br />
lasers in the setup. A pulsed Nd:YAG UV laser (λ =<br />
266 nm) irradiates the droplet to initiate photolysis.<br />
Another pulsed Nd:YAG laser (λ = 532 nm) is used<br />
to perform Raman spectroscopy, enhanced by<br />
cavity resonance effects within the microdroplets.<br />
UV laser photolysis <strong>of</strong> iodoalkanes results in the<br />
liberation <strong>of</strong> iodine radicals, which subsequently<br />
combine to form molecular iodine over the<br />
timescale <strong>of</strong> hundreds <strong>of</strong> nanoseconds. Formation<br />
<strong>of</strong> I2 within the droplets quenches the cavity<br />
enhanced Raman, as the iodine population grows<br />
until such a point that no signal is observed. By<br />
controlling the delay between UV photolysis and<br />
Raman acquisition, the decrease in Raman signal<br />
148<br />
can be plotted over time and the kinetics <strong>of</strong> iodine<br />
radical recombination can be studied. The<br />
experimental setup and findings from this study<br />
will be presented in greater detail in this poster.<br />
P71<br />
Symmetry, Pseudo-symmetry and<br />
Evolution in Protein Structures<br />
Donald G Vanselow 1<br />
1 nativeproteins.blogspot.com 54 Greenways Rd., Glen Waverley<br />
VIC 3150, Australia. dvanselow@hotmail.com<br />
Abstract<br />
The principles <strong>of</strong> Evolution and Natural Selection<br />
are applied to the existence <strong>of</strong> symmetry in<br />
tetrameric proteins and the presence <strong>of</strong> pseudosymmetry<br />
within protein subunits. The probability<br />
that protein symmetry is shaped by these<br />
principles is shown to be very useful in guiding the<br />
computer-assisted assembly (docking) <strong>of</strong> the<br />
subunits <strong>of</strong> tetrameric proteins. Without this<br />
guidance the large number <strong>of</strong> degrees <strong>of</strong> freedom<br />
involved in simultaneously docking four subunits<br />
would render the docking process impossible. The<br />
pseudo-symmetry <strong>of</strong> α and β barrels is helpful in<br />
orientating them relative to the rest <strong>of</strong> the protein,<br />
especially when the active site location is not<br />
known. Examples will be shown from the<br />
neuraminidases, galactose oxidase,<br />
dihydrodipicolinate synthase (DHDPS), and<br />
hemoglobin [1], where the tetramers [2] have been<br />
assembled with help from symmetry and<br />
pseudo-symmetry. In the case <strong>of</strong> hemoglobin,<br />
pseudo-symmetry gives rise to possible<br />
“quaternary isomers” which may explain the<br />
mechanism <strong>of</strong> the pH dependence <strong>of</strong> oxygen<br />
binding [1].<br />
The shapes <strong>of</strong> these tetramers also conform with<br />
the physics <strong>of</strong> catalysis and binding described in<br />
2002 [3].<br />
Refs:<br />
[1] The data-base <strong>of</strong> native protein structures is at www.<br />
nativeproteins.net76.net/gallery/index.htm<br />
Papers describing the structures and discussing related issues are<br />
at http://nativeproteins.blogspot.com<br />
Both sites are maintained by the author.<br />
[2] Vanselow, D. G. 2008. Haemoglobin Revisited. WATOC 2008<br />
Poster No. 321. http://posters.f1000.com/P1181<br />
[3] Vanselow, D. G. 2002. Role <strong>of</strong> constraint in catalysis and<br />
high-affinity binding by proteins, Biophys. J. 82: 2293–2303.
P72<br />
Single Microdroplet Laser<br />
Spectroscopy<br />
Bartholomew S. Vaughn1 , Phillip J.<br />
Tracey1 , Adam J. Trevitt 1 .<br />
1 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>, NSW 2522<br />
bv703@uowmail.edu.au<br />
Abstract<br />
The chemistry that occurs at the surface <strong>of</strong> single<br />
microdroplets plays an important role in various<br />
chemical systems, such as fuel sprays and<br />
atmospheric aerosols. These surface processes<br />
affect how these systems evolve chemically. This<br />
poster will outline the development <strong>of</strong> a technique<br />
that uses cavity enhanced Raman on single<br />
microdroplets in order to probe the boundary<br />
region <strong>of</strong> single microdroplets.<br />
The experimental setup uses a piezo-activated<br />
on-demand microdroplet generator to produce a<br />
stable droplet stream – as we will demonstrate.<br />
Once the droplets are formed they free-fall, where<br />
they cross a continuous He:Ne laser. The light<br />
scattered <strong>of</strong>f the droplet is collected to trigger a<br />
pulsed Nd:YAG laser operating at 532 nm, which<br />
serves as the Raman probe laser. The scattered<br />
light from the droplet is collected and focused into<br />
a Czerny-Turner spectrometer equipped with a<br />
CCD. In this way a single laser shot hits a single<br />
droplet producing a one spectrum. The droplet<br />
Raman spectra are dominated by cavity modes<br />
that are dependent on the droplet size and<br />
refractive index. Monitoring these peak positions in<br />
the spectra allows us to sensitively track the<br />
droplet size.<br />
The setup was tested using water as a liquid to<br />
master the techniques <strong>of</strong> reproducible droplet<br />
generation, as well as optimising the optical<br />
arrangement. This was extended to probe droplets<br />
composed <strong>of</strong> squalane, squalene and biodiesel<br />
fuels including methyl esters. The Raman spectra<br />
contained C-H stretch vibrational band, as well as<br />
C=C stretch in certain molecules. Interestingly the<br />
C=O band was not observed in the methyl ester<br />
droplets. This poster will discuss the experimental<br />
configuration and findings in greater detail.<br />
Poster Presentations<br />
Monday 5 December - Session 2<br />
P73<br />
Molecular Dynamics <strong>of</strong> Curcumin<br />
and Linked Cyclodextrin Dimers<br />
Samuel J. Wallace 1 , David M. Huang2 ,<br />
Takaaki Harada, 3 Tak W. Kee4 1 <strong>University</strong> <strong>of</strong> Adelaide, North Terrace, South Australia, 5005,<br />
sam.wallace@adelaide.edu.au<br />
2 <strong>University</strong> <strong>of</strong> Adelaide, North Terrace, South Australia, 5005,<br />
david.huang@adelaide.edu.au<br />
3 <strong>University</strong> <strong>of</strong> Adelaide, North Terrace, South Australia, 5005,<br />
takaaki.harada@adelaide.edu.au<br />
4 <strong>University</strong> <strong>of</strong> Adelaide, North Terrace, South Australia, 5005,<br />
tak.kee@adelaide.edu.au<br />
Abstract<br />
Curcumin, which is a naturally occurring<br />
compound known to have a wide range <strong>of</strong><br />
medicinal properties, including anti-cancer,<br />
anti-oxidant and wound healing, is a biomolecule<br />
<strong>of</strong> particular interest.1-3 The bioavailability <strong>of</strong><br />
curcumin however, is limited by its solubility and<br />
stability in the aqueous environments.4 The<br />
delivery <strong>of</strong> curcumin effectively in vivo requires<br />
stabilisation in a host medium. Various strategies<br />
to resolve the stabilisation issues have been<br />
attempted with micelles, nanoparticles and<br />
cyclodextrins.5-8 Cyclodextrins are cyclic<br />
oligosaccharides with a hydrophobic interior and<br />
hydrophilic exterior which provide an ideal<br />
environment for the stabilisation <strong>of</strong> curcumin.<br />
Recently, our group has used linked -cyclodextrin<br />
dimers to enhance the stability <strong>of</strong> curcumin by a<br />
factor <strong>of</strong> at least 700.6<br />
The work presented here uses molecular<br />
dynamics simulations running NAMD and using<br />
the Generalised Amber Force Field to determine<br />
149
Poster Presentations<br />
Monday 5 December - Session 2<br />
the binding constants and free energy <strong>of</strong> binding<br />
<strong>of</strong> curcumin to linked -cyclodextrin dimers.9,10<br />
The simulation results are compared with<br />
experimental results.<br />
(1) Masuda, T.; Jitoe, A.; Isobe, J.; Nakatani, N.; Yonemori, S.<br />
Phytochemistry 1993, 32, 1557.<br />
(2) Piper, J. T.; Singhal, S. S.; Salameh, M. S.; Torman, R. T.;<br />
Awasthi, Y. C.; Awasthi, S. Int. J. Biochem. Cell Biol. 1998, 30,<br />
445.<br />
(3) Sidhu, G. S.; Manni, H.; Gaddipati, J. P.; Singh, A. K.; Seth, P.;<br />
Banaudha, K. K.; Patnaik, G. K.; Maheshwari, R. K. Wound<br />
Repair and Regeneration 1999, 7, 362.<br />
(4) Wang, Y. J.; Pan, M. H.; Cheng, A. L.; Lin, L. I.; Ho, Y. S.; Hsieh,<br />
C. Y.; Lin, J. K. J. Pharm. Biomed. Anal. 1997, 15, 1867.<br />
(5) Aggarwal, B. B.; Anand, P.; Nair, H. B.; Sung, B. K.;<br />
Kunnumakkara, A. B.; Yadav, V. R.; Tekmal, R. R. Biochem.<br />
Pharmacol. 2010, 79, 330.<br />
(6) Kee, T. W.; Harada, T.; Pham, D. T.; Leung, M. H. M.; Huy, T. N.;<br />
Lincoln, S. F.; Easton, C. J. J. Phys. Chem. B 2011, 115, 1268.<br />
(7) Kee, T. W.; Leung, M. H. M.; Colangelo, H. Langmuir 2008, 24,<br />
5672.<br />
(8) Tonnesen, H. H.; Masson, M.; L<strong>of</strong>tsson, T. Int. J. Pharm. 2002,<br />
244, 127.<br />
(9) Schulten, K.; Phillips, J. C.; Braun, R.; Wang, W.; Gumbart, J.;<br />
Tajkhorshid, E.; Villa, E.; Chipot, C.; Skeel, R. D.; Kale, L. J.<br />
Comput. Chem. 2005, 26, 1781.<br />
(10) Wang, J. M.; Wolf, R. M.; Caldwell, J. W.; Kollman, P. A.; Case,<br />
D. A. J. Comput. Chem. 2005, 26, 114.<br />
P74<br />
De novo Structure prediction <strong>of</strong><br />
Peptide Based Biomimetic<br />
Carbohydrate Receptors<br />
Mark Waller1 1 Organic Chemistry Institute, <strong>University</strong> <strong>of</strong> Münster,<br />
Corrensstraße 40, D-48149 Münster, Germany<br />
Abstract<br />
Carbohydrate recognition poses a remarkable<br />
challenge within host-guest chemistry. In a recent<br />
study1 , cyclic oligopeptides consisting <strong>of</strong> two<br />
Cys-His-Cys tripeptide fragments were found to<br />
be able to selectively bind N-acetyl neuraminic<br />
acid (NANA) with an unprecedented high binding<br />
constant. Experimentally, a HisHis:NANA ratio <strong>of</strong><br />
1:2 was observed to exhibit strong cooperative<br />
binding. Herein we applied a meta-optimization<br />
strategy employing a multi-tiered SE, DFT and<br />
QM/MM to elucidate possible candidate<br />
structures. We carefully compare and contrast our<br />
models to determine the origins <strong>of</strong> this cooperative<br />
150<br />
behaviour.<br />
1 Ravoo et al. Angew. Chem. Int Ed. 2010, 49, 7340-7345<br />
P75<br />
Modelling Cellulase Activity upon<br />
Cellulose Surfaces using Cellular<br />
Automata<br />
Andrew C. Warden1 , Bryce A. Little2 ,<br />
Victoria S. Haritos1 1 CSIRO Ecosystem Sciences, Clunies Ross St, Acton, Canberra,<br />
ACT, 2601<br />
2 CSIRO Livestock Industries, Queensland Biosciences Precinct,<br />
306 Carmody Road, St. Lucia, QLD, 4067<br />
Abstract<br />
With increasing emphasis on energy<br />
independence and the growing concern over finite<br />
petroleum reserves, attention has turned to<br />
biomass as a potential feedstock with which to<br />
produce a significant proportion <strong>of</strong> the world’s<br />
liquid fuels. Lignocellulose is an abundant,<br />
renewable raw material that contains cellulose, a<br />
component that has the potential to replace oil as<br />
a starting material for the production <strong>of</strong> transport<br />
fuels.<br />
The saccharification <strong>of</strong> cellulose, breaking the<br />
polymer into monosaccharides, yields glucose<br />
which can undergo microbial fermentation to<br />
bi<strong>of</strong>uels such as ethanol and butanol, or be used<br />
as a feedstock for the production <strong>of</strong> lipids and<br />
chemicals. Achieving efficient saccharification from<br />
complex biomass is challenging, and one <strong>of</strong> the<br />
main factors impeding the commercialisation <strong>of</strong><br />
the biochemical method <strong>of</strong> cellulosic fuels and<br />
chemicals production.<br />
Cellulase enzymes used to break down cellulose<br />
into fermentable sugars are <strong>of</strong> several different<br />
types (generally, cellobiohydrolase I,<br />
cellobiohydrolase II, endoglucanase and<br />
β-glucosidase) that perform different functions.<br />
They form a complex, interacting network between<br />
themselves, soluble hydrolysis product molecules<br />
(both substrates and inhibitors) and the solid<br />
cellulose substrate. Most models produced to date<br />
use solution-phase kinetics to describe overall<br />
cellulase activity. We have taken a 2-phase<br />
approach whereby the solution chemistry is
modelled using traditional Michaelis-Menten<br />
kinetics and the solid phase activity is described<br />
using cellular automata.<br />
Cellulase4D is a general purpose model with<br />
which various behaviours <strong>of</strong> many individual<br />
enzymes on the cellulose surface can be<br />
examined. These include adsorption strength,<br />
mobility on the surface, crowding or jamming and<br />
adsorption probability. In addition, the model<br />
incorporates solution phase behaviours described<br />
with commonly used parameters such as kcat, Km<br />
and Ki. We have incorporated a graphical user<br />
interface with which the various parameters can<br />
be altered, real-time plotting <strong>of</strong> the production <strong>of</strong><br />
hydrolysis products such as glucose, cellobiose<br />
and higher polysaccharides, and a 3D<br />
representation <strong>of</strong> the cellulose and enzymes that<br />
can be manipulated by the user as the simulation<br />
progresses.<br />
We will present the results from simulations aimed<br />
at unravelling the effects <strong>of</strong> crowding, adsorption<br />
strength and reaction rates, and discuss the<br />
possible implications for the tuning <strong>of</strong> properties <strong>of</strong><br />
the individual cellulase components through<br />
enzyme engineering.<br />
P76<br />
Lipid Composition Regulates the<br />
Conformation and Insertion <strong>of</strong> the<br />
Antimicrobial Peptide Maculatin 1.1<br />
Thomas Whitwell1 , Marc-Antoine Sani1 ,<br />
Frances Separovic1 1 School <strong>of</strong> Chemistry, Bio21 Institute, <strong>University</strong> <strong>of</strong> Melbourne,<br />
VIC 3010<br />
Abstract<br />
The effect <strong>of</strong> the lipid composition <strong>of</strong> biological<br />
membranes on antimicrobial peptide (AMP)<br />
structure needs to be understood to rationalise<br />
AMP activity. The affinity <strong>of</strong> AMPs for anionic lipid<br />
membranes is well reported, but this has <strong>of</strong>ten<br />
been determined using dimyristoyl (tetradecyl)<br />
chain phospholipid bilayers. In this study, we<br />
highlight the importance <strong>of</strong> lipid chain length and<br />
saturation on AMP secondary structure. Maculatin<br />
1.1 is a 21 residue cationic AMP secreted from the<br />
Poster Presentations<br />
Monday 5 December - Session 2<br />
skin <strong>of</strong> Australian tree frogs that acts against Gram<br />
positive bacteria. The secondary structure <strong>of</strong><br />
maculatin 1.1 was determined by circular<br />
dichroism spectroscopy in dispersions <strong>of</strong><br />
unilamellar vesicles and oriented bilayers using a<br />
range <strong>of</strong> phospholipids. For neutral<br />
phosphatidylcholine (PC), unlike anionic<br />
phospholipids, the magnitude <strong>of</strong> the peptide<br />
interactions was dependent on the constituent<br />
acyl chain length and degree <strong>of</strong> saturation.<br />
Oriented circular dichroism (OCD) data indicated<br />
that helical structure was likely promoted by<br />
peptide insertion into the hydrophobic core <strong>of</strong> PC<br />
bilayers. The addition <strong>of</strong> cholesterol (30% mol/mol)<br />
tended to decrease the membrane interaction <strong>of</strong><br />
the peptide. OCD spectra <strong>of</strong> maculatin 1.1 with<br />
anionic lipid membranes, such as negatively<br />
charge phosphatidylglycerol (PG), indicated that<br />
the peptide was locked onto the membrane<br />
surface due to electrostatic interactions.<br />
Furthermore, increasing the membrane curvature<br />
by using small unilamellar vesicles reduced the<br />
proportion <strong>of</strong> helical structure in all systems by<br />
~10%. Clearly, lipid chain length and saturation<br />
strongly influenced the peptide interactions with<br />
phospholipid membranes. Dye release<br />
experiments are being performed to determine the<br />
AMP mechanism and further experiments with<br />
binary lipid mixtures are underway. Preliminary<br />
data has indicated that increasing the proportions<br />
<strong>of</strong> phosphatidylethanolamine (PE) in either PE/PC<br />
or PE/PG consistently reduced the helical content<br />
<strong>of</strong> maculatin 1.1 and may relate to the lack <strong>of</strong><br />
activity with Gram negative bacteria.<br />
P77<br />
The Quokka Small Angle Neutron<br />
Scattering Instrument at OPAL<br />
K. Wood1 , C. J. Garvey1 , E. P. Gilbert1 1 Bragg Institute, Australian Nuclear Science and Technology<br />
Organisation, Locked Bag 2001, Kirrawee DC, NSW 2232,<br />
Australia<br />
Abstract<br />
Quokka is a 40 metre small angle neutron<br />
scattering instrument installed on the cold source<br />
<strong>of</strong> the OPAL research reactor operated by ANSTO.<br />
Small angle scattering is a powerful technique for<br />
151
Poster Presentations<br />
Monday 5 December - Session 2<br />
looking at the structures <strong>of</strong> objects on the<br />
nanoscale (1-10nm), such as polymers, biomacromolecules,<br />
ceramics, emulsions etc… Both<br />
small angle neutron and x-ray scattering<br />
instruments are accessible at the Bragg Institute<br />
through a peer-reviewed proposal system. One <strong>of</strong><br />
the main advantages <strong>of</strong> neutrons over x-rays is the<br />
possibility to design experiments making use <strong>of</strong><br />
the large difference in neutron scattering cross<br />
section between hydrogen and its heavier isotope<br />
deuterium.<br />
In addition to the standard 20-position automatic<br />
sample changer, a wide range <strong>of</strong> sample<br />
environment equipment is available for use on the<br />
beamline including a rheometer, rapid heat/<br />
quench cell and a stop-flow mixing cell.<br />
The capabilities and performance <strong>of</strong> Quokka will<br />
be presented, along with highlights from recent<br />
experiments.<br />
Quokka will be available for beamtime requests in<br />
March 2012 through the Bragg Institute portal<br />
https://neutron.ansto.gov.au/Bragg/proposal/<br />
index.jsp. It is also possible to submit proposals for<br />
chemical and biological deuteration through the<br />
same portal.<br />
P78<br />
Structure, dynamics and interactions<br />
<strong>of</strong> malaria surface antigens<br />
Tessa R. Young 1 , David K. Chalmers1 ,<br />
Christopher A. MacRaild1 , Marie O. Pedersen1 ,<br />
Robin F. Anders2 , Raymond S. Norton1 1 Monash Institute <strong>of</strong> Pharmaceutical Sciences, Monash<br />
<strong>University</strong>, 381 Royal Parade, Parkville 3052, Australia<br />
2 Department <strong>of</strong> Biochemistry, La Trobe <strong>University</strong>, Bundoora,<br />
VIC 3086, Australia<br />
Abstract<br />
Merozoite surface protein 2 (MSP2) is one <strong>of</strong> the<br />
most abundant antigens presented on the surface<br />
<strong>of</strong> the blood stage malaria parasite, Plasmodium<br />
falciparum, and thus a leading malaria vaccine<br />
candidate. MSP2 is anchored to the parasite<br />
membrane by a C-terminal<br />
glycosylphosphatidylinositol (GPI) moiety, it is<br />
highly disordered in solution and the conformation<br />
152<br />
<strong>of</strong> the protein on the surface <strong>of</strong> the merozoite is<br />
unknown. Determination <strong>of</strong> this structure would<br />
lead to a better understanding <strong>of</strong> the way in which<br />
MSP2 interacts with the immune system.<br />
Molecular dynamics simulations <strong>of</strong> the N-terminal<br />
and C-terminal regions <strong>of</strong> MSP2 have been carried<br />
out to determine the structures <strong>of</strong> N-terminal and<br />
C-terminal regions <strong>of</strong> MSP2.<br />
MSP2 contains conserved N-terminal and<br />
C-terminal sequences flanking a central variable<br />
region. Although highly polymorphic all MSP2<br />
alleles can be grouped into two families, 3D7 and<br />
FC27. Models <strong>of</strong> the first 31 residues, including the<br />
full conserved N-terminal domain (25 residues) <strong>of</strong><br />
both 3D7 and FC27 MSP2 were built in an<br />
alpha-helical conformation, as experimental<br />
evidence suggests that the N-terminal regions<br />
show a helical propensity (1). Molecular dynamics<br />
simulations were carried out under a variety <strong>of</strong><br />
conditions: in solution, with micelles, and in a<br />
membrane environment, to observe and compare<br />
the helicity <strong>of</strong> the two peptides. Replica exchange<br />
molecular dynamics simulations have also been<br />
used, allowing the peptides to sample a larger<br />
conformational space to aid in structure definition.<br />
Molecular dynamics simulations <strong>of</strong> the GPIanchored<br />
and free C-terminal region <strong>of</strong> MSP2<br />
(MSP2193-228) in a membrane environment have<br />
also been carried out evaluate if the GPI-anchor<br />
modifies the conformation <strong>of</strong> this region <strong>of</strong> the<br />
protein (2).<br />
REFERENCES<br />
1. Zhang, X., Perugini, M. A., Yao, S., Adda, C. G., Murphy, V. J.,<br />
Low, A., Anders, R. F., and Norton, R. S. (2008) Solution<br />
conformation, backbone dynamics and lipid interactions <strong>of</strong> the<br />
intrinsically unstructured malaria surface protein MSP2., J Mol<br />
Biol 379, 105-121.<br />
2. Adda, C. G., Murphy, V. J., Sunde, M., Waddington, L. J.,<br />
Schloegel, J., Talbo, G. H., Vingas, K., Kienzle, V., Masciantonio,<br />
R., Howlett, G. J., Hodder, A. N., Foley, M., and Anders, R. F.<br />
(2009) Plasmodium falciparum merozoite surface protein 2 is<br />
unstructured and forms amyloid-like fibrils, Mol Biochem<br />
Parasitol 166, 159-171.
P79<br />
New insights into the chemical<br />
reactivity <strong>of</strong> the deazaflavin c<strong>of</strong>actor<br />
F420 through quantum chemical<br />
calculations.<br />
Peng Yuan1 , Junming Ho1 , Colin J. Jackson1 ,<br />
Michelle L. Coote1 1 Research School <strong>of</strong> Chemistry, Australian National <strong>University</strong>,<br />
ACT, 0200, coote@rsc.anu.edu.au<br />
Abstract<br />
The deazaflavin enzyme c<strong>of</strong>actor F420 is only<br />
found in certain bacterial lineages, such as<br />
Mycobacterium tuberculosis, and not in higher<br />
organisms. Recent studies have shown it to be<br />
involved in a range <strong>of</strong> redox reactions, including<br />
the degradation <strong>of</strong> environmental toxins and the<br />
activation <strong>of</strong> pro-drugs. However, we lack a<br />
detailed understanding <strong>of</strong> its chemical reactivity<br />
and its role in catalysis. To address this, we<br />
describe the application <strong>of</strong> quantum chemistry<br />
calculations to probe the reaction mechanism <strong>of</strong><br />
F420 and its substrates. This approach has the<br />
potential to shed light on the role <strong>of</strong> this c<strong>of</strong>actor,<br />
and the enzymes that utilize it, in a range <strong>of</strong><br />
important processes including detoxification and<br />
drug metabolism.<br />
P80<br />
Utilization <strong>of</strong> Ambient Ozone for<br />
Determining Double Bond Positions<br />
in Unsaturated Lipids<br />
Shane R. Ellis1 , Marc in het Panhuis2 , Todd<br />
W. Mitchell3 , Stephen J. Blanksby1 ,<br />
1 ARC Centre <strong>of</strong> Excellence for Free Radical Chemistry and<br />
Biotechnology, School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>,<br />
<strong>Wollongong</strong>, NSW, 2522<br />
2 School <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>, <strong>Wollongong</strong>,<br />
NSW, 2522<br />
3 School <strong>of</strong> Health Sciences, <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong>,<br />
<strong>Wollongong</strong>,<br />
Abstract<br />
Previously our group has reported the reaction <strong>of</strong><br />
ozone with ionized lipids within the confines <strong>of</strong> an<br />
ion-trap mass spectrometer for the elucidation <strong>of</strong><br />
Poster Presentations<br />
Monday 5 December - Session 2<br />
double bond positions. [1]. While this has proven to<br />
be a powerful analytical approach it requires an<br />
expensive high-concentration ozone generator<br />
and a specifically modified mass spectrometer.<br />
Interestingly, ambient ozone - present in the<br />
troposphere within urban environments - has<br />
previously been identified as a source <strong>of</strong> oxidation<br />
when samples are left in the ambient laboratory<br />
environment [2] . In this study the surface analysis<br />
technique Desorption Electrospray Ionization Mass<br />
Spectrometry (DESI-MS) is used to investigate the<br />
reaction <strong>of</strong> ambient ozone with unsaturated<br />
phospholipids deposited onto Teflon and silica<br />
TLC plates. Products originating from ozonolysis <strong>of</strong><br />
the deposited lipids can be observed after only 5<br />
minutes following deposition and allow the<br />
assignment <strong>of</strong> the double bond positions within<br />
the parent lipid. This work describes the powerful<br />
combination <strong>of</strong> DESI-MS and TLC in allowing both<br />
the lipid composition and double bond positions<br />
within lipids to be rapidly investigated simply by<br />
exposing the sample to the ambient laboratory<br />
environment prior to analysis, thus negating the<br />
need for expensive ozone generators.<br />
References<br />
[1] Thomas, M. C., Mitchell, T. W., Harman, D. G., Deeley, J. M.,<br />
Nealon, J. R., Blanksby, S. J. Ozone-Induced Dissociation:<br />
Elucidation <strong>of</strong> Double Bond Position within Mass-Selected Lipid<br />
Ions. Anal. Chem. 2007, 80, 303.<br />
[2] Cohen, S. L. Ozone in Ambient Air as a Source <strong>of</strong> Adventitious<br />
Oxidation. A Mass Spectrometric Study. Anal. Chem. 2006, 78,<br />
4352.<br />
P81<br />
Thi<strong>of</strong>lavin T and its derivatives:<br />
Revealing their spectroscopic<br />
properties in the absence and<br />
presence <strong>of</strong> insulin amyloid fibrils<br />
Eric H.-L. Chen 1 , Jack C.-C. Hsu 1 , Frederick<br />
Y. Luh 1 , T.-S. Lim2 , Rita P.-Y. Chen1 1 Institute <strong>of</strong> Biological Chemistry, Academia Sinica, Taipei, 11529,<br />
Taiwan<br />
2 Department <strong>of</strong> Physics, Tunghai <strong>University</strong>, Taichung 407, Taiwan<br />
Abstract<br />
Thi<strong>of</strong>lavin T (ThT) is a common tool for diagnostics<br />
<strong>of</strong> the amyloid fibril formation. Pure ThT has a<br />
strong absorption peak centered at 412 nm and<br />
low fluorescence emission at 479 nm while excited<br />
153
at 412 nm. While binding with amyloid fibrils, the<br />
ThT/amyloid complex emits strong fluorescence at<br />
487 nm while excited at 442 nm, making ThT<br />
fluorescence assay is the most common and<br />
sensitive technique in monitoring amyloid fibril<br />
formation. Pervious researches suggested that the<br />
strong fluorescence emission at 487 nm is<br />
contributed from amyloids binding with ThT dimer<br />
and that ThT monomer causes a weak absorption<br />
at ~330 nm and an emission band at 450 nm while<br />
excited at 330 nm. In this work, we found that ThT<br />
is prone to be modified and three derivatives are<br />
formed when ThT is dissolved in solution or upon<br />
UV irradiation. These three derivatives are purified<br />
by high performance liquid chromatography and<br />
characterized by mass spectroscopy and nuclear<br />
magnetic resonance. The spectroscopic<br />
properties <strong>of</strong> pure ThT and its three derivatives<br />
were carefully examined and compared. Our<br />
results suggested that the emission peak at 450<br />
nm which has been reported previously might be<br />
resulted from one oxidized ThT derivative but not<br />
ThT monomer.<br />
Notes<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
154<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................<br />
............................................................................................
Gold Sponsors<br />
Name badge Sponsor<br />
Computation Chemistry Workshop Sponsor<br />
Keynote Speaker Sponsor<br />
Trade Exhibitors<br />
Supporters<br />
Conference Sponsors<br />
155
Conference Sponsors<br />
The organising committee <strong>of</strong> BioPhysChem 2011 would like to<br />
thank the following sponsors and exhibitors for their generous<br />
support.<br />
156<br />
Gold Sponsors<br />
<strong>University</strong> <strong>of</strong> <strong>Wollongong</strong><br />
The <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong> School <strong>of</strong> Chemistry has active research<br />
programs in both pure and applied chemistry, with the recent ERA<br />
assessment highlighting the quality and breadth <strong>of</strong> chemical research carried<br />
out.<br />
Major research areas include targeted drug discovery, design and synthesis,<br />
biological chemistry, use <strong>of</strong> mass spectrometry to study both biological and<br />
chemical systems, atmospheric chemistry, food chemistry, free radical<br />
chemistry, laser chemistry, analytical and environmental chemistry,<br />
nanomaterials and intelligent polymers.<br />
Undergraduate programs include a Bachelor <strong>of</strong> Medicinal Chemistry and<br />
Bachelor <strong>of</strong> Nanotechnology alongside the Bachelor <strong>of</strong> Science.<br />
Whether you are interested in research, teaching and learning or just plain<br />
curious about Chemistry, the School <strong>of</strong> Chemistry has something to <strong>of</strong>fer.<br />
ARC Centre <strong>of</strong> Excellence for Free Radical Chemistry and<br />
Biotechnology<br />
The ARC Centre <strong>of</strong> Excellence for Free Radical Chemistry and Biotechnology<br />
is home to over 140 researchers, drawing together a unique grouping <strong>of</strong><br />
fundamental chemists, medicinal chemists, biochemists, biologists and<br />
materials scientists dedicated to the understanding and application <strong>of</strong> free<br />
radical chemistry.<br />
The Centre's research program is broad, encompassing aspects <strong>of</strong> free<br />
radical chemistry and biotechnology from the fundamental to the applied.<br />
Its interdisciplinary approach has and will continue to provide significant<br />
advancements in areas such as: pharmaceuticals, custom-designed plastics,<br />
new technology for the preservation <strong>of</strong> paints, plastics and culturally significant<br />
artwork, the characterisation <strong>of</strong> free radicals in the atmosphere and their affect<br />
on health, and "virtual laboratory" free radical chemistry using supercomputers.<br />
The Centre's research activities can be divided into eight research areas:<br />
Fundamental Radical Chemistry, Good Health & Preventing Disease, Radicals in<br />
the Environment, Radicals in Materials Technology, Kinetics & Mechanisms<br />
Facility, Pain & Inflammation, Surface Coatings, Climate Change & Energy and<br />
Biotechnology<br />
http://www.freeradical.org.au/
Name badge Sponsor<br />
Conference Sponsors<br />
NewSpec specialises in the sales and service <strong>of</strong> high-end scientific research<br />
equipment manufactured by leading international companies including:<br />
• Bruker Nano (formerly Veeco Metrology products)<br />
• Newport Corporation<br />
• Spectra-Physics Lasers<br />
• Fischer-Cripps Laboratories<br />
NewSpec’s comprehensive product portfolio includes:<br />
• Scanning Probe & Atomic Force Microscopes<br />
• Surface Pr<strong>of</strong>ilers; Confocal, Optical & Stylus models<br />
• Nano Indentation Systems<br />
• Desktop TEM/SEM systems<br />
• Lasers, Solar Simulators & Light Sources<br />
• Optical Tables & Vibration Isolation Systems<br />
• Spectroscopy, Radiometers & Optical Measurement Systems<br />
• Optics & Mechanics<br />
• Motorised & Manual Positioning Systems<br />
Contact details: Neil McMahon<br />
Tel: 08 8463 1967 Fax: 08 8410 8816<br />
sales@newspec.com.au www.newspec.com.au<br />
134 Gilbert Street, Adelaide, South Australia, 5000<br />
Computation Chemistry Workshop Sponsor<br />
Smart Infrastructure Facility<br />
The Australian community has an expectation that our cities and regions benefit<br />
from long term, holistic and strategically planned infrastructure. To address this<br />
matter <strong>of</strong> national significance the Australian Government, NSW Government and<br />
<strong>University</strong> <strong>of</strong> <strong>Wollongong</strong> partnered to establish the SMART Infrastructure Facility<br />
with $62 million in funding.<br />
The purpose <strong>of</strong> SMART is to develop a new branch <strong>of</strong> research called ‘integrated<br />
infrastructure planning and management’. Our mission is to generate, publish<br />
and disseminate ideas that support greater understanding <strong>of</strong> the interconnection<br />
and interdependencies <strong>of</strong> infrastructure.<br />
SMART stands for ‘Simulation, Modeling, Analysis, Research and Teaching’ and<br />
our work approach is multidisciplinary and collaborative.<br />
SMART has established Australia’s first Pr<strong>of</strong>essorial chairs in infrastructure<br />
governance, infrastructure systems, infrastructure modelling and simulation, and<br />
appointed a pr<strong>of</strong>essor <strong>of</strong> infrastructure economics.<br />
A modern and sustainable, purpose built, four storey facility has been developed<br />
at the <strong>University</strong> <strong>of</strong> <strong>Wollongong</strong> campus housing 30 integrated laboratories,<br />
simulation and modelling hub, rail logistics research centre and 200 higher<br />
degree research students.<br />
SMART is one <strong>of</strong> the largest facilities <strong>of</strong> its type in the world.<br />
http://smart.uow.edu.au/<br />
157
Conference Sponsors<br />
158<br />
Keynote Speaker Sponsor<br />
ARC Centre <strong>of</strong> Excellence for Electromaterials Science<br />
The Australian Research Council (ARC) Centre <strong>of</strong> Excellence for Electromaterials<br />
Science (ACES) comprises currently <strong>of</strong> six research organisations, whose<br />
common goal is to explore electromaterials and how they can be applied to the<br />
way we live. Established in 2005, ACES expands on the research program <strong>of</strong><br />
the ARC Centre for Nanostructured Electromaterials and draws together<br />
researchers from a range <strong>of</strong> disciplines, including biologists, clinicians, chemists,<br />
physicists and engineers.<br />
The ARC Centre <strong>of</strong> Excellence for Electromaterials Science aims to:<br />
• Expand upon our reputation in electromaterials science and to see the centre<br />
recognised as a world leader in the area.<br />
• Explore the science <strong>of</strong> nanomaterials having an electron or charge transfer<br />
functionality; to prepare such nanomaterials, study and develop theories for their<br />
behaviour and exploit these new behaviours in a number <strong>of</strong> selected<br />
applications.<br />
Our specific objectives are:<br />
• To create new nanostructured electrode materials based on organic materials<br />
such as inherently conductive polymers (ICPs), carbon nanotubes, fullerenes or<br />
hybrid nanocomposites containing them.<br />
• To develop and apply theory and computer modelling techniques in order to<br />
understand structure and transport behaviour <strong>of</strong> nanocomponents and<br />
nano-assemblies.<br />
• To determine the effect <strong>of</strong> nanostructure on ion transport as relates to ionic<br />
conductivity in solids and ionic liquids.<br />
• To apply this knowledge and fabrication protocols to deliver greatly enhanced<br />
performance for energy conversion/storage systems and novel energy transfer<br />
systems in bioapplications.
Trade Exhibitors<br />
Conference Sponsors<br />
ATA Scientific<br />
ATA Scientific specialises in sales, support and service for a range <strong>of</strong> specialised<br />
analytical instruments. Our suite <strong>of</strong> products is focused in the areas <strong>of</strong><br />
Biotechnology, Particle Sciences and Spectroscopy.<br />
Some <strong>of</strong> our innovative suppliers and instruments include:<br />
Malvern Particle size, particle shape, zeta potential, molecular weight,<br />
rheological properties and advanced SEC/GPC chromatography<br />
solutions<br />
Q-Sense Sensors for bio-surface structure and thin film interactions<br />
BioNavis SPR Navi 200A - low cost, high sensitivity Surface Plasmon<br />
Resonance (SPR)<br />
Attension Surface tension and Contact angle measurement<br />
KSV NIMA Instruments for Thin film fabrication and characterisation<br />
Jasco Advanced Spectroscopy and Chromatography solutions<br />
www.atascientific.com.au<br />
ATA Scientific, PO Box 2172, Taren Point, NSW, 2229<br />
enquiries@atascientific.com.au Tel: (02) 9541 3500 Fax: (02) 9525 7166<br />
facebook: www.facebook.com/ATAScientific<br />
Bio Scientific Australia<br />
Service to Science since 1972, BIOSCIENTIFIC PTY. LTD., provides Life Science<br />
Researchers with a comprehensive range <strong>of</strong> specialty chemicals, antibodies,<br />
stem cell products, cytokines, fluorescent reagents, radiolabelled products,<br />
peptides and bio & organic chemicals.<br />
BioScientific continues to source the best quality and guaranteed products.<br />
www.biosci.com.au 1300 BIOSCI ( 246724 )<br />
159
Conference Sponsors<br />
160<br />
eDAQ<br />
eDAQ is a Sydney based company that manufactures and distributes a range <strong>of</strong><br />
electrochemical instrumentation including:<br />
• high resolution INPHAZE electrical impedance spectrometers for surface and<br />
membrane studies<br />
• SDx Tethered Membrane systems for the examination <strong>of</strong> membrane ion<br />
channels<br />
• eDAQ potentiostats for voltammetric and similar experiments<br />
• eDAQ contactless conductivity detectors for capillary electrophoresis and ion<br />
chromatography<br />
• low cost eDAQ isoPods for monitoring <strong>of</strong> pH, conductivity, dissolved oxygen,<br />
amperometric sensors, etc.<br />
as well as e-corder data acquisition systems for general purpose signal<br />
monitoring.<br />
eDAQ Pty Ltd<br />
6 Doig Avenue, Denistone East, NSW 2112, Australia<br />
Phone: +61 2 9807 8855 Fax: +61 2 9807 8844<br />
Email: info@edaq.com http://www.edaq.com<br />
GE Healthcare<br />
GE Healthcare Life Sciences’ purpose is to help make the world healthier<br />
through imaginative science. Our broad laboratory instrument portfolio <strong>of</strong>fers<br />
advanced technologies for protein purification, drug discovery, biomolecular<br />
interaction, stability parameters and cellular analysis. Iconic brands as AKTA,<br />
Biacore, Microcal, Fuji, Typhoon and IN-Cell ultimately enable researchers to gain<br />
deeper insights into protein activity and function, cell biology and how potential<br />
drugs work. With a host <strong>of</strong> consumables available through distribution partners,<br />
your research needs are all consolidated at the one place. Our goal is to enable<br />
Australian scientists to discover, develop and produce therapeutics to prevent,<br />
diagnose, treat and cure disease.<br />
For further information please contact:<br />
GE Healthcare Australia Pty Limited<br />
Phone: 1800 150 522 Fax: 1300 434 232<br />
Email : sales.au@ge.com
Conference Sponsors<br />
Lastek<br />
Lastek’s scientific staff form a key group <strong>of</strong> experienced scientists representing a<br />
broad range <strong>of</strong> companies <strong>of</strong>fering state <strong>of</strong> the art equipment, and supporting<br />
cutting edge technologies. Based on the Thebarton campus <strong>of</strong> Adelaide<br />
<strong>University</strong>, Lastek has been supporting the Australian research market for the<br />
past 23 years.<br />
Areas <strong>of</strong> expertise extend from laser excitation sources through to photon<br />
sensitive detectors for routine microscopy and complex spectroscopic<br />
applications alike. Key installations include multi-photon and higher order<br />
harmonic microscopes, Raman spectroscopy for the analysis <strong>of</strong> biological<br />
samples, and time correlated single photon counting fluorescence spectroscopy.<br />
http://www.lastek.com.au/<br />
Mettler Toledo<br />
Compliance, traceability, consistency, and reliability <strong>of</strong> measurement data are<br />
primary concerns in the fields <strong>of</strong> academia and research. METTLER TOLEDO's<br />
solutions cover the basic measurement needs in all scientific work, and can be<br />
expanded into automated solutions. METTLER TOLEDO instruments are used in<br />
research, scientific and quality control labs among many others, in the<br />
pharmaceutical, chemical, food and cosmetics industries. Our scope <strong>of</strong> solutions<br />
range from standard laboratory equipment such as balances, pipettes and pH<br />
meters, through titrators, instruments for thermal analysis, melting point and<br />
density meters, to solutions for the field <strong>of</strong> automated chemistry<br />
www.mt.com<br />
NewSpec specialises in the sales and service <strong>of</strong> high-end scientific research<br />
equipment manufactured by leading international companies including:<br />
• Bruker Nano (formerly Veeco Metrology products)<br />
• Newport Corporation<br />
• Spectra-Physics Lasers<br />
• Fischer-Cripps Laboratories<br />
NewSpec’s comprehensive product portfolio includes:<br />
• Scanning Probe & Atomic Force Microscopes<br />
• Surface Pr<strong>of</strong>ilers; Confocal, Optical & Stylus models<br />
• Nano Indentation Systems<br />
• Desktop TEM/SEM systems<br />
• Lasers, Solar Simulators & Light Sources<br />
• Optical Tables & Vibration Isolation Systems<br />
• Spectroscopy, Radiometers & Optical Measurement Systems<br />
• Optics & Mechanics<br />
• Motorised & Manual Positioning Systems<br />
Contact details: Neil McMahon<br />
Tel: 08 8463 1967 Fax: 08 8410 8816<br />
sales@newspec.com.au www.newspec.com.au<br />
134 Gilbert Street, Adelaide, South Australia, 5000<br />
161
Conference Sponsors<br />
162<br />
Perkin Elmer<br />
A healthier future starts with the work we do today<br />
At PerkinElmer, we design, manufacture and deliver advanced technology<br />
solutions that address the world's most critical health and safety concerns,<br />
including maternal and fetal health, clean water and air, and safe food and toys.<br />
Our expertise combines science, innovation and a culture <strong>of</strong> operational<br />
excellence to <strong>of</strong>fer our customers technology services and support that improve<br />
the quality <strong>of</strong> people's lives worldwide.<br />
Our work in environmental health improves the quality and sustainability <strong>of</strong> our<br />
environment, and the security <strong>of</strong> people in the places where we live, work and<br />
play. This includes providing the analytical instrumentation, and lighting and<br />
sensor technologies and services that ensure clean air and water; safe food and<br />
consumer products; and efficient, renewable energy - the essential components<br />
<strong>of</strong> a healthier, safer today and tomorrow.<br />
www.perkinelmer.com.au<br />
Scitech<br />
SciTech Pty Ltd established in 1989, <strong>of</strong>fers a complete range <strong>of</strong> innovative<br />
products in the fields <strong>of</strong> Nanotechnology, Spectroscopy, Imaging and<br />
Microscopy.<br />
Some <strong>of</strong> the latest key product developments include:<br />
• The Nanowizard 3 from JPK Instruments; a high resolution and highly flexible<br />
AFM designed for both Life Science and NanoScience applications.<br />
• The JPK NanoTracker, an Optical Tweezer system which can track particles in<br />
3D space and measure interactive forces between particles down to<br />
picoNewtons.<br />
• For Researchers with ideas larger than their budget we <strong>of</strong>fer the new ultra low<br />
cost “TT-AFM” Atomic force microscope.<br />
• NanoIR combines AFM with IR spectroscopy and temperature measurement,<br />
with a resolution <strong>of</strong> 100 nm!<br />
• The Nanoink NLP2000 and DPN5000, DPN instruments for Nan<strong>of</strong>abrication<br />
applications. Features include multiplexing capability, fast prototyping and the<br />
ability to print a variety <strong>of</strong> protein inks with dot sizes from 100 nm – 10 um,<br />
• CellZscope from NanoAnalytics measures the impedance <strong>of</strong> barrier-forming cell<br />
cultures grown on permeable membranes.<br />
• The Till iMic, a compact inverted DIGITAL microscope for high end microscopy<br />
applications. The iMic is very compact, <strong>of</strong>fers greater stability, versatility,<br />
increased light throughput and faster operating speeds compared to a<br />
conventional inverted microscope.
Conference Sponsors<br />
• Featuring also the Andor range <strong>of</strong> CCD, EMCCD, InGas and sCMOS cameras<br />
for spectroscopy and high end Microscopy, Super Resolution and confocal<br />
applications. Andor also <strong>of</strong>fers the Revolution XD and DSD spinning disk<br />
confocal systems.<br />
For Further information Contact<br />
Christian Loebbe : 0400 440 426 christian@scitech.com.au<br />
Con Sapounas: 0400 440 499 con@scitech.com.au<br />
Lino Montuno: 0400 440 488 lino@scitech.com.au<br />
Supporters<br />
163