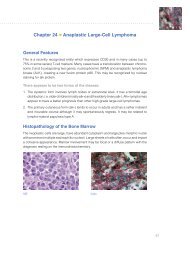
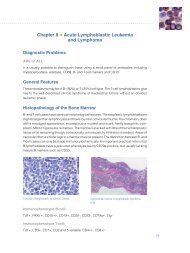
Special Stains Use in Fungal Infections - Dako
Special Stains Use in Fungal Infections - Dako
Special Stains Use in Fungal Infections - Dako
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
<strong>Special</strong> <strong>Sta<strong>in</strong>s</strong> <strong>Use</strong> <strong>in</strong> <strong>Fungal</strong> <strong>Infections</strong><br />
Abida Haque, MD<br />
Professor of Pathology<br />
Weill Medical School of Cornell University, New York<br />
Attend<strong>in</strong>g Surgical Pathologist –<br />
Department of Pathology Methodist Hospital<br />
Houston, TX, USA<br />
Histologic evaluation of tissues is a quick and easy way to identify<br />
fungal organisms, and a strong adjunct to microbiologic culture for<br />
diagnosis of fungal <strong>in</strong>fections. Histologic evaluation of granulomatous<br />
<strong>in</strong>flammation and granulomas must <strong>in</strong>clude special sta<strong>in</strong>s to exclude or<br />
<strong>in</strong>clude presence of fungi and acid fast bacteria. Gomori Methenam<strong>in</strong>e<br />
Silver (GMS) and Periodic acid-Schiff (PAS) are the two most common<br />
sta<strong>in</strong>s used to look for fungi <strong>in</strong> tissues and <strong>in</strong> cytology specimens <strong>in</strong> the<br />
daily practice of pathology. The presence of fungus <strong>in</strong> the tissue sections<br />
provides an <strong>in</strong>disputable evidence of <strong>in</strong>vasive <strong>in</strong>fection. Because of<br />
their size and morphologic diversity, many fungi can be seen <strong>in</strong> tissue<br />
sections by conventional light microscopic exam<strong>in</strong>ation of Hematoxyl<strong>in</strong><br />
and Eos<strong>in</strong> (H&E) sta<strong>in</strong>ed sections. In cytology specimens, fungi can be<br />
identified by their size and specific morphology.<br />
In the tissues, fungi usually occur either as hyphae, budd<strong>in</strong>g yeast,<br />
endosporulat<strong>in</strong>g spherules, or a comb<strong>in</strong>ation of these forms (1, 2). In some<br />
groups of fungi only one species of fungus is the cause of mycosis, and<br />
therefore when classic forms are observed, an etiologic diagnosis can<br />
be made. These groups of fungal diseases <strong>in</strong>clude adiaspiromycosis,<br />
blastomycosis, coccidioidomycosis, cryptococcosis, Histoplasmosis<br />
capsulati, Histoplasmosis duboisii, paracoccidioidomycosis, Penicilliosis<br />
marneffei, protothecosis, rh<strong>in</strong>osporidiosis, and sporotrichosis.<br />
Other mycoses are caused by any of the several species of a genus,<br />
all of which are morphologically similar <strong>in</strong> tissue sections. Although<br />
these fungi cannot be identified as to the species by conventional<br />
histology, the disease that they cause can be diagnosed generically;<br />
Application<br />
for example, aspergillosis, candidiasis, and trichosporonosis. Still other<br />
mycoses are caused by any of a number of fungi belong<strong>in</strong>g to different<br />
genera. These fungi appear similar, if not identical to one another <strong>in</strong><br />
tissues. With these fungi, it is not possible to identify the etiologic agent,<br />
however, the mycosis can be named; for example, phaeohyphomycosis<br />
and zygomycosis.<br />
Hematoxyl<strong>in</strong> & Eos<strong>in</strong> is a versatile sta<strong>in</strong> that enables the pathologist<br />
to evaluate the host response, <strong>in</strong>clud<strong>in</strong>g the Splendore-Hoeppli<br />
phenomenon, and to detect other micro-organisms (3). It is the sta<strong>in</strong><br />
of choice to confirm the presence of naturally pigmented fungi, and to<br />
demonstrate the nuclei of yeast-like cells. However, there are drawbacks<br />
to us<strong>in</strong>g just the H&E sta<strong>in</strong> for fungal diagnosis. It is often difficult to<br />
dist<strong>in</strong>guish poorly sta<strong>in</strong>ed fungi from tissue components, even at higher<br />
magnifications. When sparse, fungi are easily overlooked <strong>in</strong> H&E sta<strong>in</strong>ed<br />
sections. The morphologic features may not be evident and sometimes<br />
may be mislead<strong>in</strong>g. For example, Histoplasma, Blastomyces, and<br />
Paracoccidiodes may have cytoplasmic retraction artifact <strong>in</strong> the<br />
sections, mak<strong>in</strong>g morphologic evaluation difficult. Some of the fungal<br />
variants may have different sizes, such as the large form variant (African)<br />
histoplasma, and microform blastomycosis. Some of the dimorphic fungi<br />
can form pseudohyphae <strong>in</strong> tissues. Sometimes the fungal morphology<br />
may be altered by therapy. <strong>Special</strong> sta<strong>in</strong>s for fungi are therefore essential<br />
for histopathologic evaluation of unexpla<strong>in</strong>ed <strong>in</strong>flammatory processes<br />
(4, 5). Most fungi can be readily demonstrated with the common special<br />
sta<strong>in</strong>s, Gomori’s methenam<strong>in</strong>e silver (GMS), Gridley’s fungus (GF), and<br />
periodic acid-Schiff (PAS), also referred to as “broad spectrum” fungal<br />
�<br />
Connection 2010 | 187
188 | Connection 2010<br />
Figure 1. Hematoxyl<strong>in</strong> and Eos<strong>in</strong> sta<strong>in</strong><strong>in</strong>g of<br />
Aspergillus.<br />
Figure 2. GMS sta<strong>in</strong><strong>in</strong>g of Aspergillus.
Figure 3. Hematoxyl<strong>in</strong> and Eos<strong>in</strong> sta<strong>in</strong><strong>in</strong>g of<br />
Pneumocystis jirovici.<br />
Figure 4. GMS sta<strong>in</strong><strong>in</strong>g of<br />
Pneumocystis jirovici.<br />
�<br />
Connection 2010 | 189
190 | Connection 2010<br />
Figure 5. Alcian Blue sta<strong>in</strong><strong>in</strong>g<br />
of Cryptococcus.<br />
Figure 6. PAS sta<strong>in</strong><strong>in</strong>g of Cryptococcus.
Figure 7. Hematoxyl<strong>in</strong> and Eos<strong>in</strong> sta<strong>in</strong><strong>in</strong>g of<br />
Malassezia.<br />
Figure 8. PAS sta<strong>in</strong><strong>in</strong>g of Malassezia.<br />
�<br />
Connection 2010 | 191
192 | Connection 2010<br />
Figure 9. Hematoxyl<strong>in</strong> and Eos<strong>in</strong> sta<strong>in</strong><strong>in</strong>g of<br />
Histoplasma.<br />
Figure 10. AFB sta<strong>in</strong><strong>in</strong>g of Histoplasma.
“ Gomori Methenam<strong>in</strong>e Silver (GMS) and<br />
Periodic Acid-Schiff (PAS) are the two most<br />
common sta<strong>in</strong>s used to look for fungi <strong>in</strong> tissues<br />
and <strong>in</strong> cytology specimens <strong>in</strong> the daily practice<br />
of pathology. ”<br />
sta<strong>in</strong>s. GMS is preferred for screen<strong>in</strong>g, because it gives better contrast,<br />
and sta<strong>in</strong>s even degenerated and nonviable fungi that are sometimes<br />
refractory to the other two sta<strong>in</strong>s (Fig. 1, 2).<br />
GMS also sta<strong>in</strong>s algae (Prototheca and Chlorella spp.), cyst walls of<br />
Pneumocystis jiroveci (Fig. 3, 4), pathogenic free liv<strong>in</strong>g soil amebas,<br />
the spore coat of most microsporidian parasites, <strong>in</strong>tracytoplasmic<br />
granular <strong>in</strong>clusions of Cytomyeolovirus, Act<strong>in</strong>omyces Israeli and related<br />
species, Nocardia spp., most Mycobacterium spp., and nonfilamentous<br />
bacteria with polysaccharide capsules such as Klebsiella pneumoniae<br />
and Streptococcus pneumoniae. Prolonged sta<strong>in</strong><strong>in</strong>g <strong>in</strong> the silver nitrate<br />
solution may be required to adequately demonstrate degenerated<br />
fungal elements such as the yeast-like cells of Histoplasma capsulatum<br />
var. capsulatum <strong>in</strong> granulomas.<br />
The disadvantage of GMS and GF fungal sta<strong>in</strong>s is that they mask the<br />
natural color of pigmented fungi, mak<strong>in</strong>g it impossible to determ<strong>in</strong>e<br />
whether a fungus is colorless hyal<strong>in</strong>e or dematiaceous (pigmented).<br />
Such a determ<strong>in</strong>ation is crucial <strong>in</strong> the histologic diagnosis of mycosis<br />
caused by dematiaceous fungi such as phaeohyphomycosis (6). Except<br />
for the PAS reaction, fungal sta<strong>in</strong>s GMS and GF do not adequately<br />
demonstrate the <strong>in</strong>flammatory response to fungal <strong>in</strong>vasion. To counteract<br />
this, a GMS-sta<strong>in</strong>ed section can be countersta<strong>in</strong>ed with H&E for a<br />
simultaneous study of the fungus and the host response.<br />
The PAS sta<strong>in</strong> performs almost as well as GMS, <strong>in</strong> screen<strong>in</strong>g for fungi.<br />
It actually demonstrates fungal morphology better than the silver sta<strong>in</strong>s.<br />
PAS can sta<strong>in</strong> degenerated fungi that may not be visible on H&E sta<strong>in</strong>.<br />
Calcific bodies that are sometimes found <strong>in</strong> caseat<strong>in</strong>g granulomas are<br />
also sta<strong>in</strong>ed with PAS, and can be mistaken for yeast-like fungi. This<br />
is especially true when calcific bodies are apposed to give the false<br />
impression of budd<strong>in</strong>g yeasts, or when the bodies are lam<strong>in</strong>ated to give<br />
the appearance of a capsule or thick cell wall. Best sta<strong>in</strong>s to avoid this<br />
mis<strong>in</strong>terpretation are GMS and GF sta<strong>in</strong>s, because the chromic acid<br />
used as an oxidizer <strong>in</strong> these sta<strong>in</strong>s dissolves the calcium, leav<strong>in</strong>g the<br />
calcific bodies unsta<strong>in</strong>ed. Conversely, there are artifacts that mimic fungi<br />
on GMS and GF sta<strong>in</strong>s that are not seen on PAS sta<strong>in</strong>, therefore the use<br />
of both silver and PAS can reduce the <strong>in</strong>cidence of false positive results.<br />
Narrow-Spectrum <strong>Fungal</strong> <strong>Sta<strong>in</strong>s</strong><br />
The differential diagnosis of fungi may require the use of additional<br />
special sta<strong>in</strong>s that sta<strong>in</strong> some fungal organism and not others. These<br />
are sometimes referred to as “narrow-spectrum” fungal sta<strong>in</strong>s (7, 8).<br />
Some of the sta<strong>in</strong>s <strong>in</strong> this category are muc<strong>in</strong> sta<strong>in</strong>s such as alcian blue<br />
and Mayer’s, or Southgate’s mucicarm<strong>in</strong>e, that readily demonstrate the<br />
mucoid capsule of Cryptococcus neoformans (Fig. 5, 6).<br />
This sta<strong>in</strong><strong>in</strong>g reaction differentiates Cryptococcus from other fungi of<br />
similar morphology, such as Coccidiodes, Candida, and Histoplasma.<br />
These muc<strong>in</strong> sta<strong>in</strong>s are not specific for C. neoformans; the cell walls<br />
of B. dermatitidis and Rh<strong>in</strong>osporidium seeberi are often sta<strong>in</strong>ed<br />
to vary<strong>in</strong>g degrees with muc<strong>in</strong> sta<strong>in</strong>s. However, these two fungi<br />
are nonencapsulated and morphologically dist<strong>in</strong>ct, and not ord<strong>in</strong>arily<br />
mistaken for Cryptococcus. In some cases, poorly encapsulated<br />
cryptococci <strong>in</strong> tissue sections may not sta<strong>in</strong> positive with mucicarm<strong>in</strong>e<br />
sta<strong>in</strong>. In these cases, s<strong>in</strong>ce the cell wall of C. neoformans conta<strong>in</strong>s silver<br />
reduc<strong>in</strong>g substances, possibly melan<strong>in</strong> precursors, it can be sta<strong>in</strong>ed<br />
with Fontana-Masson’s silver procedure for melan<strong>in</strong> (9, 10). This sta<strong>in</strong> is<br />
especially useful <strong>in</strong> those cases of Cryptococcosis with <strong>in</strong>vasive yeast<br />
forms that do not have readily detectable capsules, the so-called dry<br />
variants. Such forms could possibly be confused with non-encapsulated<br />
yeasts of similar morphology. Fontana-Masson and Lillie’s ferrous iron<br />
sta<strong>in</strong>s for melan<strong>in</strong> can also be used to confirm and accentuate the<br />
presence of melan<strong>in</strong> or melan<strong>in</strong>-like pigments <strong>in</strong> the cell walls of poorly<br />
pigmented agents of phaeohyphomycosis <strong>in</strong> tissue sections (11). PAS<br />
may be used as a narrow-spectrum fungus sta<strong>in</strong>. For example, <strong>in</strong> the<br />
differential diagnosis of small budd<strong>in</strong>g yeast forms, a weak PAS and a<br />
strong GMS sta<strong>in</strong><strong>in</strong>g favors a diagnosis of Histoplasma, s<strong>in</strong>ce Candida,<br />
microforms of Blastomyces, and yeast forms of Malassezia show a<br />
strong cell wall sta<strong>in</strong><strong>in</strong>g with PAS (Fig. 7, 8).<br />
Another narrow-spectrum fungus sta<strong>in</strong> is Ziehl-Neelson (ZN). In one<br />
study, 60% of blastomyces and 47% of histoplasma organism showed<br />
positive cytoplasmic sta<strong>in</strong><strong>in</strong>g of the yeast-like cells with ZN sta<strong>in</strong><br />
(Fig. 9, 10).<br />
�<br />
Connection 2010 | 193
No sta<strong>in</strong><strong>in</strong>g was seen <strong>in</strong> Cryptococcus or Candida, and very rare acid<br />
fast sta<strong>in</strong><strong>in</strong>g was seen <strong>in</strong> coccidiodomyces endospores (12). However,<br />
these sta<strong>in</strong><strong>in</strong>g properties are <strong>in</strong>consistent and should not be used for<br />
primary diagnosis. The cell walls of fungi are <strong>in</strong> general, not acid fast.<br />
Autoflourescent Fungi<br />
Some fungi or fungal components <strong>in</strong> the H&E sta<strong>in</strong>ed tissue sections<br />
are autofluorescent when exam<strong>in</strong>ed under ultraviolet light source<br />
(13). Candida species, Coccidioides immitis and Aspergillus species<br />
can exhibit bright green to yellow-green autofluorescence (14). When<br />
sections of these fungi are sta<strong>in</strong>ed with the PAS, bright yellow fungal<br />
autofluorescence aga<strong>in</strong>st a deep red-orange background is seen (15).<br />
Autofluorescence may help del<strong>in</strong>eate sparse or poorly sta<strong>in</strong>ed fungi <strong>in</strong><br />
H&E sta<strong>in</strong>ed sections, however this property is <strong>in</strong>consistent and should<br />
not be used for def<strong>in</strong>itive diagnosis. Most fungi <strong>in</strong> frozen or paraff<strong>in</strong><br />
embedded tissue sections also sta<strong>in</strong> nonspecifically with Calcofluor<br />
white, a cotton whitener that fluoresces under ultraviolet light (16). This<br />
rapid and a simple fluorescence procedure can be rout<strong>in</strong>ely used <strong>in</strong> the<br />
<strong>in</strong>traoperative exam<strong>in</strong>ation of fresh-frozen tissues for fungi.<br />
Immunoperoxidase sta<strong>in</strong>s can be used to identify certa<strong>in</strong> fungi <strong>in</strong><br />
smears and <strong>in</strong> formal<strong>in</strong> fixed, paraff<strong>in</strong> embedded tissue section. This<br />
technique, however, has only limited diagnostic use (17).<br />
References<br />
1. Chandler FW, Watts JC. Pathologic Diagnosis of <strong>Fungal</strong><br />
<strong>Infections</strong>. Chicago: ASCP Press; 1987.<br />
2. Haque AK, McG<strong>in</strong>nis MR. Chapter 10. Dail and<br />
Hammar’s Pulmonary Pathology. New York: Spr<strong>in</strong>ger;<br />
2008<br />
3. Liber AF, Choi HS. Splendore-Hoeppli phenomenon<br />
about silk sutures <strong>in</strong> tissue. Arch Pathol Lab Med. 1973;<br />
95:217-20.<br />
4. Schwartz J. The diagnosis of deep mycoses by<br />
morphologic methods. Hum Pathol. 1982; 13:519-33.<br />
5. Vacca LL. Laboratory Manual of Histochemistry. New<br />
York: Raven; 1985.<br />
6. Chandler FW, Kaplan W, Ajello L. Color Atlas and Text of<br />
the Histopathology of Mycotic Disease. Chicago: Year<br />
Book Medical; 1980.<br />
7. Youngberg GA, Wallen EDB, Giorgadze TA. Narrow-<br />
Spectrum histochemical sta<strong>in</strong><strong>in</strong>g of fungi. Arch Pathol<br />
Lab Med 2003; 127:1529-1530.<br />
194 | Connection 2010<br />
8. Hussa<strong>in</strong> Z, Mart<strong>in</strong> A, Youngberg GA. Blastmyces<br />
dermatitides with large yeast forms. Arch Pathol Lab<br />
Med 2001; 125:663-664.<br />
9. Kwon-Chung KJ, Hill WB, Bennett E. New, special sta<strong>in</strong><br />
for histopathological diagnosis of cryptococcosis. J Cl<strong>in</strong><br />
Microbiol. 1981; 13:383-87.<br />
10. Lazcano O, Speights VO Jr, Stickler JG, Bilbao JE,<br />
Becker J, Diaz J. Comb<strong>in</strong>ed histochemical sta<strong>in</strong>s <strong>in</strong> the<br />
differential diagnosis of Cryptococcus neoformans. Mod<br />
Pathol 1993; 6:80-84.<br />
11. Wood C, Russel-Bell B. Characterization of pigmented<br />
fungi by melan<strong>in</strong> sta<strong>in</strong><strong>in</strong>g. Am J Dermatopathol. 1983;<br />
5:77-81.<br />
12. Wages DS, Wear DJ. Acid-fastness of fungi <strong>in</strong><br />
blastomycosis and histoplasmosis. Arch Pathol Lab<br />
Med. 1982; 106:440-41.<br />
13. Mann JL. Autofluorescence of fungi: an aid to detection<br />
<strong>in</strong> tissue sections. Am J Cl<strong>in</strong> Pathol. 1983; 79:587-90.<br />
14. Graham AR. <strong>Fungal</strong> autofluorescence with ultraviolet<br />
illum<strong>in</strong>ation. Am J Cl<strong>in</strong> Pathol. 1983; 79:231-34.<br />
Direct Immunofluoresence (IF)<br />
Direct immunofluoresence (IF) can improve the diagnostic capability<br />
of conventional histopathology <strong>in</strong> the diagnosis of fungal diseases<br />
(18). The IF procedure, which can be performed on smears and on<br />
formal<strong>in</strong> fixed paraff<strong>in</strong> embedded tissue sections is helpful <strong>in</strong> confirm<strong>in</strong>g<br />
a presumptive histologic diagnosis, especially when fresh tissues<br />
are not available for culture or when atypical fungus forms are seen.<br />
The Division of Mycotic Diseases, Center for Disease Control, Atlanta<br />
(United States) and others have a broad battery of sensitive and specific<br />
reagents available for identify<strong>in</strong>g the more common pathogenic fungi.<br />
The immunofluoresence procedure has several advantages. F<strong>in</strong>al<br />
identification of an unknown fungus is possible with<strong>in</strong> hours after<br />
H&E and GMS sta<strong>in</strong>ed sections are <strong>in</strong>itially exam<strong>in</strong>ed. The need<br />
for time consum<strong>in</strong>g and costly cultures is often obviated by IF, and<br />
the hazards of handl<strong>in</strong>g potentially <strong>in</strong>fectious materials are reduced<br />
when microorganisms are <strong>in</strong>activated by formal<strong>in</strong> prior to IF sta<strong>in</strong><strong>in</strong>g.<br />
Prolonged storage of formal<strong>in</strong> fixed tissues, either wet tissue or paraff<strong>in</strong><br />
embedded, does not appear to affect the antigenecity of fungi. This<br />
antigenic stability makes possible retrospective studies of paraff<strong>in</strong><br />
embedded tissue and the shipment of specimens to distant reference<br />
laboratories for confirmatory identification. Most service laboratories,<br />
however, do not rout<strong>in</strong>ely use IF, s<strong>in</strong>ce the special sta<strong>in</strong>s have been very<br />
reliable for diagnosis <strong>in</strong> the day to day pathology practice.<br />
15. Jackson JA, Kaplan W, Kaufman L. Development of<br />
fluorescent-antibody reagents for demonstration of<br />
Pseudallescheria boydii <strong>in</strong> tissue. J Cl<strong>in</strong> Microbiol. 1983;<br />
18:668-73.<br />
16. Monheit JE, Cowan DF, Moore DG. Rapid detection of<br />
fungi <strong>in</strong> tissue us<strong>in</strong>g calcofluor white and fluorescence<br />
microscopy. Arch Pathol Lab Med. 1984; 108:616-18.<br />
17. Kobayashi M, Kotani S, Fujishita M, et al.<br />
Immunohistochemical identification of Trichosporon<br />
beigelii <strong>in</strong> histologic sections by Immunoperoxidase<br />
method. Am J Cl<strong>in</strong> Pathol. 1988; 89:100-05.<br />
18. Reiss E, de-Repentgny L, Kuykendall J, et al.<br />
Monoclonal antibodies aga<strong>in</strong>st Candida tropicalis<br />
mannans antigen detection by enzyme immunoassay<br />
and immunofluorescence. J Cl<strong>in</strong> Microbiol. 1986;<br />
24:796-802.