cm m a nm a
cm m a nm a
cm m a nm a
- No tags were found...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
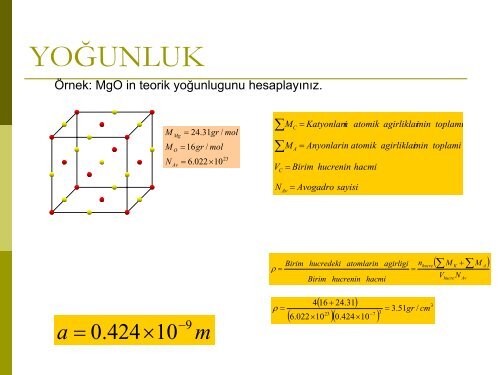
OrOr2YOĞUNLUKÖrnek: MgO in teorik yoğunlugunu hesaplayınız.MMNMgOAv= 24.31gr/ mol= 16gr/ mol= 6.022×1023∑∑VCMMCA=Katyonlari natomik= Anyonlarin atomik= Birim hucrenin ha<strong>cm</strong>iagirliklarininagirliklarinintoplamitoplamiNAv= Avogadrosayisiρ =Birim hucredeki atomlarin agirligi nhucre( ∑MK+ ∑MA)Birimhucreninha<strong>cm</strong>i=VhucreNAva=0.424×10−9mρ =( + 24.31)4 1623−7( 6.022×10 )( 0.424×10 )3= 3.51gr/ <strong>cm</strong>3