Vorlesung 5 - Redox
Vorlesung 5 - Redox
Vorlesung 5 - Redox
Sie wollen auch ein ePaper? Erhöhen Sie die Reichweite Ihrer Titel.
YUMPU macht aus Druck-PDFs automatisch weboptimierte ePaper, die Google liebt.
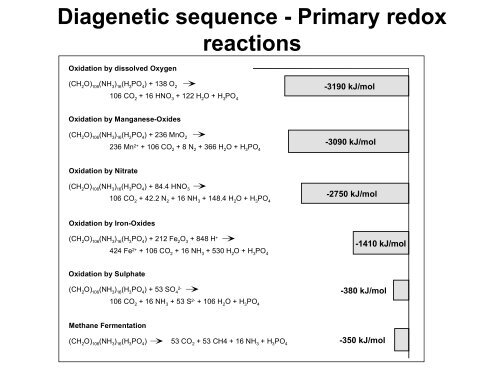
Diagenetic sequence - Primary redox<br />
reactions<br />
Oxidation by dissolved Oxygen<br />
(CH 2<br />
O) 106<br />
(NH 3<br />
) 16<br />
(H 3<br />
PO 4<br />
) + 138 O 2<br />
-3190 kJ/mol<br />
106 CO 2<br />
+ 16 HNO 3<br />
+ 122 H 2<br />
O + H 3<br />
PO 4<br />
Oxidation by Manganese-Oxides<br />
(CH 2 O) 106 (NH 3 ) 16 (H 3 PO 4 ) + 236 MnO 2<br />
236 Mn 2+ + 106 CO 2 + 8 N 2 + 366 H 2 O + H 3 PO 4<br />
-3090 kJ/mol<br />
Oxidation by Nitrate<br />
(CH 2 O) 106 (NH 3 ) 16 (H 3 PO 4 ) + 84.4 HNO 3<br />
106 CO 2<br />
+ 42.2 N 2<br />
+ 16 NH 3<br />
+ 148.4 H 2<br />
O + H 3<br />
PO 4<br />
-2750 kJ/mol<br />
Oxidation by Iron-Oxides<br />
(CH 2<br />
O) 106<br />
(NH 3<br />
) 16<br />
(H 3<br />
PO 4<br />
) + 212 Fe 2<br />
O 3<br />
+ 848 H +<br />
424 Fe 2+ + 106 CO 2<br />
+ 16 NH 3<br />
+ 530 H 2<br />
O + H 3<br />
PO 4<br />
-1410 kJ/mol<br />
Oxidation by Sulphate<br />
(CH 2<br />
O) 106<br />
(NH 3<br />
) 16<br />
(H 3<br />
PO 4<br />
) + 53 SO 4<br />
2-<br />
-380 kJ/mol<br />
106 CO 2<br />
+ 16 NH 3<br />
+ 53 S 2- + 106 H 2<br />
O + H 3<br />
PO 4<br />
Methane Fermentation<br />
(CH 2 O) 106 (NH 3 ) 16 (H 3 PO 4 ) 53 CO 2 + 53 CH4 + 16 NH 3 + H 3 PO 4<br />
-350 kJ/mol