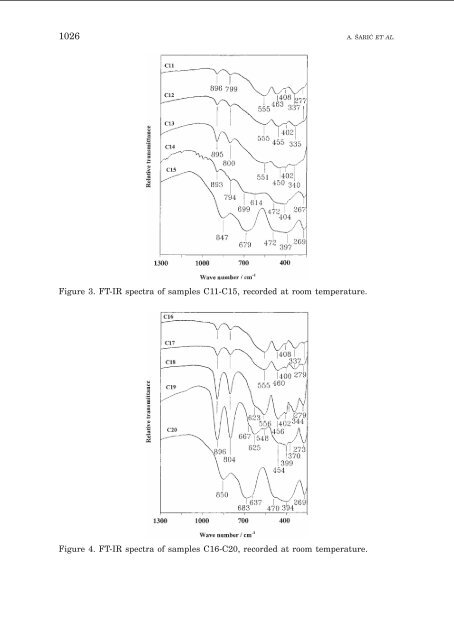
1026 A. ŠARI] ET AL. Figure 3. FT-IR spectra <str<strong>on</strong>g>of</str<strong>on</strong>g> samples C11-C15, recorded at room temperature. Figure 4. FT-IR spectra <str<strong>on</strong>g>of</str<strong>on</strong>g> samples C16-C20, recorded at room temperature.
PRECIPITATION OF IRON OXIDES 1027 Figures 3 and 4 show <strong>the</strong> FT-IR spectra <str<strong>on</strong>g>of</str<strong>on</strong>g> samples C11 to C20, precipitated <strong>from</strong> <strong>the</strong> <strong>FeCl3</strong> soluti<strong>on</strong>s with an initial urotropin c<strong>on</strong>centrati<strong>on</strong> <str<strong>on</strong>g>of</str<strong>on</strong>g> 0.1 M. At c<strong>on</strong>centrati<strong>on</strong>s up to 0.02 M <strong>FeCl3</strong> , mixtures <str<strong>on</strong>g>of</str<strong>on</strong>g> -Fe2O3 and -FeOOH were obtained (samples C11, C12 and C13), whereas at a c<strong>on</strong>centrati<strong>on</strong> <str<strong>on</strong>g>of</str<strong>on</strong>g> 0.05 M <strong>FeCl3</strong> , a mixture <str<strong>on</strong>g>of</str<strong>on</strong>g> -FeOOH, -Fe2O3 and -FeOOH was obtained, similarly as in <strong>the</strong> case <str<strong>on</strong>g>of</str<strong>on</strong>g> sample C3. The FT-IR spectrum <str<strong>on</strong>g>of</str<strong>on</strong>g> sample C15 can be assigned to -FeOOH. After 7 days <str<strong>on</strong>g>of</str<strong>on</strong>g> aging <strong>the</strong> precipitati<strong>on</strong> system C14, <strong>the</strong> -FeOOH fracti<strong>on</strong> significantly increased (IR bands at 896 and 804 cm –1 ). Figures 5 and 6 show <strong>the</strong> FT-IR spectra <str<strong>on</strong>g>of</str<strong>on</strong>g> samples C21 to C30, precipitated <strong>from</strong> <strong>the</strong> <strong>FeCl3</strong> soluti<strong>on</strong>s with an initial urotropin c<strong>on</strong>centrati<strong>on</strong> <str<strong>on</strong>g>of</str<strong>on</strong>g> 0.25 M. For c<strong>on</strong>centrati<strong>on</strong>s up to 0.05 M <strong>FeCl3</strong> and 24 hours <str<strong>on</strong>g>of</str<strong>on</strong>g> aging <str<strong>on</strong>g>of</str<strong>on</strong>g> <strong>the</strong> precipitati<strong>on</strong> systems, mixtures <str<strong>on</strong>g>of</str<strong>on</strong>g> -Fe2O3 and -FeOOH were found, whereas <strong>the</strong> -FeOOH phase was obtained for <strong>the</strong> c<strong>on</strong>centrati<strong>on</strong> <str<strong>on</strong>g>of</str<strong>on</strong>g> 0.3 M <strong>FeCl3</strong> . After 7 days <str<strong>on</strong>g>of</str<strong>on</strong>g> aging, <strong>the</strong> -FeOOH phase dissolved, so <strong>the</strong> dominant phase in sample C30 was -FeOOH (IR bands at 896, 800, 633 cm –1 ), and <strong>the</strong> minor phase was -Fe2O3 (IR shoulders at 573 and 454 cm –1 ). On <strong>the</strong> basis <str<strong>on</strong>g>of</str<strong>on</strong>g> <strong>the</strong> above-presented results, a general rule <strong>on</strong> <strong>the</strong> precipitati<strong>on</strong> <str<strong>on</strong>g>of</str<strong>on</strong>g> ir<strong>on</strong> oxides <strong>from</strong> <strong>FeCl3</strong> soluti<strong>on</strong>s, in <strong>the</strong> presence <str<strong>on</strong>g>of</str<strong>on</strong>g> urotropin, was established. After 24 hours <str<strong>on</strong>g>of</str<strong>on</strong>g> aging <strong>the</strong> precipitati<strong>on</strong> systems in <strong>the</strong> pH regi<strong>on</strong> up to 2, <strong>the</strong> -FeOOH is produced as a single phase, in <strong>the</strong> regi<strong>on</strong> 5 < pH < 6 mixtures <str<strong>on</strong>g>of</str<strong>on</strong>g> -FeOOH, -Fe2O3 and -FeOOH are produced, while at pH > 6 mixtures <str<strong>on</strong>g>of</str<strong>on</strong>g> -Fe2O3 and -FeOOH are observed. With a prol<strong>on</strong>ged time <str<strong>on</strong>g>of</str<strong>on</strong>g> aging (7 days) <strong>the</strong> precipitati<strong>on</strong> systems, -FeOOH, formed at 2.16 < pH < 5.55, was dissolved giving <strong>the</strong> feeding material for -FeOOH growth <strong>on</strong> account <str<strong>on</strong>g>of</str<strong>on</strong>g> -Fe2O3 .AtpH1.5, -FeOOH was found to precipitate as a single phase up to 7 days. At <strong>the</strong> early stage <str<strong>on</strong>g>of</str<strong>on</strong>g> <strong>the</strong> <strong>FeCl3</strong> hydrolysis, formati<strong>on</strong> <str<strong>on</strong>g>of</str<strong>on</strong>g> hydroxy complexes and polymers <str<strong>on</strong>g>of</str<strong>on</strong>g> Fe3+ i<strong>on</strong>s occurs. In <strong>the</strong>se hydroxy polymers, OH – groups are partially substituted for chlorides. 24 With an increase in pH, caused by chemical decompositi<strong>on</strong> <str<strong>on</strong>g>of</str<strong>on</strong>g> urotropin, <strong>the</strong>re is str<strong>on</strong>g competiti<strong>on</strong> between OH – and Cl – i<strong>on</strong>s, and with a prol<strong>on</strong>ged time <str<strong>on</strong>g>of</str<strong>on</strong>g> aging Cl – i<strong>on</strong>s are almost completely eliminated <strong>from</strong> <strong>the</strong> solid hydrolytical product. Only residual Cl – i<strong>on</strong>s (up to 2%) are present4 due to <strong>the</strong> specific structure <str<strong>on</strong>g>of</str<strong>on</strong>g> -FeOOH. Mössbauer Spectroscopy Mössbauer spectroscopy has found an important applicati<strong>on</strong> in <strong>the</strong> investigati<strong>on</strong> <str<strong>on</strong>g>of</str<strong>on</strong>g> ir<strong>on</strong> oxides. 25 For this reas<strong>on</strong>, we have also applied this technique in <strong>the</strong> investigati<strong>on</strong> <str<strong>on</strong>g>of</str<strong>on</strong>g> our samples to obtain additi<strong>on</strong>al informati<strong>on</strong> about <strong>the</strong> nature <str<strong>on</strong>g>of</str<strong>on</strong>g> <strong>the</strong>se samples. Typical results obtained by Mössbauer spectroscopy are summarized in Figures 7, 8 and 9, and in Table II.