Synthesis of β-Vetivone More resources available ... - ChemistforChrist
Synthesis of β-Vetivone More resources available ... - ChemistforChrist
Synthesis of β-Vetivone More resources available ... - ChemistforChrist
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
<strong>Synthesis</strong> <strong>of</strong> <strong>β</strong>-<strong>Vetivone</strong><br />
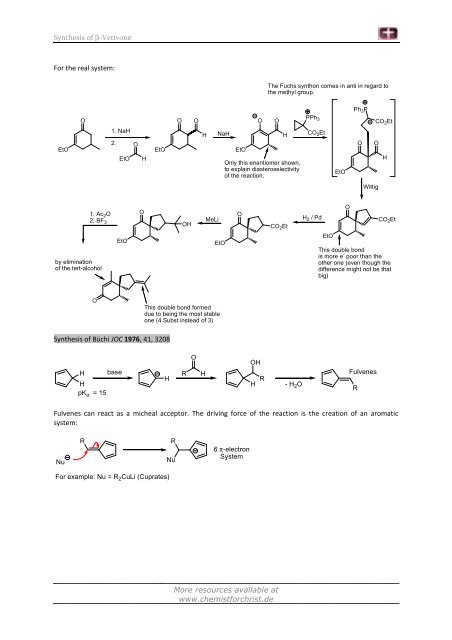
For the real system:<br />
EtO<br />
O<br />
1. Ac 2O<br />
2, BF 3<br />
by elimination<br />
<strong>of</strong> the tert-alcohol<br />
O<br />
1. NaH<br />
2.<br />
O<br />
EtO H<br />
EtO<br />
EtO<br />
<strong>Synthesis</strong> <strong>of</strong> Büchi JOC 1976, 41, 3208<br />
H<br />
H<br />
pKa = 15<br />
base<br />
O<br />
O<br />
OH<br />
O<br />
H<br />
MeLi<br />
NaH<br />
EtO<br />
This double bond formed<br />
due to being the most stable<br />
one (4 Subst instead <strong>of</strong> 3)<br />
H<br />
O<br />
R H<br />
EtO<br />
<strong>More</strong> <strong>resources</strong> <strong>available</strong> at<br />
www.chemistforchrist.de<br />
O<br />
O<br />
H<br />
Only this enantiomer shown,<br />
to explain diasteroselectivity<br />
<strong>of</strong> the reaction.<br />
O<br />
OH<br />
H<br />
R<br />
The Fuchs synthon comes in anti in regard to<br />
the methyl group.<br />
CO 2Et<br />
- H 2O<br />
PPh 3<br />
CO 2Et<br />
H 2 / Pd<br />
EtO<br />
EtO<br />
O<br />
Ph 3P<br />
O<br />
CO 2Et<br />
O<br />
Wittig<br />
H<br />
CO 2Et<br />
This double bond<br />
is more e - poor than the<br />
other one (even though the<br />
difference might not be that<br />
big)<br />
Fulvenes<br />
Fulvenes can react as a micheal acceptor. The driving force <strong>of</strong> the reaction is the creation <strong>of</strong> an aromatic<br />
system:<br />
Nu<br />
R<br />
For example: Nu = R 2CuLi (Cuprates)<br />
R<br />
Nu<br />
6 π-electron<br />
System<br />
R