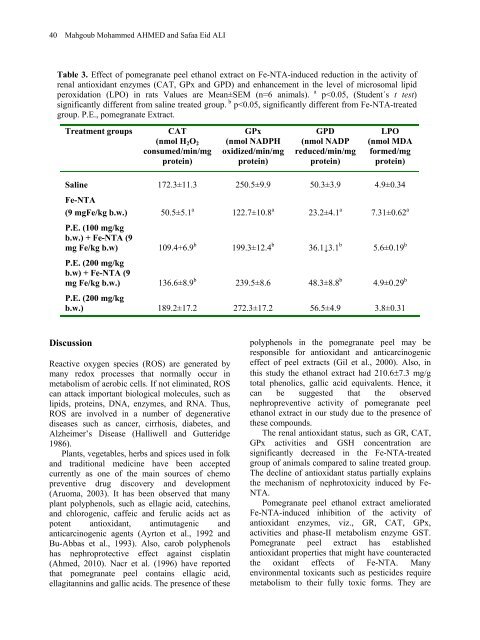
40 Mahgoub Mohammed AHMED and Safaa Eid ALI Table 3. Effect <strong>of</strong> <strong>pomegranate</strong> <strong>peel</strong> <strong>ethanol</strong> <strong>extract</strong> on Fe-NTA-induced reduction in the activity <strong>of</strong> renal antioxidant enzymes (CAT, GPx and GPD) and enhancement in the level <strong>of</strong> microsomal lipid peroxidation (LPO) in rats Values are Mean±SEM (n=6 animals). a p
<strong>of</strong>ten metabolized to proximate toxicants by phase I enzymes, e.g., cytochrome P450 which catalyze oxidative reactions. The oxidized metabolites <strong>of</strong> potentially toxic xenobiotics are then detoxified by Phase II metabolizing enzymes into the forms that are relatively inert and more easily excreted (Talalay et al., 1995). GSH depletion increases the sensitivity <strong>of</strong> organ to oxidative and chemical injury. Studies with a number <strong>of</strong> models show that the metabolism <strong>of</strong> xenobiotics <strong>of</strong>ten produced GSH depletion (Mitchell et al., 1973 and Ahmed and Zaki, 2009). The depletion <strong>of</strong> GSH, also, seems to be the prime factor that permits lipid peroxidation in the Fe- NTA treated group. Pretreatment <strong>of</strong> <strong>pomegranate</strong> <strong>peel</strong> <strong>extract</strong> reduced the depletion <strong>of</strong> GSH levels and provided protection to the kidney. The protection <strong>of</strong> GSH is by forming the substrate for GPx activity that can react directly with various aldehydes produced from the peroxidation <strong>of</strong> membrane lipid. Pomegranate <strong>peel</strong> <strong>extract</strong> pretreatment also reduced the elevated levels <strong>of</strong> serum urea and ceatinine that are marker parameters <strong>of</strong> kidney toxicity. In conclusion, we can say that, the high antioxidant and nephropreventive <strong>effect</strong> <strong>of</strong> the <strong>pomegranate</strong> <strong>peel</strong> <strong>extract</strong> appeared to be attributed to its high phenolics content. The mechanism <strong>of</strong> action <strong>of</strong> <strong>pomegranate</strong> <strong>peel</strong> <strong>extract</strong> may be through induction <strong>of</strong> various antioxidant and phase II enzymes, and scavenging reactive oxygen species. Thus our data suggest that <strong>pomegranate</strong> <strong>peel</strong> <strong>ethanol</strong> <strong>extract</strong> is a potent nephropreventive agent. Further work is required for the isolation and characterization <strong>of</strong> individual phenolic compounds present in <strong>peel</strong> <strong>ethanol</strong> <strong>extract</strong> and to determine the mechanisms involved in the nephropreventive <strong>effect</strong> <strong>of</strong> <strong>pomegranate</strong> <strong>peel</strong> <strong>extract</strong>. References Ahmed MM and Zaki NI. Assessment the ameliorative <strong>effect</strong> <strong>of</strong> <strong>pomegranate</strong> and rutin on chlorpyrifos-ethyl-induced oxidative stress in rats. Nature and Science. 7(10): 49-61, 2009. Ahmed MM. Biochemical studies on nephroprotective <strong>effect</strong> <strong>of</strong> carob (Ceratonia siliqua L.) growing in Egypt. Nature and Science. 8 (3): 41-47, 2010. Ajaikumar K.B, Asheef, M, Babu BH and Padikkala J. The inhibition <strong>of</strong> gastric mucosal injury by Punica granatum L. (<strong>pomegranate</strong>) Pomegranate <strong>peel</strong> <strong>extract</strong> <strong>against</strong> renal oxidative damage 41 m<strong>ethanol</strong>ic <strong>extract</strong>. Journal <strong>of</strong> Ethnopharma. 96: 171–176, 2005. American Institute <strong>of</strong> Nutrition. Report <strong>of</strong> the American Institute <strong>of</strong> Nutrition. Ad Hoc Committee J Nutr. 110: 1340-1348, 1980. Aruoma OI. Methodological considerations for characterizing potential antioxidant actions <strong>of</strong> bioactive components in plant foods. Mutation Research. (523-524): 9-20, 2003. Athar M and Iqbal M. Ferric nitrilotriacetate promotes N-diethylnitrosamine-induced renal tumorigenesis in the rat: implications for the involvement <strong>of</strong> oxidative stress. Carcinogenesis. 19 (6):1133-1139, 1998 Ayrton AD, Lewis DF, Waker R. and Ioannides C. Antimutagenicity <strong>of</strong> ellagic acid towards the food mutagen IQ: investigation into possible mechanisms <strong>of</strong> action. Food and Chemical Toxicology, 3D: 289-295, 1992. Bartles H, Bohmer M and Heieri C. Serum keratinin bestmmung ohno enteeissen. Clinical Chimca Acta. 37: 139-197, 1972. Bu-Abbas A, Clifford MN, Walker R and Ioannides C. Marker antimutagenic potential <strong>of</strong> aquous green tea <strong>extract</strong>s: Mechanism <strong>of</strong> action. Mutagenesis. 9: 325-331, 1994. Carlberg I and Mannervik B. Glutathione level in rat brain. J Biological Chemistry. 250: 4480- 4575, 1975. De Flora S and Ramel C. Mechanism <strong>of</strong> inhibition <strong>of</strong> mutagenesis and carcinogenesis: classification and overview. Mutation Research. 202: 285-306, 1988. De Freitas JM and Meneghini R. Iron and its sensitive balance in the cell. Mutation Research. 475: 153-159, 2001. Deiana M, Aruma OI, Rosa A, Crobu V, Piga R and Derri MA. The <strong>effect</strong> <strong>of</strong> <strong>ferric</strong>nitrilotriacetic acid on the pr<strong>of</strong>ile <strong>of</strong> polyunsaturated fatty acids in the kidney and liver <strong>of</strong> rats. Toxicol Letters. 123: 125-133, 2001. Dhawan BN, Patnaik GK, Rastogi RP, Singh KK and Tandon JS. Screening <strong>of</strong> Indian plants for biological activity. Indian J. Exp. Biol. 15: 208- 219, 1977. Ellman GL. Tissue sulfhydryl groups. Arch Biochem Biophys. 82: 70-77, 1959. Gil MI, Tomas-Barberan FA, Hess Pierce B, Holcr<strong>of</strong>t, DM. and Kader AA. Antioxidant