Selected Reports - Chest - American College of Chest Physicians
Selected Reports - Chest - American College of Chest Physicians
Selected Reports - Chest - American College of Chest Physicians
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
CHEST <strong>Selected</strong> <strong>Reports</strong><br />
Cavitating Invasive Pulmonary<br />
Aspergillosis Visualized and<br />
Diagnosed by Ultrathin<br />
Bronchoscopy*<br />
Masahide Oki, MD; Hideo Saka, MD, FCCP;<br />
Chieko Sako, MD; Shigeru Tanaka, MD;<br />
Yoshihiro Kawata, MD; Chiyoe Kitagawa, MD; and<br />
Nobuyoshi Minemura, MD<br />
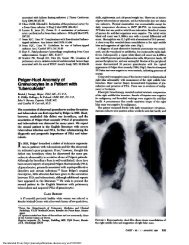
A definitive diagnosis <strong>of</strong> invasive pulmonary aspergillosis<br />
(IPA), which usually occurs in immunocompromised<br />
patients, is <strong>of</strong>ten difficult. We report<br />
two cases <strong>of</strong> cavitating IPA in a peripheral pulmonary<br />
region in patients who were receiving corticosteroids,<br />
in whom the cavity was successfully visualized<br />
and sampled during ultrathin bronchoscopy.<br />
Ultrathin bronchoscopy provides a new option for<br />
definitive diagnosis <strong>of</strong> cavitating IPA.<br />
(CHEST 2006; 129:475–479)<br />
Key words: bronchoscopy; cavity; invasive pulmonary aspergillosis;<br />
transbronchial biopsy; ultrathin bronchoscope<br />
Abbreviations: IPA invasive pulmonary aspergillosis;<br />
TBB transbronchial biopsy<br />
Invasive pulmonary aspergillosis (IPA), which commonly<br />
occurs in immunocompromised patients, is a lifethreatening<br />
infectious disease. Not only are such patients<br />
difficult to treat, the diagnosis is especially difficult.<br />
Although bronchoscopic procedures such as BAL and<br />
transbronchial biopsy (TBB) have been used to establish<br />
the diagnosis <strong>of</strong> IPA, their diagnostic yield is insufficient. 1<br />
The diagnostic yield <strong>of</strong> TBB is particularly low, 2 so its<br />
value for the diagnosis <strong>of</strong> IPA remains controversial. 3<br />
Recently, ultrathin bronchoscopes <strong>of</strong>fering a smaller<br />
outer diameter and higher image quality have been developed.<br />
Such a bronchoscope with an outer diameter <strong>of</strong> 2.8<br />
mm is now commercially available and permits the observation<br />
and manipulation <strong>of</strong> more peripheral bronchi than<br />
was previously possible with a standard bronchoscope. The<br />
*From the Departments <strong>of</strong> Respiratory Medicine (Drs. Oki, Saka,<br />
Sako, Tanaka, Kawata, and Kitagawa) and Internal Medicine (Dr.<br />
Minemura), Nagoya Medical Center, Nagoya, Japan.<br />
Manuscript received July 11, 2005; revision accepted August 20,<br />
2005.<br />
Reproduction <strong>of</strong> this article is prohibited without written permission<br />
from the <strong>American</strong> <strong>College</strong> <strong>of</strong> <strong>Chest</strong> <strong>Physicians</strong> (www.chestjournal.<br />
org/misc/reprints.shtml).<br />
Correspondence to: Masahide Oki, MD, Department <strong>of</strong> Respiratory<br />
Medicine, Nagoya Medical Center, 4-1-1 Sannomaru, Nakaku,<br />
Nagoya 460-0001, Japan; e-mail: Masahideo@aol.com<br />
ultrathin bronchoscope has been reported to be a valuable<br />
diagnostic tool for peripheral pulmonary lesions. 4–6 Although<br />
various diseases can be diagnosed histologically<br />
using the ultrathin bronchoscope, there is, to our knowledge,<br />
no report in the literature about its contribution to<br />
the diagnosis <strong>of</strong> IPA. We herewith report two cases <strong>of</strong> IPA<br />
diagnosed in patients were receiving corticosteroids, in<br />
whom the cavity was visualized and sampled during<br />
ultrathin bronchoscopy.<br />
Case 1<br />
Case <strong>Reports</strong><br />
A 57-year-old man with dermatomyositis was referred for<br />
bronchoscopic evaluation <strong>of</strong> progressive radiographic changes<br />
consisting <strong>of</strong> an enlarging cavity despite antibiotic therapy. For 3<br />
months, he had received prednisolone, 40 to 80 mg/d, and<br />
azathioprine, 100 mg/d, for intractable dermatomyositis. Two<br />
weeks earlier, he complained <strong>of</strong> fever, increased cough, purulent<br />
sputum, and dyspnea. The chest radiograph showed a cavity with<br />
surrounding infiltrates in the upper lobe <strong>of</strong> the right lung.<br />
Sputum cultures grew Pseudomonas aeruginosa and Candida<br />
glabrata. The patient had been treated with IV cefozopran and<br />
fluconazole for 2 weeks without any clinical improvement.<br />
A CT scan <strong>of</strong> the chest the day before bronchoscopy revealed<br />
a thick wall cavity in the anterior segment <strong>of</strong> the right upper lobe<br />
that had not been present 2 months previously (Fig 1). Bronchoscopic<br />
examination using a standard bronchoscope with an<br />
external diameter <strong>of</strong> 6.1 mm (BF-6C240; Olympus; Tokyo, Japan)<br />
[Fig 2] was first performed. The bronchoscope reached the<br />
segmental bronchus <strong>of</strong> the right upper lobe and revealed no<br />
abnormalities. Then, for peripheral investigation, a 2.8 mm in<br />
diameter ultrathin bronchoscope with a 1.2-mm working channel<br />
(BF-XP40; Olympus) [Fig 2] was employed. The ultrathin bronchoscope<br />
was inserted into the right B3 and advanced through<br />
the fifth-generation bronchus under direct observation, and then<br />
entered a cavity, as confirmed by fluoroscopy (Fig 3). A whitish<br />
intracavitary lesion was seen (Fig 4, left, A). Biopsies using a<br />
miniforceps (FB-56D-1; Olympus) and washing were performed<br />
with bronchoscopic visualization, and then amphotericin B, 5 mg,<br />
was instilled into the cavity through the ultrathin bronchoscope<br />
for treatment <strong>of</strong> a presumptive fungal infection. Biopsy specimens<br />
showed septate-branching hyphae suggestive <strong>of</strong> aspergillosis<br />
and cultures <strong>of</strong> the cavital washing grew Aspergillus flavus.<br />
Unfortunately, 9 days later the patient suddenly died <strong>of</strong> cerebral<br />
hemorrhage.<br />
Case 2<br />
A 69-year-old man with systemic lupus erythematosus was<br />
admitted to the hospital with cough, bloody sputum, fatigue,<br />
decreased appetite, and enlarging left lower lobe pulmonary<br />
cavity. For 6 months before hospital admission, he had received<br />
prednisolone, 20 to 30 mg/d, for systemic lupus erythematodes.<br />
Two months earlier, he complained <strong>of</strong> bloody sputum, and a<br />
chest CT showed cavitary infiltrates in the superior segment <strong>of</strong><br />
www.chestjournal.org CHEST / 129 /2/FEBRUARY, 2006 475<br />
Downloaded From: http://journal.publications.chestnet.org/ on 07/13/2013
Figure 1. <strong>Chest</strong> CT on the day before bronchoscopy showed a thick wall cavity in the anterior segment<br />
<strong>of</strong> the right upper lobe (right) that had not been present 2 months previously (left).<br />
the left lower lobe (Fig 5, left). Sputum cultures showed A flavus.<br />
Clinical deterioration with an enlarging cavity had occurred<br />
despite antibiotic treatments including itraconazole (Fig 5, right).<br />
Bronchoscopic examination was performed using a 2.8-mm<br />
ultrathin bronchoscope (BF-XP260F; Olympus), which could be<br />
inserted into the seventh-generation bronchus under direct vision<br />
and then advanced into the cavity. A yellowish white intracavitary<br />
lesion was noted (Fig 4, right, B). Biopsies and washing were<br />
Figure 2. A comparison <strong>of</strong> bronchoscopes. Left: a standard bronchoscope with an external diameter<br />
<strong>of</strong> 6.1 mm and a working channel <strong>of</strong> 2.0 mm (BF-6C240; Olympus); right: an ultrathin bronchoscope<br />
with an external diameter <strong>of</strong> 2.8 mm and a working channel <strong>of</strong> 1.2 mm (BF-XP40; Olympus).<br />
476 <strong>Selected</strong> <strong>Reports</strong><br />
Downloaded From: http://journal.publications.chestnet.org/ on 07/13/2013
Figure 3. Tip <strong>of</strong> the ultrathin bronchoscope shown entering a<br />
cavity.<br />
performed under ultrathin bronchoscopic visualization. Histologic<br />
examination <strong>of</strong> biopsy specimens showed septate-branching<br />
hyphae, and cultures <strong>of</strong> the cavital washing grew Aspergillus<br />
fumigatus. IV amphotericin B was administered at a total dose <strong>of</strong><br />
2,000 mg, and the patient showed clinical improvement with<br />
resolution <strong>of</strong> the extracavitary and intracavitary infiltrates but<br />
persistence <strong>of</strong> a thin wall cavity.<br />
Discussion<br />
IPA occurs mainly in severely immunocompromised<br />
patients, particularly those with prolonged severe granu-<br />
locytopenia. 7 Although the incidence is relatively uncommon,<br />
mildly immunocompromised patients, such as those<br />
receiving corticosteroids similar to our cases, can also<br />
acquire IPA. 8,9 Palmer et al 9 reported six patients with<br />
lung disease receiving corticosteroids who acquired IPA,<br />
and they indicated that IPA should be included in the<br />
differential diagnosis when pneumonia and cavitary infiltrates<br />
occur in such patients.<br />
Although the diagnosis <strong>of</strong> IPA is <strong>of</strong>ten admittedly<br />
difficult, 10 when compatible clinical and radiographic findings<br />
are supported by the presence <strong>of</strong> positive culture<br />
results for Aspergillus species from respiratory secretions,<br />
it is highly likely that the patient has IPA. BAL during<br />
bronchoscopy has proved helpful in diagnosing IPA. In<br />
one review article, 11 it was reported that the diagnostic<br />
sensitivity <strong>of</strong> BAL was 43% in histologically proven IPA.<br />
However, the reported diagnostic sensitivity <strong>of</strong> BAL for<br />
IPA appears to vary with the investigator. Saito et al 12<br />
reported that BAL was diagnostic for pulmonary aspergillosis<br />
in none <strong>of</strong> nine patients with leukemia. Most specific<br />
diagnoses for pulmonary aspergillosis were established<br />
solely from autopsy, so BAL was reportedly useless in most<br />
cases. Moreover, the specificity <strong>of</strong> BAL for IPA is good,<br />
but it may be difficult to exactly distinguish infection from<br />
colonization.<br />
The definitive diagnosis <strong>of</strong> IPA requires histopathologic<br />
demonstration <strong>of</strong> septate acute branching hyphae with<br />
positive culture results for Aspergillus. 13 TBB is a safe and<br />
established bronchoscopic method in the histopathologic<br />
diagnosis <strong>of</strong> pulmonary infections in immunocompromised<br />
patients. As for diagnosing IPA, however, the<br />
diagnostic sensitivity <strong>of</strong> TBB has been reported to be quite<br />
low. 2 Some authors 14,15 advocate the use <strong>of</strong> invasive surgical<br />
procedures for the diagnosis and treatment <strong>of</strong> IPA. In<br />
our case 2, the clinical and radiologic progress was relatively<br />
less acute, and the immunosuppression was mild, so<br />
it might be categorized as subacute IPA. However, a<br />
Figure 4. Ultrathin bronchoscopic views <strong>of</strong> the lung cavity. A whitish intracavitary lesion caused by A<br />
flavus was seen in case 1 (left, A), and a yellowish white lesion caused by A fumigatus was seen in case<br />
2(right, B).<br />
www.chestjournal.org CHEST / 129 /2/FEBRUARY, 2006 477<br />
Downloaded From: http://journal.publications.chestnet.org/ on 07/13/2013
Figure 5. A cavity in the superior segment <strong>of</strong> the left lower lobe shows enlargement (right) compared<br />
to 2 months previously (left).<br />
precise distinction has not been made between IPA and<br />
other chronic forms <strong>of</strong> Aspergillus, such as subacute IPA<br />
or chronic necrotizing pulmonary aspergillosis. Moreover,<br />
the diagnostic yield <strong>of</strong> TBB for chronic cavitary pulmonary<br />
aspergillosis has also been reported to be quite low. 16<br />
A commercially available ultrathin bronchoscope with<br />
an external diameter <strong>of</strong> 2.8 mm has been used as a<br />
diagnostic 4–6 and therapeutic tool 17 in the peripheral<br />
airways. It can reach the peripheral area <strong>of</strong> the lung and<br />
occasionally even the visceral pleura. 18 Nevertheless, the<br />
closer the ultrathin bronchoscope is brought to the peripheral<br />
bronchi, the poorer the visibility is. S<strong>of</strong>t peripheral<br />
bronchi, which easily collapse under bronchoscopic<br />
suction, and bronchial secretions obstruct the visibility.<br />
However, a cavitary lesion in the peripheral area <strong>of</strong> the<br />
lung may be a good indication for ultrathin bronchoscopy.<br />
Once the tip <strong>of</strong> the ultrathin bronchoscope enters the<br />
cavity through a small bronchus, the range <strong>of</strong> vision<br />
becomes broad. Biopsy <strong>of</strong> a lesion with bronchoscopic<br />
visualization should result in higher diagnostic yield.<br />
Moreover, a biopsy with direct visualization using miniforceps<br />
may decrease the risks <strong>of</strong> bleeding.<br />
Visualization <strong>of</strong> intracavitary lesions during bronchoscopy<br />
in patients with pulmonary aspergillosis, mostly<br />
aspergilloma, which is characterized by mycelial growth in<br />
a preexisting lung cavity and usually has a good prognosis<br />
without any treatments, has been rarely reported. 19–21<br />
However, there may be only a limited number <strong>of</strong> cases in<br />
which the diameter <strong>of</strong> the bronchus leading to the cavity is<br />
relatively large. In our case 1, a conventional bronchoscope<br />
could reach no farther than the segmental bronchus.<br />
Our results indicate an ultrathin bronchoscope can enter a<br />
cavity more frequently than a conventional bronchoscope.<br />
In some cases, it appears that ultrathin bronchoscopy can<br />
be useful in the diagnosis <strong>of</strong> cavitating IPA. Further<br />
studies are needed to elucidate in more detail its utility for<br />
the diagnosis <strong>of</strong> IPA.<br />
References<br />
1 Reichenberger F, Habicht J, Matt P, et al. Diagnostic yield <strong>of</strong><br />
bronchoscopy in histologically proven invasive pulmonary<br />
aspergillosis. Bone Marrow Transplant 1999; 24:1195–1199<br />
2 Albelda SM, Talbot GH, Gerson SL, et al. Role <strong>of</strong> fiberoptic<br />
bronchoscopy in the diagnosis <strong>of</strong> invasive pulmonary aspergillosis<br />
in patients with acute leukemia. Am J Med 1984;<br />
76:1027–1034<br />
3 Stevens DA, Kan VL, Judson MA, et al. Practice guidelines<br />
for diseases caused by Aspergillus: Infectious Diseases Society<br />
<strong>of</strong> America. Clin Infect Dis 2000; 30:696–709<br />
4 Saka H, Oki M, Kumazawa A, et al. Diagnosis <strong>of</strong> pulmonary<br />
peripheral lesions using ultrathin bronchoscope. J Jpn Soc<br />
Bronchol 2000; 22:617–619<br />
5 Shinagawa N, Yamazaki K, Onodera Y, et al. CT-guided<br />
transbronchial biopsy using an ultrathin bronchoscope with<br />
virtual bronchoscopic navigation. <strong>Chest</strong> 2004; 125:1138–1143<br />
6 Yamamoto S, Ueno K, Imamura F, et al. Usefulness <strong>of</strong><br />
ultrathin bronchoscopy in diagnosis <strong>of</strong> lung cancer. Lung<br />
Cancer 2004; 56:43–48<br />
7 Gerson SL, Talbot GH, Hurwitz S, et al. Prolonged granulocytopenia:<br />
the major risk factor for invasive pulmonary aspergillosis<br />
in patients with acute leukemia. Ann Intern Med<br />
1984; 100:345–351<br />
8 Karam GH, Griffin FM. Invasive pulmonary aspergillosis in<br />
478 <strong>Selected</strong> <strong>Reports</strong><br />
Downloaded From: http://journal.publications.chestnet.org/ on 07/13/2013
nonimmunocompromised, nonneutropenic hosts. Rev Infect<br />
Dis 1986; 8:357–363<br />
9 Palmer LB, Greenberg HE, Schiff MJ. Corticosteroid treatment<br />
as a risk factor for invasive aspergillosis in patients with<br />
lung disease. Thorax 1991; 46:15–20<br />
10 Denning DW, Marinus A, Cohen J, et al. An EORTC<br />
multicentre prospective survey <strong>of</strong> invasive aspergillosis in<br />
haematological patients: diagnosis and therapeutic outcome.<br />
EORTC Invasive Fungal Infections Cooperative Group. J Infect<br />
1998; 37:173–180<br />
11 Reichenberger F, Habicht JM, Gratwohl A, et al. Diagnosis<br />
and treatment <strong>of</strong> invasive pulmonary aspergillosis in neutropenic<br />
patients. Eur Respir J 2002; 19:743–755<br />
12 Saito H, Anaissie EJ, Morice RC, et al. Bronchoalveolar<br />
lavage in the diagnosis <strong>of</strong> pulmonary infiltrates in patients<br />
with acute leukemia. <strong>Chest</strong> 1988; 94:745–749<br />
13 Tablan OC, Anderson LJ, Besser R, et al. Guidelines for<br />
preventing health-care-associated pneumonia, 2003: recommendations<br />
<strong>of</strong> CDC and the Healthcare Infection Control<br />
Practices Advisory Committee. MMWR Morb Mortal Wkly<br />
Rep Recomm Rep 2004; 53:1–36<br />
14 Baron O, Guillaume B, Moreau P, et al. Aggressive surgical<br />
management in localized pulmonary mycotic and nonmycotic<br />
infections for neutropenic patients with acute leukemia:<br />
report <strong>of</strong> eighteen cases. J Thorac Cardiovasc Surg 1998;<br />
115:63–69<br />
15 Reichenberger F, Habicht J, Kaim A, et al. Lung resection for<br />
invasive pulmonary aspergillosis in neutropenic patients with<br />
hematologic diseases. Am J Respir Crit Care Med 1998;<br />
158:885–890<br />
16 Denning DW, Riniotis K, Dobrashian R, et al. Chronic<br />
cavitary and fibrosing pulmonary and pleural aspergillosis:<br />
case series, proposed nomenclature change, and review. Clin<br />
Infect Dis 2003; 37:265S–280S<br />
17 Oki M, Saka H, Kumazawa A, et al. Extraction <strong>of</strong> peripheral<br />
endobronchial foreign body using an ultrathin flexible bronchoscope.<br />
J Bronchol 2004; 11:37–39<br />
18 Oki M, Saka H, Kitagawa C, et al. Visceral pleural perforation<br />
in two cases <strong>of</strong> ultrathin bronchoscopy. <strong>Chest</strong> 2005; 127:<br />
2271–2273<br />
19 Hargis JL, Bone RC, Stewart J, et al. Intracavitary amphotericin<br />
B in the treatment <strong>of</strong> symptomatic pulmonary aspergillomas.<br />
Am J Med 1980; 68:389–394<br />
20 Smith RL, Morelli MJ, Aranda CP. Pulmonary aspergilloma<br />
diagnosed by fiberoptic bronchoscopy. <strong>Chest</strong> 1987; 92:948–<br />
949<br />
21 Rohatgi PK, Chasse RT. Endoscopic visualization <strong>of</strong> aspergilloma.<br />
Respiration 1991; 58:112–114<br />
Closure <strong>of</strong> a Bronchopleural<br />
Fistula Using Bronchoscopic<br />
Placement <strong>of</strong> an Endobronchial<br />
Valve Designed for the<br />
Treatment <strong>of</strong> Emphysema*<br />
J. Scott Ferguson, MD, FCCP; Kimberly Sprenger, BSN; and<br />
Timothy Van Natta, MD<br />
Pneumothoraces are sometimes complicated by a<br />
persistent air leak or bronchopleural fistula requiring<br />
prolonged chest tube drainage. Non-surgical<br />
treatment <strong>of</strong> persistent bronchopleural fistulas is<br />
<strong>of</strong>ten performed in patients who are poor surgical<br />
candidates, but the ideal method <strong>of</strong> closure is not<br />
known. Here we report closure <strong>of</strong> a persistent distal<br />
bronchopleural fistula using a one-way endobronchial<br />
valve designed for the treatment <strong>of</strong> emphysema.<br />
(CHEST 2006; 129:479–481)<br />
Key words: bronchial fistula; bronchoscopy; pneumothorax<br />
Abbreviations: BPF bronchopleural fistula; RML right<br />
middle lobe<br />
Pneumothoraces are sometimes complicated by a persistent<br />
air leak or bronchopleural fistula (BPF) requiring<br />
prolonged chest tube drainage. It is generally recommended<br />
that surgical treatment be undertaken when possible. However,<br />
many patients are very poor surgical candidates. In this<br />
article, we report the successful closure <strong>of</strong> a prolonged air<br />
leak with a one-way endobronchial valve originally designed<br />
for the palliative treatment <strong>of</strong> emphysema.<br />
Case Report<br />
A 63-year-old man with widely metastatic breast cancer was<br />
admitted for sudden onset <strong>of</strong> severe dyspnea 8 days after<br />
thoracoscopic placement <strong>of</strong> an indwelling cuffed pleural catheter.<br />
A large hydropneumothorax was discovered, and the indwelling<br />
catheter was connected to suction, relieving the dyspnea. A chest<br />
tube was then placed and connected to a water-seal device. A<br />
large air leak was noted with each exhalation and continued for<br />
the next 9 days. A new chest tube was placed, but the air leak<br />
continued.<br />
The etiology <strong>of</strong> the pneumothorax was not certain but was<br />
assumed to be a delayed complication <strong>of</strong> the thoracoscopy, as<br />
opposed to necrosis and breakdown <strong>of</strong> a metastatic focus <strong>of</strong> tumor.<br />
A thin-section, multidetector CT scan <strong>of</strong> the chest revealed multiple<br />
metastases, hydropneumothorax, and airspace disease <strong>of</strong> the lower<br />
lobes. No radiographic evidence <strong>of</strong> BPF was present.<br />
Surgical correction with plication <strong>of</strong> the <strong>of</strong>fending lobe was<br />
considered but judged to be risky given that the patient’s pulmonary<br />
reserve was limited due to the metastatic disease. Endoscopic<br />
therapies with glue, gel foam, and coiling were considered but were<br />
judged to have a low likelihood <strong>of</strong> success based on the endoscopist’s<br />
previous experience with these methods in other patients. Therefore,<br />
these techniques <strong>of</strong> BPF closure were not attempted in this<br />
patient.<br />
One-way endobronchial valves for the treatment <strong>of</strong> emphysema<br />
became available in the United States last year as part <strong>of</strong> a national<br />
clinical trial. 1 We thought placement <strong>of</strong> an endobronchial valve that<br />
would allow escape <strong>of</strong> air from the <strong>of</strong>fending segment or lobe, but<br />
*From Carver <strong>College</strong> <strong>of</strong> Medicine, Departments <strong>of</strong> Internal<br />
Medicine (Dr. Ferguson and Ms. Sprenger) and Thoracic and<br />
Cardiovascular Surgery (Dr. Van Natta), University <strong>of</strong> Iowa, Iowa<br />
City, IA.<br />
Dr. Ferguson, Ms. Sprenger, and Dr. Van Natta were supported in<br />
part by the Endobronchial Valve for Emphysema Palliation Trial<br />
(VENT) Trial.<br />
Manuscript received August 31, 2005; revision accepted October 1,<br />
2005.<br />
Reproduction <strong>of</strong> this article is prohibited without written permission<br />
from the <strong>American</strong> <strong>College</strong> <strong>of</strong> <strong>Chest</strong> <strong>Physicians</strong> (www.chestjournal.<br />
org/misc/reprints.shtml).<br />
Correspondence to: J. Scott Ferguson, MD, Department <strong>of</strong> Internal<br />
Medicine, C-33 GH 200, Hawkins Dr, Iowa City, IA 52242;<br />
e-mail: john-s-ferguson@uiowa.edu<br />
www.chestjournal.org CHEST / 129 /2/FEBRUARY, 2006 479<br />
Downloaded From: http://journal.publications.chestnet.org/ on 07/13/2013