a thermodynamic approach to cement hydration - Eawag-Empa ...
a thermodynamic approach to cement hydration - Eawag-Empa ...
a thermodynamic approach to cement hydration - Eawag-Empa ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
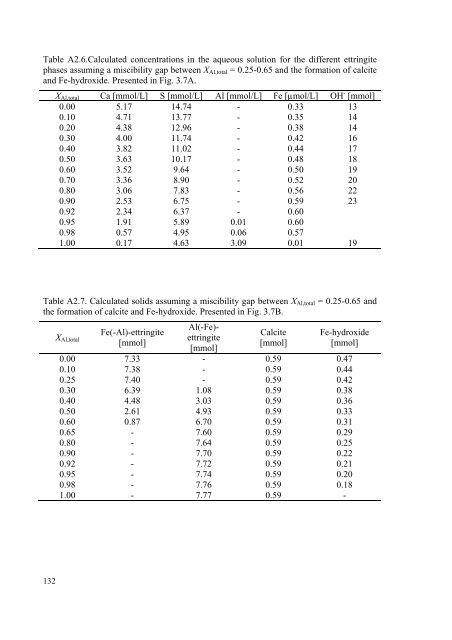
Table A2.6.Calculated concentrations in the aqueous solution for the different ettringite<br />
phases assuming a miscibility gap between XAl,<strong>to</strong>tal = 0.25-0.65 and the formation of calcite<br />
and Fe-hydroxide. Presented in Fig. 3.7A.<br />
132<br />
XAl,<strong>to</strong>tal Ca [mmol/L] S [mmol/L] Al [mmol/L] Fe [µmol/L] OH - [mmol]<br />
0.00 5.17 14.74 - 0.33 13<br />
0.10 4.71 13.77 - 0.35 14<br />
0.20 4.38 12.96 - 0.38 14<br />
0.30 4.00 11.74 - 0.42 16<br />
0.40 3.82 11.02 - 0.44 17<br />
0.50 3.63 10.17 - 0.48 18<br />
0.60 3.52 9.64 - 0.50 19<br />
0.70 3.36 8.90 - 0.52 20<br />
0.80 3.06 7.83 - 0.56 22<br />
0.90 2.53 6.75 - 0.59 23<br />
0.92 2.34 6.37 - 0.60<br />
0.95 1.91 5.89 0.01 0.60<br />
0.98 0.57 4.95 0.06 0.57<br />
1.00 0.17 4.63 3.09 0.01 19<br />
Table A2.7. Calculated solids assuming a miscibility gap between XAl,<strong>to</strong>tal = 0.25-0.65 and<br />
the formation of calcite and Fe-hydroxide. Presented in Fig. 3.7B.<br />
XAl,<strong>to</strong>tal<br />
Fe(-Al)-ettringite<br />
[mmol]<br />
Al(-Fe)ettringite<br />
[mmol]<br />
Calcite<br />
[mmol]<br />
Fe-hydroxide<br />
[mmol]<br />
0.00 7.33 - 0.59 0.47<br />
0.10 7.38 - 0.59 0.44<br />
0.25 7.40 - 0.59 0.42<br />
0.30 6.39 1.08 0.59 0.38<br />
0.40 4.48 3.03 0.59 0.36<br />
0.50 2.61 4.93 0.59 0.33<br />
0.60 0.87 6.70 0.59 0.31<br />
0.65 - 7.60 0.59 0.29<br />
0.80 - 7.64 0.59 0.25<br />
0.90 - 7.70 0.59 0.22<br />
0.92 - 7.72 0.59 0.21<br />
0.95 - 7.74 0.59 0.20<br />
0.98 - 7.76 0.59 0.18<br />
1.00 - 7.77 0.59 -