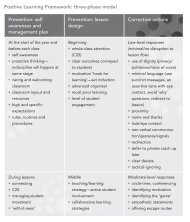
2 Chapter 6 ⢠organising elements Organising elements
2 Chapter 6 ⢠organising elements Organising elements
2 Chapter 6 ⢠organising elements Organising elements
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
6<br />
Properties and structure<br />
Elements in our oceans<br />
Fig 6.39 There are more<br />
<strong>elements</strong> in our oceans<br />
than you may think.<br />
32 <strong>Chapter</strong> 6 • <strong>organising</strong> <strong>elements</strong><br />
We all know that water<br />
has the formula H 2 O and<br />
therefore contains oxygen and<br />
hydrogen. Sea water contains<br />
salt, so it also has the <strong>elements</strong><br />
sodium and chlorine. But did<br />
you know that our oceans<br />
contain a whole range of other<br />
<strong>elements</strong>, such as magnesium,<br />
sulfur, calcium, potassium,<br />
bromine and even gold? So<br />
how did the <strong>elements</strong> get into<br />
the water that covers twothirds<br />
of the surface of our<br />
planet? And in what form are<br />
these <strong>elements</strong> present within<br />
the sea water?<br />
Many of the metals present in<br />
sea water have been leached<br />
from minerals on the floor<br />
of the ocean. Sodium is an<br />
example of this; it dissolves<br />
into the water in the form<br />
of sodium ions (Na + ). Over<br />
time, gases from volcanic<br />
activity on Earth have passed<br />
through the oceans. Some of<br />
these gases, such as hydrogen<br />
chloride, contain chlorine and<br />
this element also becomes<br />
dissolved in sea water in the<br />
form of ions. Sulfur is also a<br />
product of volcanic activity<br />
and most sulfur in today’s<br />
oceans exists in the form of<br />
sulfate ions.<br />
UNCORRECTED PAGE PROOFS<br />
CAS_SB10_TXT_06_1pp.indd 32<br />
11/11/11 4:58 PM