2 Chapter 6 ⢠organising elements Organising elements
2 Chapter 6 ⢠organising elements Organising elements
2 Chapter 6 ⢠organising elements Organising elements
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
10<br />
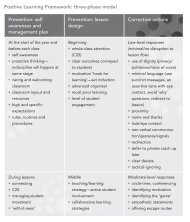
Properties and structure<br />
6.1<br />
Fig 6.9<br />
1<br />
9<br />
H<br />
Fe<br />
17<br />
Cl<br />
Why do we organise atoms?<br />
Remember and understand<br />
1 What is the atomic number of the element<br />
known as ununpentium?<br />
2 What is the overall order of <strong>elements</strong> in the<br />
periodic table based on?<br />
3 Arrange the following people in chronological<br />
(time) order and matching them with the<br />
concepts listed.<br />
<br />
<strong>Chapter</strong> 6 • <strong>organising</strong> <strong>elements</strong><br />
• People: Bohr, Lavoisier, Seaborg,<br />
Mendeleev, Newlands, Berzelius, Dobereiner<br />
• Concepts: periodic table developed, modern<br />
concept of an element, atomic symbols and<br />
weights standardised<br />
4 What is the difference between an atom<br />
and an element?<br />
2 3<br />
He Li<br />
10 11<br />
Ne Na<br />
18 19<br />
Ar K<br />
Apply<br />
10 Some <strong>elements</strong> are very new, whereas<br />
others have been known for hundreds of<br />
years. What is the relationship between<br />
the date of discovery and the features of<br />
the element? Use the table below as a<br />
guide to answer this question.<br />
5 The element mendelevium (101)<br />
is named after the scientist who<br />
developed the first version of<br />
the periodic table. Mendeleev<br />
combined the ideas of two<br />
earlier scientists: who were these<br />
scientists and what did they do?<br />
Analyse and evaluate<br />
Properties and structure<br />
6 Scientists like Berzelius and Mendeleev<br />
worked on their own to produce new ideas.<br />
Others, like Seaborg, worked in a team. Now<br />
most scientists work in teams. What are the<br />
advantages of working in a team?<br />
7 Scientists have had to deduce what it is like<br />
inside the atom from indirect evidence, similar<br />
to the work of astronomers in determining the<br />
temperature and composition of stars. List<br />
three advantages and three disadvantages of<br />
using indirect evidence to develop scientific<br />
theories.<br />
Ethical behaviour<br />
8 Meyer and Mendeleev both published a<br />
periodic table within months of each other.<br />
However, Mendeleev is given sole credit for the<br />
developing the periodic table.<br />
a Is it fair that the person who first discovers/<br />
develops/publishes something gets the<br />
credit for this discovery?<br />
b What did Mendeleev do for him to get sole<br />
credit for developing the periodic table?<br />
Critical and creative thinking<br />
9 Make a spiral periodic table of the first twenty<br />
<strong>elements</strong>. Use Figure 6.9 as a guide. Glue a<br />
paper strip onto a piece of wood, such as an<br />
ice block stick.<br />
Element Date of discovery Notes<br />
Gold, tin Ancient times Found on the surface of the Earth<br />
Iron, copper 1500 bce Separated in the heat of an open fire<br />
Aluminium, sodium 1800s Separated using electricity<br />
Argon, neon Early 1900s Separated from liquid air<br />
Neptunium, plutonium 1940s Synthesised in nuclear reactors or cyclotrons<br />
UNCORRECTED PAGE PROOFS<br />
CAS_SB10_TXT_06_1pp.indd 10<br />
11/11/11 4:58 PM