Nanomedicine - European Science Foundation
Nanomedicine - European Science Foundation
Nanomedicine - European Science Foundation
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
APPENDIX V 45<br />
Nanoarchitecture<br />
Objectives: Nanostructuring modification of biomaterials<br />
for tissue engineering and other biomimetic applications.<br />
Contact: Vasif Hasirci<br />
Email: chasirci@metu.edu.tr<br />
NanoBone<br />
Objectives: To apply nanotechnology, in terms of<br />
structuring, to bone repair and regeneration.<br />
Contact: Fernando Monteiro<br />
Email: fjmont@ineb.up.pt<br />
Nanostres<br />
Objectives: To use nanotechnology techniques to create<br />
implants and implant technologies for skeletal tissues.<br />
Contact: Josep Anton Planell<br />
Email: plannell@cmem.upc.es<br />
NB-TISS-INTER-MED<br />
Objectives: To redress the issue of the decreasing<br />
<strong>European</strong> market share in medical devices.<br />
Contact: Ian McKay<br />
Email: ian.mckay@pera.com<br />
NMMA<br />
Objectives: To carry out interdisciplinary research (for<br />
medical applications) into the possibilities<br />
nanotechnology offers for shaping surface and internal<br />
structure, and using molecular biology to control<br />
interactions between materials and cells.<br />
Contact: Krzysztof Kurzydlowski<br />
Email: KJK@inmat.pw.edu.pl<br />
NONMETALLICIMPLANTS<br />
Objectives: To create new/improved polymer materials<br />
for implants and scaffolds, with the necessary structure to<br />
optimise their function.<br />
Contact: Jan Chlopek<br />
Email: chlopek@uci.agh.edu.pl<br />
SAM-MED-NET<br />
Objectives: To gain a better understanding of self<br />
assembly mechanisms for applications in biomimetic<br />
materials (e.g. tissue engineering).<br />
Contact: Frederic Cuisinier<br />
Email: fred.cuisinier@odonto-ulp.u-strabg.fr<br />
Appendix V<br />
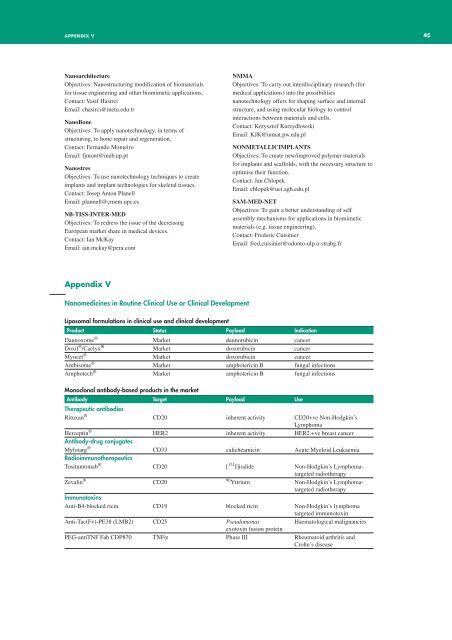
<strong>Nanomedicine</strong>s in Routine Clinical Use or Clinical Development<br />
Liposomal formulations in clinical use and clinical development<br />
Product Status Payload Indication<br />
Daunoxome ® Market daunorubicin cancer<br />
Doxil ® /Caelyx ® Market doxorubicin cancer<br />
Myocet ® Market doxorubicin cancer<br />
Ambisome ® Market amphotericin B fungal infections<br />
Amphotech ® Market amphotericin B fungal infections<br />
Monoclonal antibody-based products in the market<br />
Antibody Target Payload Use<br />
Therapeutic antibodies<br />
Rituxan ® CD20 inherent activity CD20+ve Non-Hodgkin’s<br />
Lymphoma<br />
Herceptin ® HER2 inherent activity HER2 +ve breast cancer<br />
Antibody-drug conjugates<br />
Mylotarg ® CD33 calicheamicin Acute Myeloid Leukaemia<br />
Radioimmunotherapeutics<br />
Tositumomab ® CD20 [ 131 I]iodide Non-Hodgkin’s Lymphomatargeted<br />
radiotherapy<br />
Zevalin ®<br />
CD20<br />
90 Yttrium Non-Hodgkin’s Lymphomatargeted<br />
radiotherapy<br />
Immunotoxins<br />
Anti-B4-blocked ricin CD19 blocked ricin Non-Hodgkin’s lymphoma<br />
targeted immunotoxin<br />
Anti-Tac(Fv)-PE38 (LMB2) CD25 Pseudomonas Haematological malignancies<br />
exotoxin fusion protein<br />
PEG-antiTNF Fab CDP870 TNFα Phase III Rheumatoid arthritis and<br />
Crohn’s disease