Reaction of Furan and Thiophene
Reaction of Furan and Thiophene
Reaction of Furan and Thiophene
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
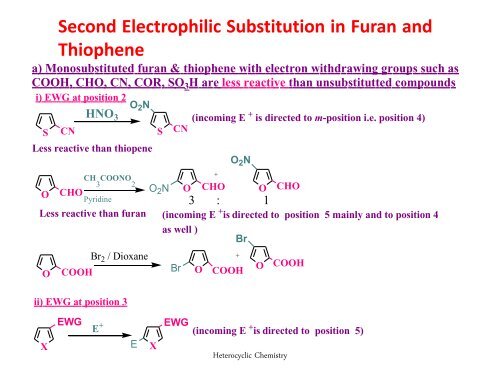
Second Electrophilic Substitution in <strong>Furan</strong> <strong>and</strong><br />
<strong>Thiophene</strong><br />
a) Monosubstituted furan & thiophene with electron withdrawing groups such as<br />
COOH, CHO, CN, COR, SO 3 H are less reactive than unsubstitutted compounds<br />
i) EWG at position 2<br />
O 2 N<br />
HNO 3 (incoming E + is directed to m-position i.e. position 4)<br />
S CN S CN<br />
Less reactive than thiopene<br />
O 2 N<br />
+<br />
CH COONO 3 2<br />
O CHO<br />
O 2 N O CHO O CHO<br />
Pyridine<br />
Less reactive than furan<br />
O<br />
COOH<br />
Br 2 / Dioxane<br />
ii) EWG at position 3<br />
3 : 1<br />
(incoming E + is directed to position 5 mainly <strong>and</strong> to position 4<br />
as well )<br />
Br<br />
+<br />
Br O COOH<br />
O COOH<br />
X<br />
EWG<br />
E +<br />
E<br />
X<br />
EWG<br />
(incoming E + is directed to position 5)<br />
Heterocyclic Chemistry