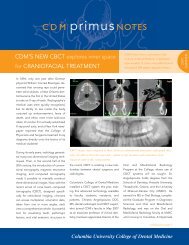
Jarvie Journal - College of Dental Medicine - Columbia University
Jarvie Journal - College of Dental Medicine - Columbia University
Jarvie Journal - College of Dental Medicine - Columbia University
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
Multiphase Bioscaffold for Integrated Regeneration <strong>of</strong> Root-Periodontium Complex<br />
Jeffrey Hajibandeh 1 , Chang H. Lee 1,2 , Jeffrey Ahn 1,2 , Andrew Fan 1,2 , Jeremy J. Mao 1,2 *<br />
1 <strong>College</strong> <strong>of</strong> <strong>Dental</strong> <strong>Medicine</strong>, <strong>Columbia</strong> <strong>University</strong>, New York, NY,<br />
2 Center for Crani<strong>of</strong>acial Regeneration, <strong>College</strong> <strong>of</strong> <strong>Dental</strong> <strong>Medicine</strong><br />
<strong>Columbia</strong> <strong>University</strong>, New York, NY; *Faculty Mentor<br />
Objectives: Regeneration <strong>of</strong> root and periodontium holds a promise to overcome current limitations <strong>of</strong> dental implant<br />
therapy, including its dependence upon supporting bone structure and lack <strong>of</strong> biological integration and remodeling with<br />
host alveolar bone. Here, we develop an integrated scaffold with multiphase microstructure and spatial-delivered<br />
bioactive cues, and its potential in generating root-periodontium complex from dental stem/progenitor cells is tested both<br />
in vitro and in vivo.<br />
Materials and Methods: Polycaprolactone (PCL)-hydroxylapatite (HA) (90:10wt%) scaffolds were fabricated<br />
(5×5×3mm 3 ) using 3D printing per our prior works. The scaffolds consisted <strong>of</strong> three phases: A) 100µm microchannels<br />
with 2.25mm in width, B) 600µm microchannels with 500µm in width, and C) 300µm microchannels with 2.25mm in<br />
width. Phases A, B, and C were designed to guide formation <strong>of</strong> dentin/cementum, periodontal ligament (PDL), and<br />
alveolar bone, respectively. To promote cell differentiation, PLGA microspheres encapsulated with Amelogenin, CTGF,<br />
and BMP2 were incorporated in phase A, B, and C <strong>of</strong> the scaffolds, respectively. To test various cell types and their<br />
responses, scaffolds were seeded with human dental pulp stem/progenitor cells (DPSCs), periodontal ligament<br />
stem/progenitor cells (PDLSCs), or alveolar bone stem/progenitor cells (ABSCs) with approximately 100,000 cells per<br />
scaffold. In vitro scaffolds were grown in modified DMEM media, while in vivo scaffolds were implanted<br />
subcutaneously into nude mice. Both models were harvest at four weeks and multiphase tissue formation in the scaffolds<br />
was evaluated by multitude <strong>of</strong> assays.<br />
Results and Conclusions: Immuno-/histomorphometric analysis demonstrated that multiphase scaffold microstructure<br />
with spatial-delivered bioactive cues successfully generated multiphase putative tissues consisting <strong>of</strong> collagen I-rich<br />
fibrous matrix (Phase B) sandwiched between mineralized regions in both in vitro and in vivo (Phase A and C). DSPpositive<br />
mineralized structure in Phase C was highly dense and polarized (reminiscent <strong>of</strong> native dentin) in comparison<br />
with that <strong>of</strong> Phase C. DPSC’s were superior to the other cell types in mineralization, whereas PDLSC’s yielded highly<br />
aligned fibrous structure as compared to the other cell types. In vivo results further demonstrated highly aligned fibrous<br />
tissues inserting into CEMP + mineralized matrix. PCR demonstrated amplified levels <strong>of</strong> tissue specific markers in the<br />
GF+ scaffolds: Phase A (putative alveolar bone) expressed relatively high levels <strong>of</strong> BSP (p