Form 3 Science Test Test 4: Acids and basis Total: 50 ... - Pearson
Form 3 Science Test Test 4: Acids and basis Total: 50 ... - Pearson
Form 3 Science Test Test 4: Acids and basis Total: 50 ... - Pearson
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
<strong>Form</strong> 3 <strong>Science</strong> <strong>Test</strong><br />
<strong>Test</strong> 4: <strong>Acids</strong> <strong>and</strong> <strong>basis</strong><br />
<strong>Total</strong>: <strong>50</strong> marks<br />
Name: ______________________________________ Date: ______________<br />
Section A<br />
Write down the correct letter for each of the following questions. You must choose one<br />
answer only.<br />
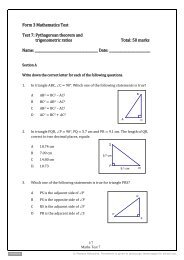
1. Which of the following statements is true<br />
A<br />
B<br />
C<br />
D<br />
We use the pH scale to indicate how strong an acid or alkali is.<br />
Substances with a pH from 1 to 7 are acids.<br />
Substances with a pH from 7 to 14 are alkalis.<br />
All of the above.<br />
2. What do we call a substance that forms an alkaline solution when dissolved<br />
in water<br />
A<br />
B<br />
C<br />
D<br />
An acid<br />
An alkaline<br />
An atom<br />
A base<br />
3. What is used to tell the difference between an acid <strong>and</strong> a base<br />
A<br />
B<br />
C<br />
D<br />
An indicator<br />
An abductor<br />
An incident<br />
An adjudicator<br />
1/6<br />
<strong>Science</strong> <strong>Test</strong> 4<br />
© <strong>Pearson</strong> Botswana. Permission is given to photocopy these pages for school use.
4. What do we call the reaction that takes place when an acid reacts with<br />
an alkali<br />
A<br />
B<br />
C<br />
D<br />
Neutralisation<br />
Naturalisation<br />
Alkalination<br />
Acidity<br />
5. Which equation shows the reaction between zinc <strong>and</strong> hydrochloric acid<br />
A<br />
B<br />
C<br />
D<br />
hydrochloric acid + zinc ➝ zinc chloride +hydrogen<br />
hydrochloric acid – zinc ➝ zinc chloride + hydrogen<br />
hydrochloric acid + zinc ➝ zinc chloride + oxygen<br />
None of the above<br />
6. Which of the following statements is true of chemical reactions<br />
A<br />
B<br />
C<br />
D<br />
The more concentrated the acid, the faster the chemical reaction<br />
The less concentrated the acid, the faster the chemical reaction<br />
The more concentrated the acid, the slower the chemical reaction<br />
None of the above<br />
7. The chemical reaction that happens when we make soap is:<br />
A<br />
B<br />
C<br />
D<br />
fat + alkali ➝ soap + glycerol<br />
fat + alkali ➝ soap – glycerol<br />
fat + alkali ➝ soap + glycerine<br />
fat + acid ➝ soap + glycerol<br />
2/6<br />
<strong>Science</strong> <strong>Test</strong> 4<br />
© <strong>Pearson</strong> Botswana. Permission is given to photocopy these pages for school use.
8. Which of the following statements is true<br />
A<br />
B<br />
C<br />
D<br />
Water that contains dissolved calcium or magnesium salts is hard.<br />
Hard water is water that forms a scum when we try to make soap lather.<br />
The scum is formed by a chemical reaction between the calcium salt in<br />
the water <strong>and</strong> the soap.<br />
All of the above.<br />
9. Which metal is contained in a torch battery<br />
A<br />
B<br />
C<br />
D<br />
Copper<br />
Carbon<br />
Zinc<br />
Silver<br />
10. Calcium chloride is a:<br />
A<br />
B<br />
C<br />
D<br />
Salt<br />
Metal<br />
Solute<br />
Substrate<br />
[10]<br />
3/6<br />
<strong>Science</strong> <strong>Test</strong> 4<br />
© <strong>Pearson</strong> Botswana. Permission is given to photocopy these pages for school use.
Section B<br />
11. Name three examples of substances that are acids. (3)<br />
_________________________________________________________________<br />
_________________________________________________________________<br />
_________________________________________________________________<br />
12. Name three examples of substances that are alkalines. (3)<br />
_________________________________________________________________<br />
_________________________________________________________________<br />
_________________________________________________________________<br />
13. What do we call a solution with a pH of 7 on the pH scale (1)<br />
_________________________________________________________________<br />
14. Complete the following:<br />
a) A solution that turns blue litmus paper red is a _____________________. (1)<br />
b) A solution that turns red litmus paper blue is a _____________________. (1)<br />
15. Explain the difference between the following in respect of acids:<br />
a) Strong (2)<br />
____________________________________________________________<br />
____________________________________________________________<br />
4/6<br />
<strong>Science</strong> <strong>Test</strong> 4<br />
© <strong>Pearson</strong> Botswana. Permission is given to photocopy these pages for school use.
) Weak (2)<br />
____________________________________________________________<br />
____________________________________________________________<br />
c) Concentrated (2)<br />
____________________________________________________________<br />
____________________________________________________________<br />
d) Dilute (2)<br />
____________________________________________________________<br />
____________________________________________________________<br />
16. Write down the formulae for the following:<br />
a) Sulphuric acid (1)<br />
____________________________________________________________<br />
b) Nitric acid (1)<br />
____________________________________________________________<br />
c) Calcium hydroxide (1)<br />
____________________________________________________________<br />
[20]<br />
5/6<br />
<strong>Science</strong> <strong>Test</strong> 4<br />
© <strong>Pearson</strong> Botswana. Permission is given to photocopy these pages for school use.
Section C<br />
17. Using your knowledge of <strong>Science</strong>, explain what happens when a person suffers<br />
from heartburn. (10)<br />
_________________________________________________________________<br />
_________________________________________________________________<br />
_________________________________________________________________<br />
_________________________________________________________________<br />
_________________________________________________________________<br />
_________________________________________________________________<br />
_________________________________________________________________<br />
_________________________________________________________________<br />
_________________________________________________________________<br />
_________________________________________________________________<br />
18. Explain the science experiment you would conduct to show what happens when<br />
an alkali is added to an acid. (10)<br />
_________________________________________________________________<br />
_________________________________________________________________<br />
_________________________________________________________________<br />
_________________________________________________________________<br />
_________________________________________________________________<br />
_________________________________________________________________<br />
_________________________________________________________________<br />
_________________________________________________________________<br />
_________________________________________________________________<br />
_________________________________________________________________<br />
6/6<br />
<strong>Science</strong> <strong>Test</strong> 4<br />
[20]<br />
[<strong>Total</strong>: <strong>50</strong>]<br />
© <strong>Pearson</strong> Botswana. Permission is given to photocopy these pages for school use.