Polyatomic molecules - Cobalt
Polyatomic molecules - Cobalt
Polyatomic molecules - Cobalt
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
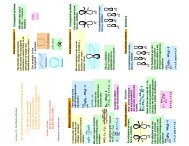
We have for conformation (a) the energies<br />
H 3 + : 2 α +2 2β<br />
H<br />
H<br />
3<br />
3 −<br />
: 3 α +2 2β<br />
: 4 α +2 2β<br />
For the second conformation (b)<br />
α-E<br />
β β<br />
β α-E<br />
β<br />
β β α-E<br />
=<br />
ε2 = ε3<br />
= α−β<br />
α-E<br />
β<br />
-E<br />
( α -E)<br />
− β β β + β β α<br />
β α-E β α-E<br />
β β<br />
3 + 3<br />
ε1 = α+<br />
2β<br />
3 − : 4 α +2β<br />
3 2 2 3<br />
3 2 3<br />
1 = + 2 ; 2 = 3 = −<br />
3 + α β<br />
3 : 3 α +3β<br />
3 -<br />
( α-E) −( α-E) β − 2β ( α-E)-2β<br />
=<br />
( α-E)<br />
− 3(<br />
α-E)<br />
β + 2β<br />
[( α-E)-β][ ( α-E)-β][ ( α-E)+2β]=<br />
0<br />
Thus<br />
ε α β ε ε α β<br />
We have for conformation (b) the energies<br />
H : 2 +4<br />
H<br />
H<br />
Thus H and H will prefer (b) but H<br />
will prefer (a)