Isolated ileal interposition in enteroendocrine L cells differentiation
Isolated ileal interposition in enteroendocrine L cells differentiation
Isolated ileal interposition in enteroendocrine L cells differentiation
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
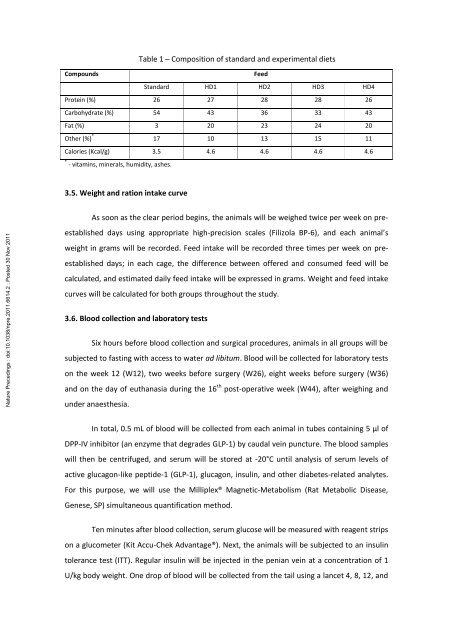
Table 1 – Composition of standard and experimental diets<br />
Compounds<br />
Feed<br />
Standard HD1 HD2 HD3 HD4<br />
Prote<strong>in</strong> (%) 26 27 28 28 26<br />
Carbohydrate (%) 54 43 36 33 43<br />
Fat (%) 3 20 23 24 20<br />
Other (%) * 17 10 13 15 11<br />
Calories (Kcal/g) 3.5 4.6 4.6 4.6 4.6<br />
* - vitam<strong>in</strong>s, m<strong>in</strong>erals, humidity, ashes.<br />
3.5. Weight and ration <strong>in</strong>take curve<br />
Nature Preced<strong>in</strong>gs : doi:10.1038/npre.2011.6614.2 : Posted 30 Nov 2011<br />
As soon as the clear period beg<strong>in</strong>s, the animals will be weighed twice per week on preestablished<br />
days us<strong>in</strong>g appropriate high-precision scales (Filizola BP-6), and each animal’s<br />
weight <strong>in</strong> grams will be recorded. Feed <strong>in</strong>take will be recorded three times per week on preestablished<br />
days; <strong>in</strong> each cage, the difference between offered and consumed feed will be<br />
calculated, and estimated daily feed <strong>in</strong>take will be expressed <strong>in</strong> grams. Weight and feed <strong>in</strong>take<br />
curves will be calculated for both groups throughout the study.<br />
3.6. Blood collection and laboratory tests<br />
Six hours before blood collection and surgical procedures, animals <strong>in</strong> all groups will be<br />
subjected to fast<strong>in</strong>g with access to water ad libitum. Blood will be collected for laboratory tests<br />
on the week 12 (W12), two weeks before surgery (W26), eight weeks before surgery (W36)<br />
and on the day of euthanasia dur<strong>in</strong>g the 16 th post-operative week (W44), after weigh<strong>in</strong>g and<br />
under anaesthesia.<br />
In total, 0.5 mL of blood will be collected from each animal <strong>in</strong> tubes conta<strong>in</strong><strong>in</strong>g 5 μl of<br />
DPP-IV <strong>in</strong>hibitor (an enzyme that degrades GLP-1) by caudal ve<strong>in</strong> puncture. The blood samples<br />
will then be centrifuged, and serum will be stored at -20°C until analysis of serum levels of<br />
active glucagon-like peptide-1 (GLP-1), glucagon, <strong>in</strong>sul<strong>in</strong>, and other diabetes-related analytes.<br />
For this purpose, we will use the Milliplex® Magnetic-Metabolism (Rat Metabolic Disease,<br />
Genese, SP) simultaneous quantification method.<br />
Ten m<strong>in</strong>utes after blood collection, serum glucose will be measured with reagent strips<br />
on a glucometer (Kit Accu-Chek Advantage®). Next, the animals will be subjected to an <strong>in</strong>sul<strong>in</strong><br />
tolerance test (ITT). Regular <strong>in</strong>sul<strong>in</strong> will be <strong>in</strong>jected <strong>in</strong> the penian ve<strong>in</strong> at a concentration of 1<br />
U/kg body weight. One drop of blood will be collected from the tail us<strong>in</strong>g a lancet 4, 8, 12, and