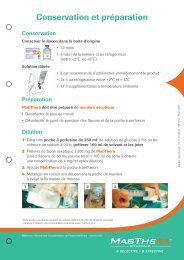
Intravenous TAMIFLU
Intravenous TAMIFLU
Intravenous TAMIFLU
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
COMPASSIONATE USE – CU 056<br />
<strong>Intravenous</strong> <strong>TAMIFLU</strong> ® (oseltamivir)<br />
4. DRUG ELIGIBILITY CRITERIA FOR THIS CU PROGRAM<br />
This product is actually under evaluation in clinical trials. On January 21 2010, the<br />
CHMP granted positive opinion for a Compassionate use program of Tamiflu intravenous<br />
formulation within the EU.<br />
5. SCOPE AND PATIENTS ELIGIBILITY CRITERIA FOR THIS CU PROGRAM<br />
IV oseltamivir should be considered only to treat critically ill adults and children older<br />
than 1 year of age with acute severe illness due to suspected or confirmed pandemic<br />
(H1N1) infection or infection due to seasonal influenza A or B virus.<br />
Specifically, oseltamivir infusion should only be used for the following patients who are<br />
admitted to a hospital and under the care or consultation of a licensed clinician (skilled in<br />
the diagnosis and management of patients with potentially life-threatening illness and the<br />
ability to recognise and manage medication-related adverse events):<br />
<br />
Adult or paediatric patients > 1 year of age with severe influenza illness for<br />
whom therapy with an IV agent is clinically indicated, based upon one or more of<br />
the following reasons:<br />
(1) patient not responding to either oral or inhaled authorised antiviral<br />
medication, or<br />
(2) drug delivery by a route other than IV (e.g. enteral oseltamivir or inhaled<br />
zanamivir) is not expected to be dependable or is not feasible, or<br />
The patient or legally authorized consent giver should provide written consent; for<br />
unconscious patients unable to provide consent for treatment with oseltamivir IV a<br />
surrogate decision maker must sign for the patient.<br />
Pregnant patients may be considered for treatment with oseltamivir IV only if the<br />
potential benefit justifies the potential risk to the fetus.<br />
For infants below 1 year of age, no dose recommendations can be provided, given the<br />
lack of data. Should a physician decide to treat an infant < 1 year of age, based upon a<br />
benefit /risk assessment, the doses which will be investigated in clinical study NP25138<br />
are provided in Section 9 for information.<br />
Oseltamivir IV should not be used in patients on probenecid as probenecid inhibits OAT<br />
function.<br />
Oseltamivir IV should not be administered to patients with clinical evidence of severe<br />
hepatic decompensation or with a history of allergy to the medication.<br />
In case of renal impairment, special dosing instructions must be followed (see further).<br />
Compassionate Use 056 – <strong>Intravenous</strong> Tamiflu ® (oseltamivir) - Version 20 December<br />
2011<br />
2/15