Drugs in the Pipeline Analysis
Drugs in the Pipeline Analysis
Drugs in the Pipeline Analysis
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
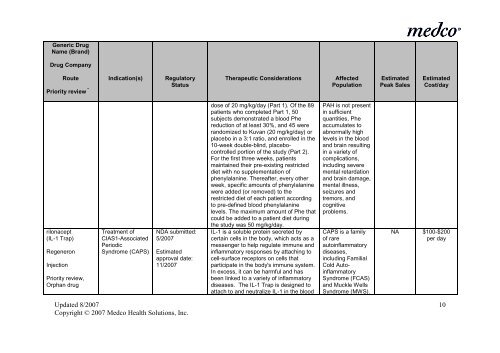
Generic Drug<br />
Name (Brand)<br />
Drug Company<br />
Route Indication(s) Regulatory<br />
Priority review * Status<br />
Therapeutic Considerations<br />
Affected<br />
Population<br />
Estimated<br />
Peak Sales<br />
Estimated<br />
Cost/day<br />
rilonacept<br />
(IL-1 Trap)<br />
Regeneron<br />
Injection<br />
Priority review,<br />
Orphan drug<br />
Treatment of<br />
CIAS1-Associated<br />
Periodic<br />
Syndrome (CAPS)<br />
NDA submitted:<br />
5/2007<br />
Estimated<br />
approval date:<br />
11/2007<br />
dose of 20 mg/kg/day (Part 1). Of <strong>the</strong> 89<br />
patients who completed Part 1, 50<br />
subjects demonstrated a blood Phe<br />
reduction of at least 30%, and 45 were<br />
randomized to Kuvan (20 mg/kg/day) or<br />
placebo <strong>in</strong> a 3:1 ratio, and enrolled <strong>in</strong> <strong>the</strong><br />
10-week double-bl<strong>in</strong>d, placebocontrolled<br />
portion of <strong>the</strong> study (Part 2).<br />
For <strong>the</strong> first three weeks, patients<br />
ma<strong>in</strong>ta<strong>in</strong>ed <strong>the</strong>ir pre-exist<strong>in</strong>g restricted<br />
diet with no supplementation of<br />
phenylalan<strong>in</strong>e. Thereafter, every o<strong>the</strong>r<br />
week, specific amounts of phenylalan<strong>in</strong>e<br />
were added (or removed) to <strong>the</strong><br />
restricted diet of each patient accord<strong>in</strong>g<br />
to pre-def<strong>in</strong>ed blood phenylalan<strong>in</strong>e<br />
levels. The maximum amount of Phe that<br />
could be added to a patient diet dur<strong>in</strong>g<br />
<strong>the</strong> study was 50 mg/kg/day.<br />
IL-1 is a soluble prote<strong>in</strong> secreted by<br />
certa<strong>in</strong> cells <strong>in</strong> <strong>the</strong> body, which acts as a<br />
messenger to help regulate immune and<br />
<strong>in</strong>flammatory responses by attach<strong>in</strong>g to<br />
cell-surface receptors on cells that<br />
participate <strong>in</strong> <strong>the</strong> body's immune system.<br />
In excess, it can be harmful and has<br />
been l<strong>in</strong>ked to a variety of <strong>in</strong>flammatory<br />
diseases. The IL-1 Trap is designed to<br />
attach to and neutralize IL-1 <strong>in</strong> <strong>the</strong> blood<br />
PAH is not present<br />
<strong>in</strong> sufficient<br />
quantities, Phe<br />
accumulates to<br />
abnormally high<br />
levels <strong>in</strong> <strong>the</strong> blood<br />
and bra<strong>in</strong> result<strong>in</strong>g<br />
<strong>in</strong> a variety of<br />
complications,<br />
<strong>in</strong>clud<strong>in</strong>g severe<br />
mental retardation<br />
and bra<strong>in</strong> damage,<br />
mental illness,<br />
seizures and<br />
tremors, and<br />
cognitive<br />
problems.<br />
CAPS is a family<br />
of rare<br />
auto<strong>in</strong>flammatory<br />
diseases,<br />
<strong>in</strong>clud<strong>in</strong>g Familial<br />
Cold Auto<strong>in</strong>flammatory<br />
Syndrome (FCAS)<br />
and Muckle Wells<br />
Syndrome (MWS).<br />
NA $100-$200<br />
per day<br />
Updated 8/2007<br />
Copyright © 2007 Medco Health Solutions, Inc.<br />
10