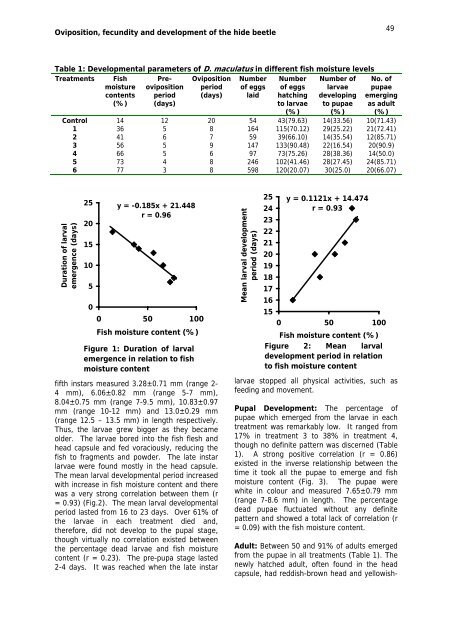
Oviposition, fecundity and development <strong>of</strong> the hide beetle49Table 1: Developmental parameters <strong>of</strong> D. maculatus in different fish moisture levelsTreatments Fishmoisturecontents(%)Preovipositionperiod(days)Ovipositionperiod(days)Number<strong>of</strong> eggslaidNumber<strong>of</strong> eggshatchingto larvae(%)Number <strong>of</strong>larvaedevelopingto pupae(%)No. <strong>of</strong>pupaeemergingas adult(%)Control 14 12 20 54 43(79.63) 14(33.56) 10(71.43)1 36 5 8 164 115(70.12) 29(25.22) 21(72.41)2 41 6 7 59 39(66.10) 14(35.54) 12(85.71)3 56 5 9 147 133(90.48) 22(16.54) 20(90.9)4 66 5 6 97 73(75.26) 28(38.36) 14(50.0)5 73 4 8 246 102(41.46) 28(27.45) 24(85.71)6 77 3 8 598 120(20.07) 30(25.0) 20(66.07)Duration <strong>of</strong> larvalemergence (days)2520151050y = -0.185x + 21.448r = 0.960 50 100Fish moisture content (%)Figure 1: Duration <strong>of</strong> larvalemergence in relation to fishmoisture contentfifth instars measured 3.28±0.71 mm (range 2-4 mm), 6.06±0.82 mm (range 5-7 mm),8.04±0.75 mm (range 7-9.5 mm), 10.83±0.97mm (range 10-12 mm) and 13.0±0.29 mm(range 12.5 – 13.5 mm) in length respectively.Thus, the larvae grew bigger as they becameolder. The larvae bored into the fish flesh andhead capsule and fed voraciously, reducing thefish to fragments and powder. The late instarlarvae were found mostly in the head capsule.The mean larval developmental period increasedwith increase in fish moisture content and therewas a very strong correlation between them (r= 0.93) (Fig.2). The mean larval developmentalperiod lasted <strong>from</strong> 16 to 23 days. Over 61% <strong>of</strong>the larvae in each treatment died and,therefore, did not develop to the pupal stage,though virtually no correlation existed betweenthe percentage dead larvae and fish moisturecontent (r = 0.23). The pre-pupa stage lasted2-4 days. It was reached when the late instarMean larval developmentperiod (days)2524232221201918171615y = 0.1121x + 14.474r = 0.930 50 100Fish moisture content (%)Figure 2: Mean larvaldevelopment period in relationto fish moisture contentlarvae stopped all physical activities, such asfeeding and movement.Pupal Development: The percentage <strong>of</strong>pupae which emerged <strong>from</strong> the larvae in eachtreatment was remarkably low. It ranged <strong>from</strong>17% in treatment 3 to 38% in treatment 4,though no definite pattern was discerned (Table1). A strong positive correlation (r = 0.86)existed in the inverse <strong>relationship</strong> between thetime it took all the pupae to emerge and fishmoisture content (Fig. 3). The pupae werewhite in colour and measured 7.65±0.79 mm(range 7-8.6 mm) in length. The percentagedead pupae fluctuated without any definitepattern and showed a total lack <strong>of</strong> correlation (r= 0.09) with the fish moisture content.Adult: Between 50 and 91% <strong>of</strong> adults emerged<strong>from</strong> the pupae in all treatments (Table 1). Thenewly hatched adult, <strong>of</strong>ten found in the headcapsule, had reddish-brown head and yellowish-
EZENWAJI, Henry Maduka Godfrey and OBAYI, Nicodermus Sunday50Duration <strong>of</strong> pupalemergence (days)454035302520151050y = -0.4297x + 44.571r = 0.860 50 100Fish moisture content (%)Figure 3: Duration <strong>of</strong> pupalemergence in relation to fishmoisture contentbrown elytra, which later became black. Theabdomen was white in colour. There wasmoderate correlation (r = 0.77) in the inverse<strong>relationship</strong> between percentage eggs whichdeveloped to adult and the fish moisturecontent.DISCUSSIONThe longer pre-oviposition and ovipositionperiods <strong>of</strong> D. maculatus females in the controlare attributed to the low fish moisture content(14%). This level <strong>of</strong> fish moisture seems toprohibit oviposition by the beetle and probablyexplains why ‘banda’, made up <strong>of</strong> cut pieces <strong>of</strong>mainly Clarias species, is charred and dried tomoisture content <strong>of</strong> 15% before storage (Osuji,1977). To provide a better condition for egglaying, the fish in the control absorbed moisture<strong>from</strong> the free water in the plate which thenenabled it to became gradually more conducivefor egg deposition. This process wasundoubtedly accomplished over a period <strong>of</strong>time, hence the longer oviposition period andthe fewer number <strong>of</strong> eggs (54) laid. Ourpreliminary investigation before the start <strong>of</strong> thisstudy showed that dipterans, particularly blowflies, found very high levels <strong>of</strong> fish moisturecontent extremely suitable for egg deposition,as also do D. maculatus females <strong>of</strong> this study,which laid very high numbers <strong>of</strong> eggs on fishwith moisture content <strong>of</strong> 73% and 77%.However, the vast majority <strong>of</strong> the eggs laid atthese high fish moisture levels died resulting inthe low percentage ( < 42%) number <strong>of</strong> eggshatching to larvae as against the highpercentage (> 66%) observed at the other fishmoisture levels, which had fewer eggs (Table1). It is probably this observation that led Osuji(1977) and Johnson and Esser (2000) to assertthat fish with moisture content <strong>of</strong> 32-47% arevery suitable for infestation with dermestideggs.The fecundity <strong>of</strong> female D. maculatus (54-598eggs), though extremely variable, comparesfavourably with that reported for dermestids byKreyenberg (1928, 198 – 845 eggs), Taylor(1964, 160 eggs), Osuji (1985, 250 eggs) andMichael (2000, 318 eggs). The lower limit <strong>of</strong> 54eggs recorded in the control is the lowestdermestid fecundity so far reported. It appearsthat the eggs laid on the fish with lowermoisture content (14-66%) at the warm, dryperiod <strong>of</strong> January to April were very viable suchthat a higher percentage <strong>of</strong> them survived toadult life. This is consistent with Osuji(1974a,b), Proctor (1977) and Barwal and Devi(1993) who have shown acceleration <strong>of</strong>development in dermestids during the warm,dry season, given fish with the right moisturelevel and high lipid content. C. ebriensis, aswell as other Clarias species, has high lipid level<strong>of</strong> > 16.4% (Osuji, 1974b; Ezenwaji, 1989) andthis nutritional requirement makes the clariidgood substrate for the rapid development <strong>of</strong> D.maculatus. That this is indeed so iscollaborated by the short larval developmentalstages and period reported here. The larvalstages (5 instars) are short, falling into thelower limit reported by Osuji (1975, 1985, 5 - 7instars) but contrasting with Taylor (1964, 6 -10 instars) and Michael (2000, 7 - 9 instars).Similarly, the larval developmental period <strong>of</strong> 16-23 days is considerably less than those reportedby these workers (Taylor, 1964, 39 – 46 days;Osuji, 1975, 33.5 days; Michael, 2000, 50 days).The temperature (29 0 C) and relative humidity(58%) which prevailed during the warm, dryperiod also appear to be optimum for dermestiddevelopment.An overview shows that female D.maculatus is able to lay eggs on fish withvarying moisture levels (14-77%). The eggs atthe lower moisture levels (14-66%) survivemuch better than the eggs laid at higher fishmoisture levels. Low moisture levels, thenutritional status <strong>of</strong> C. ebriensis and the climaticconditions enable acceleration <strong>of</strong> thedevelopment <strong>of</strong> D. maculatus in C. ebriensis. Itmay be in this way that high numbers <strong>of</strong> D.maculatus are maintained in dry Clarias speciesmarketed in Nigeria resulting in high quality andeconomic losses.
- Page 2 and 3: Animal ResearchInternational ®Anim
- Page 4 and 5: Animal Research International (2004
- Page 6 and 7: Length-weight relationships of fish
- Page 9 and 10: EZENWAJI, Henry Maduka Godfrey 6sta
- Page 11 and 12: NGWU, Godwin Ikechukwu et al.8Studi
- Page 13 and 14: NGWU, Godwin Ikechukwu et al.betwee
- Page 15 and 16: Animal Research International (2004
- Page 17 and 18: Metal concentrations in Alau reserv
- Page 19 and 20: Animal Research International (2004
- Page 21 and 22: Oxygen consumption rate of crab18Ef
- Page 23 and 24: Oxygen consumption rate of crab20O
- Page 25 and 26: Oxygen consumption rate of crab22MA
- Page 27 and 28: UBACHUKWU, Patience Obiageli24group
- Page 29: UBACHUKWU, Patience Obiageli26Table
- Page 32 and 33: Perception and treatment of onchoce
- Page 34 and 35: Animal Research International (2004
- Page 36 and 37: Differences in meristic counts of t
- Page 38 and 39: Differences in meristic counts of t
- Page 40 and 41: EYO, Joseph Effiong and INYANG, Nic
- Page 42 and 43: EYO, Joseph Effiong and INYANG, Nic
- Page 44 and 45: EYO, Joseph Effiong and INYANG, Nic
- Page 46 and 47: OSAKWE, Isaac, Ikechukwu and STEING
- Page 48 and 49: OSAKWE, Isaac, Ikechukwu and STEING
- Page 50 and 51: Animal Research International (2004
- Page 54 and 55: Oviposition, fecundity and developm
- Page 56 and 57: OLUAH, Ndubuisi Stanley and MGBENKA
- Page 58 and 59: OLUAH, Ndubuisi Stanley and MGBENKA
- Page 60 and 61: Animal Research International (2004
- Page 62 and 63: Community participation in the cont
- Page 64 and 65: Community participation in the cont
- Page 66 and 67: Community participation in the cont
- Page 68 and 69: OKAFOR, Fabian Chukwuemenam et al.6
- Page 70 and 71: OKAFOR, Fabian Chukwuemenam et al.6
- Page 72 and 73: OKAFOR, Fabian Chukwuemenam et al.6