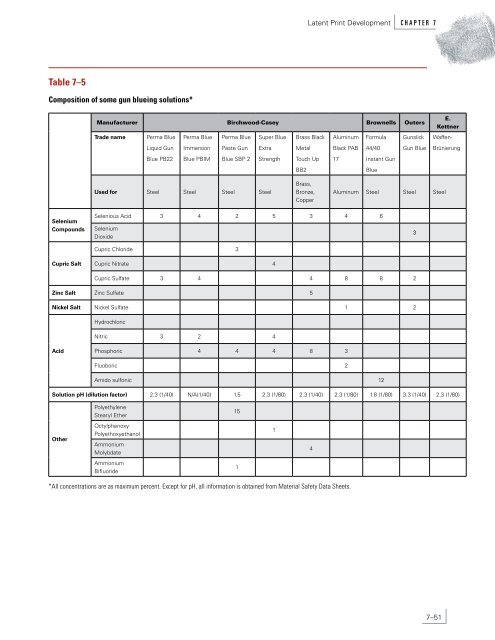
C H A P T E R 7 <strong>Latent</strong> <strong>Print</strong> <strong>Development</strong> As each of these reagents etches, the metal ions get deposited on what is etched. For example, on aluminum, the oxidation and reduction (etching and deposition) reaction is 3H 2 SeO 3 + 12H + + 4Al 3Se + 9H 2 O + 4Al 3+ E o = 2402 mV redox 3Cu 2+ + 2Al 3Cu + 2Al 3+ E o redox = 2003.9 mV If both reagents are present together, as in the gun blueing solution, then the ratio of Cu to Se that deposits depends on the speed (kinetics) of each of the two competing reactions; it is possible that a 1:1 Cu–Se alloy is formed, but it is not certain. The final result is a blue-black metallic coating (everywhere except where a sebaceous latent print exists). The composition of some gun blueing solutions is given in Table 7–5. Note that all involve selenious acid (one involves selenium dioxide, which is the anhydrous form of selenious acid), a cupric salt, and an acid. Interestingly, Bentsen et al. (1996, pp 3–8) used a 0.4% selenious acid solution (without cupric ions or acid) to develop prints on metal surfaces such as spent cartridge cases. This deposits selenium metal on the metal being treated and this solution, along with the vacuum cyanoacrylate ester treatment, was rated highly among other methods tested. 7.13.4.4 Formulations. Cantu et al. (1998, pp 294–298) recommend a 1:80 dilution of a commercial gun blueing solution. Leben and Ramotowski (1996, pp 8, 10) recommend a stronger solution (a 1:40 dilution) and indicate that an improvement over just using the diluted gun blueing reagent is to treat the metallic specimens first (e.g., cartridge cases) with CA fumes. Table 7–6 is a summary of their recommendations. 7.13.4.5 Application. Since its introduction by the BKA, gun blueing is now used in several laboratories because of its ease. As mentioned above, a CA ester treatment prior to gun blueing improves the detection of latent prints on metal. After treatment, there is a tendency for the gun blueing solution to continue its deposition, and several arresting methods have been proposed (Cantu et al., 1998, pp 294–298). These include dipping in a sodium bicarbonate solution, dipping in clear varnish (Bentsen et al., 1996, pp 3–8), applying a lacquer spray (private communication from Anton Theeuwen and Josita Limborgh, Netherlands Ministry of <strong>Justice</strong>, Forensic Science Laboratory), and using fingernail polish (private communication from Vici Inlow, U.S. Secret Service forensic laboratory). 7–50 If overdevelopment occurs, then acidified hydrogen peroxide is recommended for removing excess gun blue deposit (Cantu et al., 1998, pp 294–298). If we assume the copper– selenium alloy is a 1:1 adduct, then the net reaction for its removal is 3H 2 O 2 + 2H + + Cu–Se H 2 SeO 3 + Cu 2+ + 3H 2 O A suggested composition for the acidified hydrogen peroxide solution is 5 volumes of household vinegar (5% acetic acid) and 7 volumes of household hydrogen peroxide (3% hydrogen peroxide). This is based on stoichiometry of the above equation. However, a 1:1 mixture also works well. 7.13.4.6 Comments on Etching. Etching without metal deposition can also reveal prints on metal surfaces by the contrast formed between the etched background and the unetched latent print. From Table 7–4 it can be seen that acid can displace iron, lead, nickel, zinc, and aluminum. Acidified hydrogen peroxide, however, will also displace copper. Cantu et al. (1998, pp 294–298) noted that acidified hydrogen peroxide visualizes prints rather well on many metal surfaces. They provide a lengthy discussion and explanation of why the etching process should be carefully watched: the metals that are etched out as ions can redeposit as the process continues. Schütz et al. (2000, pp 65–68) compared etching and gun blueing methods with the multimetal deposition method on their ability to develop latent prints on cartridge cases. They found that (1) gun blueing excelled in visualizing sebaceous prints on brass cartridge cases, (2) for aluminum cartridge cases, MMD worked best (the modified physical developer step, performed after the colloidal gold step, brought out the print contrast), and (3) nothing worked well for lacquered steel cartridge cases. For the latter, they recommend CA fuming. 7.13.5 Sudan Black B 7.13.5.1 History and Background. Sudan black B (herein referred to as Sudan black) was initially used in laboratories for biological testing or chemical screening for fatty components (Figure 7–24). The reaction produces a blue-black product or image. Sudan black was initially reported for use as a friction ridge development technique in 1980 by Mitsui, Katho, Shimada, and Wakasugi of the <strong>Criminal</strong> Science Laboratory in Nagoya-shi, Japan (Mitsui et al., 1980, pp 9–10; 1981, pp 84–85).
Table 7–5 Composition of some gun blueing solutions* Selenium Compounds Cupric Salt Manufacturer Birchwood-Casey Brownells Outers Trade name Perma Blue Liquid Gun Blue PB22 Perma Blue Immersion Blue PBIM Perma Blue Paste Gun Blue SBP 2 Used for Steel Steel Steel Steel Super Blue Extra Strength Brass Black Metal Touch Up BB2 Brass, Bronze, Copper Aluminum Black PAB 17 Formula 44/40 Instant Gun Blue Selenious Acid 3 4 2 5 3 4 6 Selenium Dioxide Cupric Chloride 3 Cupric Nitrate 4 Gunslick Gun Blue Aluminum Steel Steel Steel Cupric Sulfate 3 4 4 8 8 2 Zinc Salt Zinc Sulfate 5 Nickel Salt Nickel Sulfate 1 2 Acid Hydrochloric Nitric 3 2 4 Phosphoric 4 4 4 8 3 Fluoboric 2 Amido sulfonic 12 3 E. Kettner Waffen- Brünierung Solution pH (dilution factor) 2.3 (1/40) N/A(1/40) 1.5 2.3 (1/80) 2.3 (1/40) 2.3 (1/80) 1.8 (1/80) 3.3 (1/40) 2.3 (1/80) Other Polyethylene Stearyl Ether Octylphenoxy Polyethoxyethanol Ammonium Molybdate Ammonium Bifluoride *All concentrations are as maximum percent. Except for pH, all information is obtained from Material Safety Data Sheets. 15 1 1 <strong>Latent</strong> <strong>Print</strong> <strong>Development</strong> C H A P T E R 7 4 7–51