Magnetic Susceptibility
Magnetic Susceptibility
Magnetic Susceptibility
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
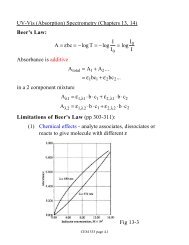
CEM 472 Fall 2000<strong>Magnetic</strong> <strong>Susceptibility</strong>IntroductionIn this experiment the magnetic susceptibility of transition metal ion complexes will be measured. Basedupon the sign and magnitude of the susceptibilities, substances will be classified as paramagnetic ordiammagnetic. The number of unpaired electrons in each paramagnetic complex will be determined andusing this information, the ligand field strength in each complex will be assessed. Two methods will beused to measure magnetic susceptibility: a Gouy method using aqueous solutions of the complex ions anda Gouy balance, and the Evans method using solid salts of the complex ions and a portable magneticsusceptibility balance.ProcedureI. Gouy MethodAll but one of the solutions for this part of the experiment will be provided. Make sure that you get thehandout from the TA that gives the concentration and density of these solutions – you’ll need thisinformation in order to do the calculations. Ferrous ammonium sulfate (F.W. 392.14 g/mol) is subject toslow air oxidation so you will need to prepare this solution yourself and determine its density. To do this,weigh a clean, dry 25-mL volumetric flask. Weigh by difference the appropriate amount of(NH 4 ) 2 Fe(SO 4 ) 2 ⋅6H 2 O to make a ~0.5 M solution. Add it to the flask and partially fill with distilled water.Swirl and shake to mix. When completely dissolved, fill to the mark with distilled water. Reweigh thevolumetric flask and calculate the actual concentration and density of the solution. Be sure to mix thesolution well before determining the magnetic susceptibility of this solution.The Gouy balance you will be using for this experiment consists of an analytical balance and anelectromagnet. A solution of known concentration and density is placed in a Gouy tube and hung onfishing line from the bottom of the analytical balance. The change in weight is measured when themagnetic field is turned on.Operation of the Gouy balance:1) Operation of the electromagnet:a) Make sure that the cooling water and the chiller are on. These are used for other instruments inthe lab, so they may be on when you come to lab. Do not turn this off unless you are sure that noone else is working in the lab.b) Turn the controller on by pressing the toggle on the lower left front side. The oscilloscope willshow a noisy signal – just ignore it.c) Set the magnetic field to 7,000 Gauss. The knobs are a little hard to turn.d) The magnet is turned on by turning the 10,000 th ’s place from “OFF” to “0”.2) The analytical balance responds to the magnetic field with an increase in the measured weight. Thisvalue is sensitive to both the magnitude of the applied field and to the position of the balance. Youmust measure this response and then subtract it from the change in weight measured for your samples.To measure:a) Put a cap on the empty Gouy tube and secure it with rubber bands. With the magnet off, hang theempty Gouy tube from the fishing line so that it is centered between the poles of the magnet andsuspended so that its middle is at the center of the poles. This may require an adjustment of the
length of the fishing line. (NOTE: When measuring solutions, the solution only fills the top halfof the Gouy tube. In order to get good measurements, the bottom of the solution should be wherethe field is the strongest (center of the poles) and the top where it is weakest (at or above thepoles). Make these adjustments now and then use the same Gouy tube for all measurements.)b) Stop the tube from swinging then press the “Tare” button (right side of the long bar). Thebalance should read 0.0000 g fairly steadily. You may need to press it again.c) Turn on the magnet and wait for the weight to stop changing. This may take about a minute.Record this value and turn the magnet off.d) Wait a minute or two and repeat these steps. Do this for a total of three times. If you do not get areasonably consistent reading, repeat again.e) IMPORTANT: Anytime that you move the balance, you need to re-determine the response of theempty tube.3) The TA will provide you with solutions of all of the complex ions to be studied except for ferrousammonium sulfate. You will also be given the density of these solutions.4) Measuring the sample:a) Using a pasteur pipette rinse the Gouy tube with distilled water. Then rinse the tube with severalportions of the solution to be analyzed, putting the wash solution into the waste containerprovided. Finally, carefully fill the tube to the mark with the solution. Secure the top with therubberbands and hang the tube from the line.b) Check the position and remember that if you have to shift the balance you need to re-determinethe response of the empty tube to the field.c) Make sure that the tube is not swinging. Tare the balance. Turn on the magnetic field. Wait forthe weight to stabilize and record the value.d) Turn off the magnet.e) Do the above in triplicate, checking for reproducibility.f) Empty the tube using a pasteur pipette and put the solution into the waste container provided.II. Evans MethodUsing the Gouy method, the electromagnet is stationary and the sample is pulled into or pushed out of themagnet when the magnetic field is turned on. The force that the magnet exerts on the sample is measuredby the analytical balance. The portable magnetic susceptibility balance uses the same configuration as theGouy method but the sample is stationary and the magnet is suspended. The equal and opposite forcewhich the sample exerts on a suspended permanent magnet is measured.Pairs of magnets are fixed at opposite ends of a beam which is balanced on a fulcrum. Without a sample,the system is in balance. Introduction of a sample between the poles of one pair of magnets produces adeflection of the beam. The extent that it tips depends upon the magnetic susceptibility of the sample.The deflection is registered by means of photodiodes. A current passes through a coil mounted betweenthe poles of the second pair of magnets in such a way as to return the system to equilibrium. Atequilibrium, the current through the coil is proportional to the force exerted by the sample and can bemeasured as a voltage drop.2
Operation of the portable <strong>Magnetic</strong> <strong>Susceptibility</strong> Balance1) Turn the range knob to the x1 scale and allow 10 minutes for the apparatus to warmup.2) Put a clean, empty sample tube in the balance and adjust the zero knob until the display reads 000.3) Pack a sample tube with the sample to be analyzed:a) The sample should be a reasonably fine powder. If necessary, crush crystalline aggregatesusing a non-magnetic spatula (small ferromagnetic fragments can introduce large errors to theexperiment).b) A small amount of the solid is then introduced to a WEIGHED sample tube, and the bottomgently tapped a number of times to shake the solid down. Homogeneous packing is essentialto making accurate measurements. Repeat until the length of sample is 2.5 – 3.5 cm.c) Weigh the filled sample tube and calculate the mass of the sample m.d) With the top of the sample horizontal, measure the length l of the sample.4) Place the packed sample tube into the guide and take the reading R. Note the sign, a negative readingindicates that the sample is diamagnetic. If the reading is off scale, turn the range knob to the x10scale, re-zero the balance with an empty tube, and multiply the reading by 10.5) Remove the sample from the balance and tap it on the benchtop several times, then replace it in thebalance. If the sample was well packed value of R will be unchanged. If R is different, repeat thisprocedure until a constant value of R is obtained.6) Repeat the procedure for a second sample of the same powder.7) The mass susceptibility χ mass can be calculated using the following:χwhere C is the calibration constant for the balance (1.258), l is the length of the sample in centimeters,R is the reading on the balance with the sample in the tube, and m is the mass of the sample in grams.The mass susceptibility will then be in c.g.s. units. It can be converted to S.I. units (those used in theGouy balance calculations) using the following relationship:S.I. mass susceptibility χ mass : multiply c.g.s. value by 4π ×10 -3The S.I. units on mass susceptibility are m 3 /kg.ClR=10 mmass 9Data AnalysisUsing the mass susceptibility determined for the nickle chloride solution, determine the apparatusconstant for the Gouy balance.Determine the mass and molar susceptibilities for each of the substances studied. Make thisdetermination based upon the results of both analysis methods.Determine if the complexes are diamagnetic or paramagnetic.Based upon your results, calculate the number of unpaired electrons for each paramagnetic substancestudied. From this determine if the metal ion complex is a high or low spin octahedral complex.3
QuestionsCompare the mass susceptibility of the measurements made with the Gouy balance and the portablesusceptibility balance with literature values.Comment on the accuracy of the two methods and suggest sources of error.For each complex ion, compare the number of electrons calculated for the complex with the possibleelectron configurations of the transition metal ion.Comment on the spin type and ligand field strength of each complex studied.4