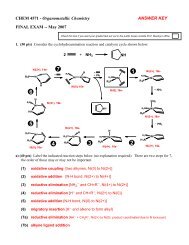
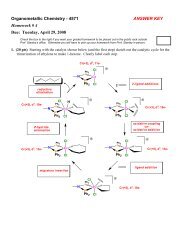
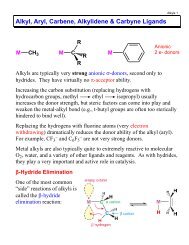
REDOX & Electrochemistry - LSU Chemistry
REDOX & Electrochemistry - LSU Chemistry
REDOX & Electrochemistry - LSU Chemistry
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
<strong>REDOX</strong> 19To calculate the voltage of an electrochemical cellone can simply add together the two half-cellreactions that make up the overall galvanic(electrochemical) cell.Let’s calculate the spontaneous cell potential(voltage) for our Cu/Zn cell. First look up the twohalf cell reactions from the table of StandardReduction Potentials:Cu 2+ (aq) + 2e - Cu(s) +0.34Zn 2+ (aq) + 2e - Zn(s) -0.76The more positive potential (relative to thehydrogen electrode) for the Cu 2+ reductioncompared to the Zn 2+ reduction means that Cu 2+ isa stronger oxidizing agent (wants to be reducedmore) than Zn 2+ .