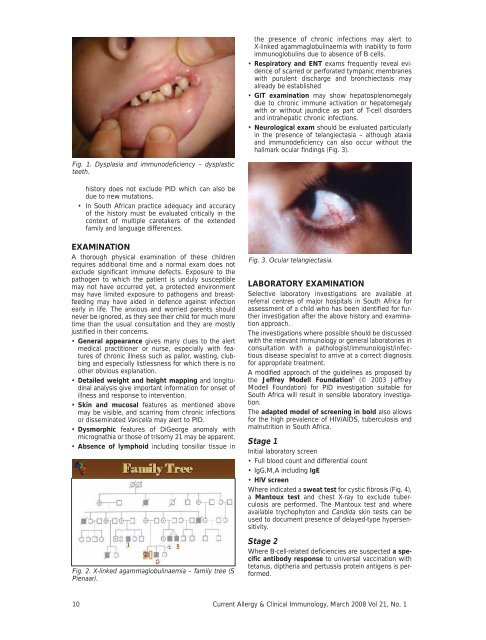
Burkholderia cepacia may be serious warning signsof significant lack of cellular immunity or phagocytedysfunction respectively.Recurrent pneumococcal infections may signal lackof specific antibody or IRAK4 deficiency. The recurrenceof a pneumonia or pneumonia with anunusual/opportunistic organism such as Pneumocystiscarinii or infections with Giardia should beinvestigated.Recurrence of meningococcal meningitis should precipitateinvestigation for complement deficiencies.Deficient antibody responses especially to polysaccharideantigens may persist after infancy <strong>and</strong> result inrecurrent respiratory infections despite normal levelsof immunoglobulins.Recurrence of infection at one site obviously shouldalert to anatomical defects or obstructions.Recurrent fevers in an otherwise healthy child whereno pathogens can be documented <strong>and</strong> resolution isspontaneous may signify one of the periodic fever syndromeswhere maintenance treatment with colchicinein some variants may prevent onset of amyloidosis.More recently described defects of the interferongamma <strong>and</strong> interleukin 12, as well as the nuclear factorkappa B essential modulator (NEMO), pathways maypresent with disseminated mycobacterial <strong>and</strong>Salmonella infections. Patients with NEMO mutationsalso frequently have features of ectodermal dysplasiaof varying degrees such as conical teeth, sparse hair<strong>and</strong> decreased or absent sweat production.The Jeffrey Modell Foundation Medical AdvisoryBoard 6 has also developed a useful list of 10 warningsigns which include most of the above, as well as a relevantfamily history, <strong>and</strong> these should prompt immunologicalinvestigations.Contributing factors for recurrent infections in childhoodmust be excluded early on to prevent unnecessary<strong>and</strong> costly investigation for PID:• Increased exposure including overcrowded housing<strong>and</strong> day care• Second-h<strong>and</strong> smoke• Atopy/asthma• Foreign body• Cystic fibrosis – incidence of about 1/2 500 in someCaucasian populations• Anatomical obstructions• Gastro-oesophageal reflux• Prematurity• Not having been breastfedMedical causes/associations for secondary oracquired immunodeficiency may mimic PID <strong>and</strong> mustbe investigated if appropriate:• Malnutrition• Infectious diseases – suspected HIV, Epstein Barrvirus, Mycobacterium tuberculosis, Cryptococcus• Malignancy• Immune suppressant treatment• Chronic inflammatory diseases such as systemiclupus erythematosus, rheumatoid arthritis• Protein loss• Chronic illness of organ systems• Asplenia, sickle cell diseaseHISTORYAfter review of the above factors <strong>and</strong> associations,specific questions are important which will guide theexamination <strong>and</strong> further investigation for PID:• History of age of onset of infections. These detailsare very important as severe combined deficienciesare likely to start in the first months of life, whereasantibody deficiencies may only become symptomaticin later childhood even in the virtual absence ofserum IgG. Passively transferred maternal antibodiesare detectable until 18 months of life in the infant'sserum <strong>and</strong> prolonged breastfeeding may protectagainst common pathogens. Although most patientswith PID will present in infancy or early childhoodbecause of the combined effects of immaturity <strong>and</strong>inborn deficiency of the immune system, deficienciesother than severe combined forms may even bediagnosed as late as mid-adulthood despite significantmorbidity.• Detailed perinatal history <strong>and</strong> history of the pregnancy• Pathogen history can point towards the broadgroup of defects in the immune defence: 7T cells – Pneumocystis carinii, mycobacteriae,Cryptococcus neoformans,herpes virusesB cells – Streptocccus pneumoniae, Haemophilusinfluenzae, Giardia lamblia,enteroviruses, staphylococci, CampylobactersppComplement – Neisseria sppPhagocytes – staphylococci, Aspergillus spp,Burkholderia cepaciaBecause of the close co-operation between differentcomponents of the immune defence system these categories<strong>and</strong> divisions are artificial <strong>and</strong> serve as guidelinesonly. Overlap of functions also termed immunologicalredundancy can further obscure a specific susceptibility.Comprehensive practice parameters, classifications<strong>and</strong> algorithms are available for these specificdefects. 8-10• History for hallmark special features• Severe eczema <strong>and</strong> peticheae of Wiskott-Aldrichsyndrome• Delayed umbilical stump separation of leukocyteadhesion defect (although rarely observed in clinicalpractice) or poor wound healing• Postvaccination disseminated BCG or paralyticpolio in T-cell or B-cell disorders• Hypocalcaemic tetany of the neonate in DiGeorgeanomaly• Graft-versus-host disease caused by maternalengraftment or non-irradiated blood transfusion inT-cell defects• Arthritis which does not fit the defined categoriesof juvenile idiopathic arthritis (JIA) in agammaglobulinaemia(but also in aquired immunodeficiencysyndrome)• Autoimmunity of B-cell-related disorders or earlycomplement component deficiencies• Generalised molluscum contagiosum or extensivewarts of T-cell disorders• Dysplastic features (Fig. 1)• History of the family• Unexplained death in infancy or recurrent infectionhistory in maternal male relatives may be a clue toX-linked immunodeficiency where the mother isa carrier of the condition (Fig. 2).• A history of autoimmunity in family members maybe positive for the patient with common variableimmunodeficiency or IgA deficiency.• It is important to remember that a negative family<strong>Current</strong> <strong>Allergy</strong> & <strong>Clinical</strong> <strong>Immunology</strong>, <strong>March</strong> <strong>2008</strong> Vol 21, No. 1 9
the presence of chronic infections may alert toX-linked agammaglobulinaemia with inability to formimmunoglobulins due to absence of B cells.• Respiratory <strong>and</strong> ENT exams frequently reveal evidenceof scarred or perforated tympanic membraneswith purulent discharge <strong>and</strong> bronchiectasis mayalready be established• GIT examination may show hepatosplenomegalydue to chronic immune activation or hepatomegalywith or without jaundice as part of T-cell disorders<strong>and</strong> intrahepatic chronic infections.• Neurological exam should be evaluated particularlyin the presence of telangiectasia – although ataxia<strong>and</strong> immunodeficiency can also occur without thehallmark ocular findings (Fig. 3).Fig. 1. Dysplasia <strong>and</strong> immunodeficiency – dysplasticteeth.history does not exclude PID which can also bedue to new mutations.• In South African practice adequacy <strong>and</strong> accuracyof the history must be evaluated critically in thecontext of multiple caretakers of the extendedfamily <strong>and</strong> language differences.EXAMINATIONA thorough physical examination of these childrenrequires additional time <strong>and</strong> a normal exam does notexclude significant immune defects. Exposure to thepathogen to which the patient is unduly susceptiblemay not have occurred yet, a protected environmentmay have limited exposure to pathogens <strong>and</strong> breastfeedingmay have aided in defence against infectionearly in life. The anxious <strong>and</strong> worried parents shouldnever be ignored, as they see their child for much moretime than the usual consultation <strong>and</strong> they are mostlyjustified in their concerns.• General appearance gives many clues to the alertmedical practitioner or nurse, especially with featuresof chronic illness such as pallor, wasting, clubbing<strong>and</strong> especially listlessness for which there is noother obvious explanation.• Detailed weight <strong>and</strong> height mapping <strong>and</strong> longitudinalanalysis give important information for onset ofillness <strong>and</strong> response to intervention.• Skin <strong>and</strong> mucosal features as mentioned abovemay be visible, <strong>and</strong> scarring from chronic infectionsor disseminated Varicella may alert to PID.• Dysmorphic features of DiGeorge anomaly withmicrognathia or those of trisomy 21 may be apparent.• Absence of lymphoid including tonsillar tissue inFig. 2. X-linked agammaglobulinaemia – family tree (SPienaar).Fig. 3. Ocular telangiectasia.LABORATORY EXAMINATIONSelective laboratory investigations are available atreferral centres of major hospitals in South Africa forassessment of a child who has been identified for furtherinvestigation after the above history <strong>and</strong> examinationapproach.The investigations where possible should be discussedwith the relevant immunology or general laboratories inconsultation with a pathologist/immunologist/infectiousdisease specialist to arrive at a correct diagnosisfor appropriate treatment.A modified approach of the guidelines as proposed bythe Jeffrey Modell Foundation 6 (© 2003 JeffreyModell Foundation) for PID investigation suitable forSouth Africa will result in sensible laboratory investigation.The adapted model of screening in bold also allowsfor the high prevalence of HIV/AIDS, tuberculosis <strong>and</strong>malnutrition in South Africa.Stage 1Initial laboratory screen• Full blood count <strong>and</strong> differential count• IgG,M,A including IgE• HIV screenWhere indicated a sweat test for cystic fibrosis (Fig. 4),a Mantoux test <strong>and</strong> chest X-ray to exclude tuberculosisare performed. The Mantoux test <strong>and</strong> whereavailable trychophyton <strong>and</strong> C<strong>and</strong>ida skin tests can beused to document presence of delayed-type hypersensitivity.Stage 2Where B-cell-related deficiencies are suspected a specificantibody response to universal vaccination withtetanus, diptheria <strong>and</strong> pertussis protein antigens is performed.10 <strong>Current</strong> <strong>Allergy</strong> & <strong>Clinical</strong> <strong>Immunology</strong>, <strong>March</strong> <strong>2008</strong> Vol 21, No. 1