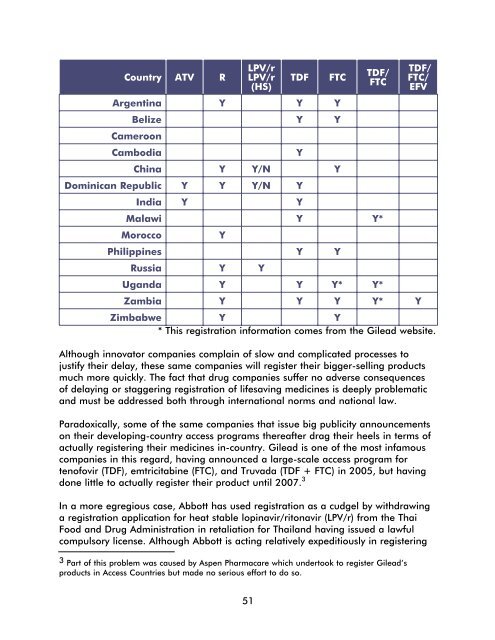
Country ATV RLPV/rLPV/r(HS)TDFArgentina Y Y YFTCBelize Y YCameroonCambodiaChina Y Y/N YDominican Republic Y Y Y/N YIndia Y YYTDF/FTCMalawi Y Y*MoroccoYPhilippines Y YRussia Y YUganda Y Y Y* Y*TDF/FTC/EFVZambia Y Y Y Y* YZimbabwe Y Y* This registration information comes from <strong>the</strong> Gilead website.Although innovator companies complain of slow and complicated processes tojustify <strong>the</strong>ir delay, <strong>the</strong>se same companies will register <strong>the</strong>ir bigger-selling productsmuch more quickly. The fact that drug companies suffer no adverse consequencesof delaying or staggering registration of lifesaving medicines is deeply problematicand must be addressed both through international norms and national law.Paradoxically, some of <strong>the</strong> same companies that issue big publicity announcementson <strong>the</strong>ir developing-country access programs <strong>the</strong>reafter drag <strong>the</strong>ir heels in terms ofactually registering <strong>the</strong>ir medicines in-country. Gilead is one of <strong>the</strong> most infamouscompanies in this regard, having announced a large-scale access program fortenofovir (TDF), emtricitabine (FTC), and Truvada (TDF + FTC) in 2005, but havingdone little to actually register <strong>the</strong>ir product until 2007. 3In a more egregious case, Abbott has used registration as a cudgel by withdrawinga registration application for heat stable lopinavir/ritonavir (LPV/r) from <strong>the</strong> ThaiFood and <strong>Drug</strong> Administration in retaliation for Thailand having issued a lawfulcompulsory license. Although Abbott is acting relatively expeditiously in registering3 Part of this problem was caused by Aspen Pharmacare which undertook to register Gilead’sproducts in <strong>Access</strong> Countries but made no serious effort to do so.51
LPV/r in developing countries, it is simply outrageous that drug companies cannot only delay registration with impunity, but that <strong>the</strong>y can use registration delaysor withdrawals to retaliate against lawful use of TRIPS-compliant flexibilities.Fortunately, Thai activists have filed a competition complaint to Thai authoritieschallenging <strong>the</strong> product withdrawal.It is important to note that at <strong>the</strong> same time that <strong>the</strong>y are delaying <strong>the</strong>ir marketentry, drug companies benefit from <strong>the</strong>ir pre-existing patent rights that give<strong>the</strong>m rights to exclude competitors (China). This is where local working rules incompulsory licensing schemes can play a role: failure to register <strong>the</strong> product andto satisfy <strong>the</strong> local market within a certain period of time can be an independentjustification for issuing a compulsory license.Delayed registration by generic companiesGeneric companies are also required to register <strong>the</strong>ir <strong>the</strong>rapeutically equivalentproducts before marketing <strong>the</strong>m, but <strong>the</strong>y too frequently prioritize registrationapplications based on market incentives, e.g., <strong>the</strong> size of <strong>the</strong> PLWHA communityand whe<strong>the</strong>r international donors are supporting treatment access. Because genericcompanies work on smaller margins, inefficient and costly registration processesare doubly problematic. For this reason, <strong>the</strong> newly WHO prequalified and FDAapprovedtriple fixed-dose pediatric formulation from Cipla is not widely available(e.g., Uganda). In <strong>the</strong> same token, although 142 ARVs have been WHO prequalifiedto date— <strong>the</strong> vast majority of <strong>the</strong>m generics, to <strong>the</strong> best of our knowledge— mostof <strong>the</strong>se products have not been separately registered in most developing countrymarkets, reducing competition and potential availability of alternative sources ofsupply.Global institutions are playing an insufficient role in expediting generic registrationwith three exceptions. The most proactive entity is <strong>the</strong> WHO PrequalificationProgramme, which has granted prequalification to 142 ARVs and multiple OIdrugs since it was launched in 2001. However, even if a generic companynavigates prequalification, it must still file separate registration applications ineach developing country where it wants to do business. In addition, <strong>the</strong> ClintonFoundation has worked behind <strong>the</strong> scenes to assist generic companies in itsconsortium to prosecute successful registration applications in Clinton Foundationbuying countries (Clinton Foundation consultation, Sept. 15, 2007). Finally, <strong>the</strong>U.S. Supply Chain Management System has been proactive in seeking to expediteregistration of FDA-approved products.However, in general, international institutions, including WHO and regionalorganizations like <strong>the</strong> Sou<strong>the</strong>rn African Development Community (SADC), havedone little to harmonize and streamline <strong>the</strong> registration process for genericproducers, to provide technical assistance in filing registration applications, or toprovide reliable market incentives for generic producers to take <strong>the</strong> risk of product52
- Page 1 and 2:
Missing the Target #5:Improving AID
- Page 4 and 5:
ArgentinaDr. María Lorena Di Giano
- Page 8 and 9: Executive SummaryAt the G8 meeting
- Page 12: • UN agencies should provide incr
- Page 16 and 17: Price should not be a barrier when
- Page 18 and 19: The Global Fund and UNITAID: The Fu
- Page 20 and 21: The real cost of free treatmentBy a
- Page 22 and 23: Remaining work for asuccessful prog
- Page 24 and 25: DOMINICAN REPUBLICto access rapid t
- Page 26 and 27: DOMINICAN REPUBLICIn Santo Domingo
- Page 28 and 29: ZIMBABWEtreatment, representing 35
- Page 30 and 31: ZIMBABWEAccording to the Medicines
- Page 32 and 33: ZIMBABWEZimbabwe’s application fo
- Page 34 and 35: ZIMBABWENational government• Addr
- Page 36 and 37: RUSSIAMigrantsRussia, with the seco
- Page 38 and 39: a person with HIV will die very qui
- Page 40 and 41: FamiliesIn Kenya, families are the
- Page 42 and 43: INDIA2. Provide pediatric formulati
- Page 44 and 45: Linking nutrition and treatmentBy W
- Page 46 and 47: Most people living with advanced HI
- Page 48 and 49: only available in the capital, Yaou
- Page 50 and 51: ZAMBIAFindings in particular provin
- Page 52 and 53: The DACA for Chadiza District was a
- Page 54 and 55: Many health care workers are inadeq
- Page 56 and 57: DRUG REGISTRATION BARRIERS & LOGJAM
- Page 60 and 61: development and registration. Likew
- Page 62 and 63: Capacity issues and delays in the W
- Page 64 and 65: Lack of post-approval quality assur
- Page 66 and 67: ARV PROCUREMENT, REGISTRATION,AND S
- Page 68 and 69: ARGENTINAParallel importingRelating
- Page 70 and 71: ARGENTINAAccess to ARVsNo difficult
- Page 72 and 73: Viral load tests are available, but
- Page 74 and 75: Cambodiaby Mony PenOnly pharmacists
- Page 76 and 77: ChinaBy anonymous Missing the Targe
- Page 78 and 79: CHINAThe current process for regist
- Page 80 and 81: Dominican RepublicBy Eugene Schiff
- Page 82 and 83: IndiaBy Abraham KK, Celina D’Cost
- Page 84 and 85: MalawiBy Lot Nyirenda and Grace Bon
- Page 86 and 87: MALAWIStock-outsAccording to an ext
- Page 88 and 89: MoroccoBy Othman MelloukMorocco has
- Page 90 and 91: Determine the patent status of all
- Page 92 and 93: MOROCCOThe shortage occurred becaus
- Page 94 and 95: As of November 2007, the unit cost
- Page 96 and 97: NIGERIADiagnostic testsDiagnostic t
- Page 98 and 99: The government’s initial inabilit
- Page 100 and 101: Increase training and capacity buil
- Page 102 and 103: treatment practice. The Minster ann
- Page 104 and 105: Ugandaby Richard Hasunira, Prima Ka
- Page 106 and 107: UGANDAExtracts from the MoH’s rep
- Page 108 and 109:
ZAMBIAIn gathering this information
- Page 110 and 111:
However, since manufacturing prices
- Page 112 and 113:
105SHORT SUMMARY
- Page 114:
PAKISTANAccess for marginalized gro