- Page 2 and 3:
Reference Tables for Physical Setti
- Page 4 and 5:
150. 140. Table G Solubility Curves
- Page 6 and 7:
Table I Heats of Reaction at 101.3
- Page 8 and 9:
Table O Symbols Used in Nuclear Che
- Page 10 and 11:
0 6.941 +1 Li 3 2-1 Na 39.0983 K +1
- Page 12 and 13:
First Atomic Symbol Name Ionization
- Page 14 and 15:
Collier County CHEMISTRY EXAM FORMU
- Page 16 and 17:
CHEMISTRY EXAM FORMULA AND RESOURCE
- Page 18 and 19:
Honors Chemistry Class Policies and
- Page 20 and 21:
Unit 1 (22 days) Chapter 1 Introduc
- Page 22 and 23:
4 Lorenzo Walker Technical High Sch
- Page 24 and 25:
MUSTANG LABORATORIES COMMITMENT TO
- Page 26 and 27:
Chapter 1 Unit 1 Introduction to Ch
- Page 28 and 29:
The Learning Goal for this assignme
- Page 30 and 31:
2. One cereal bar has a mass of 37
- Page 32 and 33:
1. How many meters are in one kilom
- Page 34 and 35:
The Learning Goal for this assignme
- Page 36 and 37:
RULE #3: To add/subtract in scienti
- Page 38 and 39:
The Learning Goal for this assignme
- Page 40 and 41:
Rule 3: A final zero or trailing ze
- Page 42 and 43: 24 For addition and subtraction, lo
- Page 44 and 45: Dimensional Analysis This is a way
- Page 46 and 47: Chapter 4 Unit 2 Atomic Structure T
- Page 48 and 49: The Learning Goal for this assignme
- Page 50 and 51: Looking at Ions We haven’t talked
- Page 52 and 53: Neutron Madness We have already lea
- Page 54 and 55: Create groups for these Scientist a
- Page 56 and 57: 38
- Page 58 and 59: The Learning Goal for this assignme
- Page 60 and 61: In the space below, write the unabb
- Page 62: Skyler Wild Name __________________
- Page 66 and 67: The Learning Goal for this Assignme
- Page 68 and 69: Atomic Size Define Atomic Size: The
- Page 70 and 71: Define Electronegativity: Electrone
- Page 72 and 73: Learning Goal for this section: Exp
- Page 74 and 75: Beta Particle Decay Beta decay is a
- Page 76 and 77: Inside the sun, fusion reactions ta
- Page 78 and 79: 58 Chapter 7 Unit 3 Ionic and Metal
- Page 80 and 81: The Learning Goal for this assignme
- Page 82 and 83: Ion Charge? What is the charge on f
- Page 84 and 85: Here are some examples for you: Zn
- Page 86 and 87: The Learning Goal for this assignme
- Page 88 and 89: C 2 H 6 O Ethanol CH 3 CH 2 O Step
- Page 90 and 91: Name Formula Charge Dichromate Cr
- Page 94 and 95: Carbon Dioxide Chemical Name CO 2 C
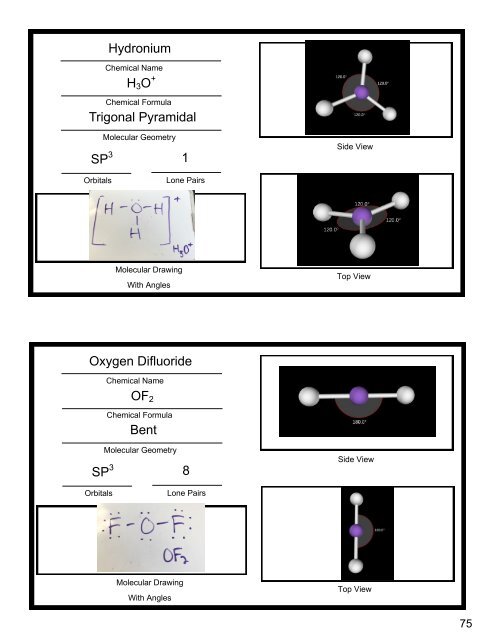
- Page 96 and 97: Hydronium Chemical Name H 3 O + Che
- Page 98 and 99: Sulfur Hexafluoride Chemical Name S
- Page 100 and 101: Bent: They look, well, bent. Bond a
- Page 102 and 103: 4. the chlorine atom in hydrochlori
- Page 104 and 105: Chapter 23 Functional Groups The st
- Page 106 and 107: 85
- Page 108 and 109: anched chains: Hexane - a 6-carbon
- Page 110 and 111: Isomers Because carbon can bond in
- Page 112 and 113: Chapter 12 Stoichiometry The studen
- Page 114 and 115: Other atomic weights are listed on
- Page 116 and 117: Find Molecular Mass 1- 10 1. NaBr 2
- Page 118 and 119: Step 3 Always leave hydrogen and ox
- Page 120 and 121: 1) ___ 2 NaNO3 + ___ 1 PbO ___ 1 P
- Page 122 and 123: 1) 2 NaNO3 + PbO Pb(NO3)2 + Na2O 2
- Page 124 and 125: 4) Double displacement: This is whe
- Page 126 and 127: Determine the Type of Reaction for
- Page 128 and 129: we can determine that 2 moles of HC
- Page 130 and 131: When there is no limiting reagent b
- Page 132 and 133: Example 7 How much 5M stock solutio
- Page 134 and 135: Theoretical and Actual Yields Key T
- Page 136 and 137: Review Purposes To get an over
- Page 138 and 139: 30.0gCl2 1 mol Cl2 1 mol S2Cl2 135g
- Page 140 and 141: Chapter 14 The Behavior of Gases Th
- Page 142 and 143:
Chapter 13: States of Matter Lesson
- Page 144 and 145:
Lesson #3: A Model for Solids: Esse
- Page 146 and 147:
It is important that you realize th
- Page 148 and 149:
3. The pressure of nitrogen gas in
- Page 150 and 151:
This formula is strictly valid only
- Page 152 and 153:
Electrolytes and Nonelectrolytes Ke
- Page 154 and 155:
Create and interpret potential ener
- Page 156 and 157:
Structure effects: determines what
- Page 158 and 159:
Specific Heat Here is the definitio
- Page 160 and 161:
Step One: solid ice rises in temper
- Page 162 and 163:
Step Three: liquid water rises in t
- Page 164 and 165:
Step Five: steam rises in temperatu
- Page 166 and 167:
then it is exothermic, meaning the
- Page 168 and 169:
Define the types of reactions. Give
- Page 170 and 171:
1. If this compound was at room tem
- Page 172 and 173:
1. In what part of the curve would
- Page 174 and 175:
Reduction Oxidation reduction react
- Page 176 and 177:
So far, the properties have an obvi
- Page 178 and 179:
The acid base theory of Brønsted a
- Page 180 and 181:
IV. Problems with the Theory This t
- Page 182 and 183:
Let's discuss significant figures a
- Page 184 and 185:
On your calculator you would input
- Page 186 and 187:
+1 0 0 +2 Ag+ --> Ag Reduction Cu -
- Page 188 and 189:
7. Half Reactions A half-reaction i
- Page 190:
Notice that each separate substance