Refractive Lens Surgery
Refractive Lens Surgery
Refractive Lens Surgery
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
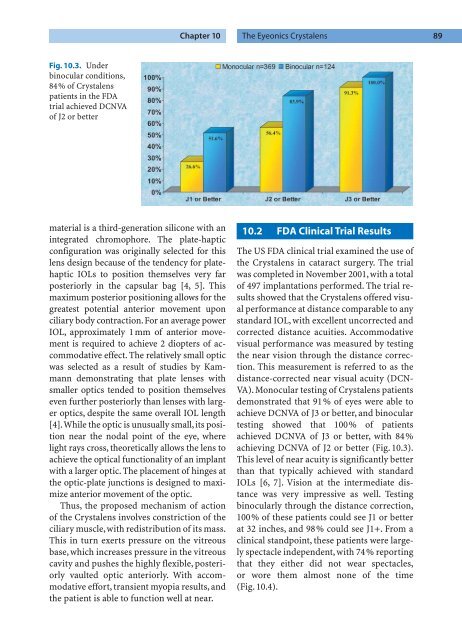
Fig. 10.3. Under<br />
binocular conditions,<br />
84% of Crystalens<br />
patients in the FDA<br />
trial achieved DCNVA<br />
of J2 or better<br />
material is a third-generation silicone with an<br />
integrated chromophore. The plate-haptic<br />
configuration was originally selected for this<br />
lens design because of the tendency for platehaptic<br />
IOLs to position themselves very far<br />
posteriorly in the capsular bag [4, 5]. This<br />
maximum posterior positioning allows for the<br />
greatest potential anterior movement upon<br />
ciliary body contraction. For an average power<br />
IOL, approximately 1 mm of anterior movement<br />
is required to achieve 2 diopters of accommodative<br />
effect. The relatively small optic<br />
was selected as a result of studies by Kammann<br />
demonstrating that plate lenses with<br />
smaller optics tended to position themselves<br />
even further posteriorly than lenses with larger<br />
optics, despite the same overall IOL length<br />
[4].While the optic is unusually small, its position<br />
near the nodal point of the eye, where<br />
light rays cross, theoretically allows the lens to<br />
achieve the optical functionality of an implant<br />
with a larger optic. The placement of hinges at<br />
the optic-plate junctions is designed to maximize<br />
anterior movement of the optic.<br />
Thus, the proposed mechanism of action<br />
of the Crystalens involves constriction of the<br />
ciliary muscle,with redistribution of its mass.<br />
This in turn exerts pressure on the vitreous<br />
base, which increases pressure in the vitreous<br />
cavity and pushes the highly flexible, posteriorly<br />
vaulted optic anteriorly. With accommodative<br />
effort, transient myopia results, and<br />
the patient is able to function well at near.<br />
Chapter 10 The Eyeonics Crystalens 89<br />
10.2 FDA Clinical Trial Results<br />
The US FDA clinical trial examined the use of<br />
the Crystalens in cataract surgery. The trial<br />
was completed in November 2001, with a total<br />
of 497 implantations performed. The trial results<br />
showed that the Crystalens offered visual<br />
performance at distance comparable to any<br />
standard IOL, with excellent uncorrected and<br />
corrected distance acuities. Accommodative<br />
visual performance was measured by testing<br />
the near vision through the distance correction.<br />
This measurement is referred to as the<br />
distance-corrected near visual acuity (DCN-<br />
VA). Monocular testing of Crystalens patients<br />
demonstrated that 91% of eyes were able to<br />
achieve DCNVA of J3 or better, and binocular<br />
testing showed that 100% of patients<br />
achieved DCNVA of J3 or better, with 84%<br />
achieving DCNVA of J2 or better (Fig. 10.3).<br />
This level of near acuity is significantly better<br />
than that typically achieved with standard<br />
IOLs [6, 7]. Vision at the intermediate distance<br />
was very impressive as well. Testing<br />
binocularly through the distance correction,<br />
100% of these patients could see J1 or better<br />
at 32 inches, and 98% could see J1+. From a<br />
clinical standpoint, these patients were largely<br />
spectacle independent, with 74% reporting<br />
that they either did not wear spectacles,<br />
or wore them almost none of the time<br />
(Fig. 10.4).