annual report 2011 - Office for Research - Northwestern University
annual report 2011 - Office for Research - Northwestern University
annual report 2011 - Office for Research - Northwestern University
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
Melina R. Kibbe<br />
Feinberg School of Medicine<br />
Stem Cells That Reside in the<br />
Arterial Wall May Control the<br />
Fate of Vascular Interventions<br />
Failure of vascular interventions, such as<br />
bypass grafting and balloon angioplasty<br />
and stenting, results from neointimal<br />
hyperplasia. Classically, this process has<br />
been thought to result from overgrowth of the cells<br />
that line the media—the middle layer of the arterial<br />
wall—and that ultimately leads to restenosis and arterial<br />
occlusion. In the last decade, however, this view has<br />
been changing, as it is now recognized that the arterial<br />
wall’s outer layer, the adventitial, may play a much<br />
more prominent role in this process. Specifically, the<br />
adventitia has been identified as home to a progenitor<br />
cell, the Sca1+ cell, which may be integral to the process<br />
of neointimal hyperplasia.<br />
Melina Kibbe, vascular surgery, has had a long-<br />
standing interest in the development and prevention<br />
of neointimal hyperplasia. Her ef<strong>for</strong>ts have focused on<br />
using nitric oxide as a therapeutic agent, given that nitric<br />
oxide is a potent inhibitor of neointimal hyperplasia.<br />
While studying the effects of nitric oxide on the<br />
vascular wall, Kibbe discovered that following vascular<br />
interventions, nitric oxide dramatically increases the<br />
Sca1+ progenitor cells residing in the adventitia just<br />
external to the external elastic lamina. Whether this<br />
increase elicited by nitric oxide is due to an increase in<br />
recruitment of these cells to the vascular wall following<br />
injury or due to inhibition of phenotypic differentiation<br />
of these cells to myofibroblasts, thereby preventing<br />
them from contributing to neointimal hyperplasia,<br />
remains to be determined.<br />
Kibbe was recently awarded a grant from the National<br />
Institutes of Health to study the role of Sca1+ cells<br />
following vascular interventions and nitric oxide therapy.<br />
Kibbe’s research has also been supported by grants from<br />
the Department of Veterans Affairs, the American Heart<br />
Association, the American Recovery and Reinvestment<br />
Act, and the <strong>Northwestern</strong> Memorial Foundation.<br />
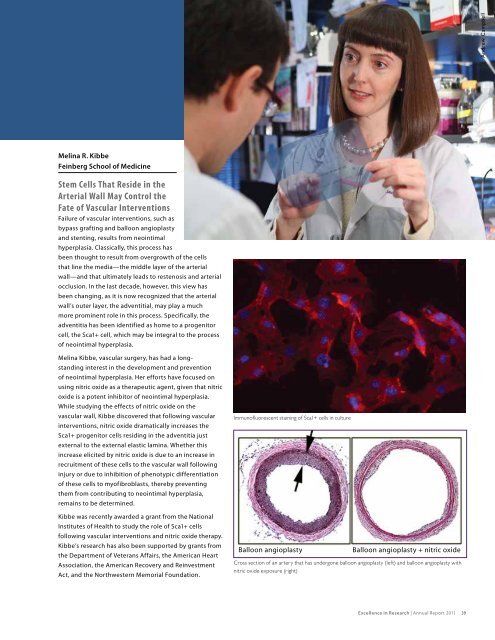
Immuno�uorescent staining of Sca1+ cells in culture<br />
Balloon angioplasty Balloon angioplasty + nitric oxide<br />
Cross section of an artery that has undergone balloon angioplasty (left) and balloon angioplasty with<br />
nitric oxide exposure (right)<br />
Excellence in <strong>Research</strong> | Annual Report <strong>2011</strong> 39<br />
Andrew Campbell