HPLC Analysis of Biomolecules Technical Guide - Interscience
HPLC Analysis of Biomolecules Technical Guide - Interscience
HPLC Analysis of Biomolecules Technical Guide - Interscience
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
12<br />
<strong>HPLC</strong> <strong>Analysis</strong> <strong>of</strong> <strong>Biomolecules</strong><br />
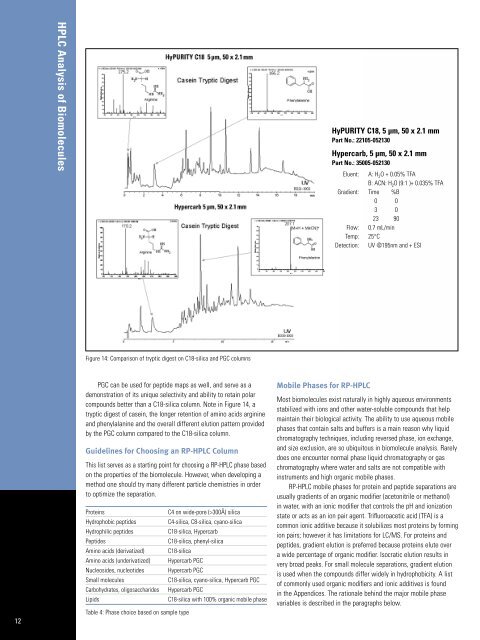
Figure 14: Comparison <strong>of</strong> tryptic digest on C18-silica and PGC columns<br />
PGC can be used for peptide maps as well, and serve as a<br />
demonstration <strong>of</strong> its unique selectivity and ability to retain polar<br />
compounds better than a C18-silica column. Note in Figure 14, a<br />
tryptic digest <strong>of</strong> casein, the longer retention <strong>of</strong> amino acids arginine<br />
and phenylalanine and the overall different elution pattern provided<br />
by the PGC column compared to the C18-silica column.<br />
<strong>Guide</strong>lines for Choosing an RP-<strong>HPLC</strong> Column<br />
This list serves as a starting point for choosing a RP-<strong>HPLC</strong> phase based<br />
on the properties <strong>of</strong> the biomolecule. However, when developing a<br />
method one should try many different particle chemistries in order<br />
to optimize the separation.<br />
Proteins C4 on wide-pore (>300Å) silica<br />
Hydrophobic peptides C4-silica, C8-silica, cyano-silica<br />
Hydrophilic peptides C18-silica, Hypercarb<br />
Peptides C18-silica, phenyl-silica<br />
Amino acids (derivatized) C18-silica<br />
Amino acids (underivatized) Hypercarb PGC<br />
Nucleosides, nucleotides Hypercarb PGC<br />
Small molecules C18-silica, cyano-silica, Hypercarb PGC<br />
Carbohydrates, oligosaccharides Hypercarb PGC<br />
Lipids C18-silica with 100% organic mobile phase<br />
Table 4: Phase choice based on sample type<br />
Mobile Phases for RP-<strong>HPLC</strong><br />
HyPURITY C18, 5 µm, 50 x 2.1 mm<br />
Part No.: 22105-052130<br />
Hypercarb, 5 µm, 50 x 2.1 mm<br />
Part No.: 35005-052130<br />
Eluent: A: H 2 O + 0.05% TFA<br />
B: ACN: H 2 O (9:1 )+ 0.035% TFA<br />
Gradient: Time %B<br />
0 0<br />
3 0<br />
23 90<br />
Flow: 0.7 mL/min<br />
Temp: 25°C<br />
Detection: UV @195nm and + ESI<br />
Most biomolecules exist naturally in highly aqueous environments<br />
stabilized with ions and other water-soluble compounds that help<br />
maintain their biological activity. The ability to use aqueous mobile<br />
phases that contain salts and buffers is a main reason why liquid<br />
chromatography techniques, including reversed phase, ion exchange,<br />
and size exclusion, are so ubiquitous in biomolecule analysis. Rarely<br />
does one encounter normal phase liquid chromatography or gas<br />
chromatography where water and salts are not compatible with<br />
instruments and high organic mobile phases.<br />
RP-<strong>HPLC</strong> mobile phases for protein and peptide separations are<br />
usually gradients <strong>of</strong> an organic modifier (acetonitrile or methanol)<br />
in water, with an ionic modifier that controls the pH and ionization<br />
state or acts as an ion pair agent. Trifluoroacetic acid (TFA) is a<br />
common ionic additive because it solubilizes most proteins by forming<br />
ion pairs; however it has limitations for LC/MS. For proteins and<br />
peptides, gradient elution is preferred because proteins elute over<br />
a wide percentage <strong>of</strong> organic modifier. Isocratic elution results in<br />
very broad peaks. For small molecule separations, gradient elution<br />
is used when the compounds differ widely in hydrophobicity. A list<br />
<strong>of</strong> commonly used organic modifiers and ionic additives is found<br />
in the Appendices. The rationale behind the major mobile phase<br />
variables is described in the paragraphs below.