HPLC Analysis of Biomolecules Technical Guide - Interscience
HPLC Analysis of Biomolecules Technical Guide - Interscience
HPLC Analysis of Biomolecules Technical Guide - Interscience
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
16<br />
<strong>HPLC</strong> <strong>Analysis</strong> <strong>of</strong> <strong>Biomolecules</strong><br />
RP-LC/MS <strong>of</strong> <strong>Biomolecules</strong><br />
<strong>HPLC</strong> with mass spectrometric detection (LC/MS) is fast becoming<br />
the method <strong>of</strong> choice for many biomolecule analyses. A full discussion<br />
<strong>of</strong> LC/MS, the instruments, ionization techniques, and interfaces, is<br />
beyond the scope <strong>of</strong> this guide. However, the focus <strong>of</strong> this guide, the<br />
<strong>HPLC</strong> column, is more than a MS filter. The chemistry <strong>of</strong> the particle<br />
and the configuration <strong>of</strong> the hardware significantly affect the relevance<br />
and reliability <strong>of</strong> the data that is obtained from the LC/MS instrument.<br />
In addition to the column, the mobile phase conditions have a pr<strong>of</strong>ound<br />
influence on the quality <strong>of</strong> LC/MS data. Mass spectrometric detection<br />
has high compound specificity and selectivity which lessens the requirements<br />
for high selectivity separations; however, it brings limitations<br />
to the method development process. For instance, the choice <strong>of</strong> mobile<br />
phase composition is restricted since mobile phase needs to balance<br />
the chromatographic requirements with the ionization efficiency.<br />
Figure 23: LC/MS method selection guidelines<br />
Column Selection for RP-LC/MS: Column ID<br />
Usually the aim <strong>of</strong> an LC/MS experiment is to maximize sensitivity;<br />
to be able to see compounds that exist at very low concentration in<br />
the sample or that may have adequate concentration but are not<br />
easily detected (ionized) by the MS. When samples are limited, as<br />
in most proteomics applications, it is essential that maximum LC/MS<br />
information is obtained from the smallest injection volume. To<br />
increase sensitivity, the concentration <strong>of</strong> the compound entering the<br />
ionization source must be maximized. The more concentrated the<br />
analyte, the stronger the signal and the higher the sensitivity. Using<br />
concentration to increase sensitivity is especially important for<br />
compounds that are not readily ionizable. The ID (internal diameter) <strong>of</strong><br />
the <strong>HPLC</strong> column affects the concentration <strong>of</strong> the sample. Samples<br />
are diluted in proportion to the cross-sectional area <strong>of</strong> the column.<br />
Therefore, smaller ID columns yield less dilution. Sensitive LC/MS<br />
analyses are typically run on very small ID columns or capillaries.<br />
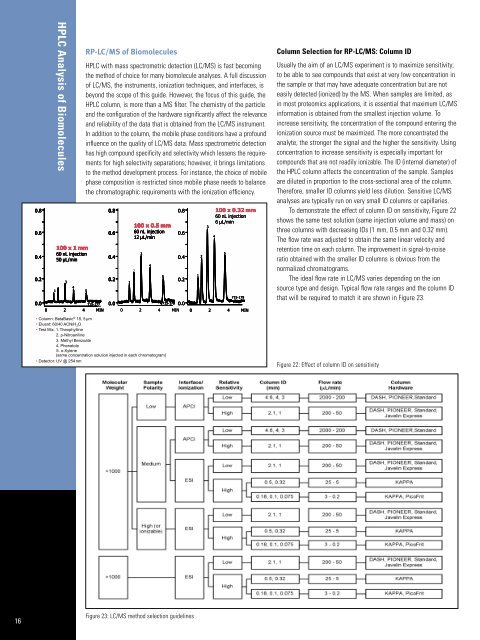
To demonstrate the effect <strong>of</strong> column ID on sensitivity, Figure 22<br />
shows the same test solution (same injection volume and mass) on<br />
three columns with decreasing IDs (1 mm, 0.5 mm and 0.32 mm).<br />
The flow rate was adjusted to obtain the same linear velocity and<br />
retention time on each column. The improvement in signal-to-noise<br />
ratio obtained with the smaller ID columns is obvious from the<br />
normalized chromatograms.<br />
The ideal flow rate in LC/MS varies depending on the ion<br />
source type and design. Typical flow rate ranges and the column ID<br />
that will be required to match it are shown in Figure 23.<br />
Figure 22: Effect <strong>of</strong> column ID on sensitivity