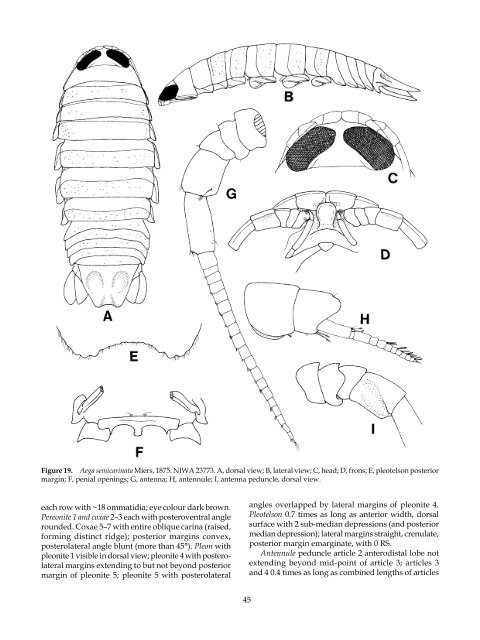
mediately recognisable by the highly textured dorsal surface, heavily ‘spined’ pleotelson and uropods, the subtruncate pleotelson posterior margin with a welldefined median point, the pleotelson with a prominent longitudinal ridge, the antennule peduncle articles being flattened and expanded, the huge eyes which appear to be medially united and the characteristic shape <strong>of</strong> the frontal lamina. Juvenile specimens are not as nodular, and can be identified by the characteristic shape <strong>of</strong> the frontal lamina, and the shape, ornamentation and setation <strong>of</strong> the pleotelson and uropodal rami. <strong>The</strong> ovigerous female is slightly wider in body shape, and the prominent robust setae are missing from the margins <strong>of</strong> the pleotelson and uropods; the frontal lamina is the same as in the male, and the appendages are otherwise similar. Although the distribution is vast, there is no doubt that all the material identified here is the one species, and furthermore such a distribution is not unique (e.g. Metacirolana caeca (Hansen, 9 6), see Svavarsson & Bruce 2000), possible influenced by the Great Global Conveyer currents (e.g. see Manighetti 200 ). A rather similar Aega sp. collected east <strong>of</strong> Heron Island, Queensland (NTM, unregistered, see p. 244) can be distinguished by the far smaller antennule peduncle articles 1 and 2, flat frontal lamina, more anteriorly rounded head, more and larger robust setae on pereopods –3, lack <strong>of</strong> large robust setae on the pleotelson and uropods, more rounded uropodal exopod, and by the different nature <strong>of</strong> the robust pitting on the pereon and pleotelson. prey: <strong>The</strong>re are no recent prey identifications. Gadus morrhua and Scymnum microcephalum (= Scymno microcephalo), Somniosus microcephalus (= Greenland shark, Dalatiidae) (Schioedte & Meinert 879b); Centrophorus squamosus (gulper shark, Centrophoridae) (Kussakin 979); Hyperoglyphe antarctica [bluenose and matiri (<strong>New</strong> <strong>Zealand</strong>) or Antarctic butterfish, Centro- lophidae]. distribution: North Atlantic, South Africa and southwestern Pacific. Localities: Schioedte and Meinert ( 879b) cite Iceland, Bergen, Floroe, Lodshagen and Farsun (all Norway), German Sea (= German Bight?) and Herne, Skagerak. South Africa (Barnard, 9 4); Bahamas (Treat 980); Hansen ( 9 6) cites the Færoe Islands and Jutland (Denmark) as regional records; Moray Firth and Shetland, Scotland (Norman 904). Present material is from the Chatham Rise, <strong>New</strong> <strong>Zealand</strong>, <strong>New</strong> Caledonia and from southeastern Australia <strong>of</strong>f the mid- and southern <strong>New</strong> South Wales coast. Previously recorded at depths <strong>of</strong> 460–730 m, present material at depths between 440 and 022 metres. 44 Aega semicarinata Miers, 875 (Figs 9–22) Æga semicarinata Miers, 1875: 115.– 1879: 201, pl. 11, figs. 1–1d.– Dollfus, 1891: 57, pl. 8, figs 2–2a.– Bouvier, 1911: 39, pl. 2, fig. 1. Aega semicarinata.– Barnard, 9 6: 06.– Nierstrasz, 93 : 83.– Hale, 937: 9.– Barnard, 940: 40 .– Stephensen, 1947: 23.– Menzies, 1962: 118, fig. 38A–C.– Moreira & Sadowsky, 1979: 109.– Kensley, 1978: 57, fig. 24I–J; 980b: 59; 200 : 227.– Kussakin & Vasina, 982: 264.– Branch, Griffiths, Kensley & Sieg, 1991: 26.– Bruce, Lew Ton & Poore, 2002: 62. Æga semicarinatus.– Stebbing, 920: 334. Aega bicavata Nordenstam, 1930, 547, fig. 11, Pl. 20, fig. 11. Aega (Aega) bicavata.– Brusca, 983: 0. Aega (Aega) semicarinata.– Brusca, 983: . type LoCaLity: Kerguelen, southern Indian Ocean (Miers 875). types: At the then British Museum <strong>of</strong> Natural History, London, according to Miers ( 875). Not listed by Ellis ( 98 ), though one unregistered specimen, labelled as ‘type’ is held at <strong>The</strong> Natural History Museum, London. <strong>The</strong> type information states: ‘outside label destroyed at Godstone’ and the only other data is ‘HMS Sylvia’. materiaL examined: Holotype(?), ♀ (non-ovig 27 mm), ‘outside label destroyed at Godstone’, HMS Sylvia (BMNH). <strong>New</strong> <strong>Zealand</strong> material: ♀ (74 mm ovig, previously dissected), I685, vicinity <strong>of</strong> Bounty Plateau, 48° 9.50– 7.20’S, 79°29.50–40’W, 6 March 979, dredged, 722 m and ♂ (35 mm), stn Z3, labels in tube: “Z3, 40F, A, 8/63”; “?John Graham, Oamaru, “♂ <strong>of</strong> giant I685”; therefore presumably same data as I685 (NIWA 2377 ♀, 23772 ♂). ♂ (38 mm), Chatham Rise, 42°43.95’S, 79°53.9 ’W, 8 April 200 , 076–990 m, coll. S. O’Shea on RV Tangaroa (NIWA 23773). ♂ (3 mm), Chatham Rise, 43°49.605’S, 78°29.284’E, 6 October 200 , 454 m, Agassiz trawl, coll. RV Tangaroa (NIWA 23774). ♂ (3 mm), North Otago, 27.5 m, 962, coll. John Graham (NMNZ Cr. 20 6). ♀ (~4–5 cm, ovig, broken, two pieces), 44°4 .35’S, 72°34.0’E, 390–360 m, RV James Cook (NMNZ Cr.4969). Additional material: ♀ (63 mm, non-ovig), Juan Fernandez Islands, Chile, 920, A 463 (LACM 20- 2. ). 2♀ (44, 38 mm), <strong>of</strong>f Table Bay, South Africa (BMNH 93 . . 0. 8-20, part). ♂ (39 mm), Chatham Rise, 42.7 60–7 08°S, 80.0390–0297°E, 28 May 2006, 935– 2 0 m, coll. RV Tangaroa (NIWA 25658). desCription: Body 2.6 times as long as greatest width, dorsal surfaces punctate, widest at pereonite 6, lateral margins subparallel. Rostral point projecting anteriorly, not ventrally folded. Eyes large, not medially united, separated by about 9% width <strong>of</strong> head; each eye made up <strong>of</strong> ~36 transverse rows <strong>of</strong> ommatidia,
Figure 19. Aega semicarinata Miers, 875. NIWA 23773. A, dorsal view; B, lateral view; C, head; D, frons; E, pleotelson posterior margin; F, penial openings; G, antenna; H, antennule; I, antenna peduncle, dorsal view. each row with ~ 8 ommatidia; eye colour dark brown. Pereonite 1 and coxae 2–3 each with posteroventral angle rounded. Coxae 5–7 with entire oblique carina (raised, forming distinct ridge); posterior margins convex, posterolateral angle blunt (more than 45°). Pleon with pleonite visible in dorsal view; pleonite 4 with posterolateral margins extending to but not beyond posterior margin <strong>of</strong> pleonite 5; pleonite 5 with posterolateral 45 angles overlapped by lateral margins <strong>of</strong> pleonite 4. Pleotelson 0.7 times as long as anterior width, dorsal surface with 2 sub-median depressions (and posterior median depression); lateral margins straight, crenulate, posterior margin emarginate, with 0 RS. Antennule peduncle article 2 anterodistal lobe not extending beyond mid-point <strong>of</strong> article 3; articles 3 and 4 0.4 times as long as combined lengths <strong>of</strong> articles
- Page 1 and 2: NATIONAL INSTITUTE OF WATER AND ATM
- Page 3 and 4: NATIONAL INSTITUTE OF WATER AND ATM
- Page 5 and 6: cONtENts ABSTRACT .................
- Page 7 and 8: The isopod fauna of New Zealand has
- Page 9 and 10: mAtErIAl ExAmINEd Material examined
- Page 11 and 12: end on preservation, rendering appa
- Page 13 and 14: Figure 3. Terms used in description
- Page 15 and 16: y the fact that while some species
- Page 17 and 18: Figure 4. Clades in Aega: Strict co
- Page 19 and 20: parallel lateral margins (Ch 28.2)
- Page 21 and 22: CharaCter List for AegA body charac
- Page 23 and 24: Traditionally the Aegidae have been
- Page 25 and 26: subOrdEr cymOthOIdA wägElE, 1989 B
- Page 27 and 28: 3. Body dorsally compressed; fronta
- Page 29 and 30: Figure 8. Aega falklandica Kussakin
- Page 31 and 32: Figure 10. Aega falklandica Kussaki
- Page 33 and 34: Figure 11. Aega falklandica Kussaki
- Page 35 and 36: Figure 12. Aega komai Bruce, 996. N
- Page 37 and 38: size: Female 26 mm, two males 6.5 a
- Page 39 and 40: Figure 15. Aega monophthalma Johnst
- Page 41 and 42: Figure 16. Aega monophthalma Johnst
- Page 43: Figure 18. Aega monophthalma Johnst
- Page 47 and 48: Figure 21. Aega semicarinata Miers,
- Page 49 and 50: pod 2.2 times as long as wide, dist
- Page 51 and 52: Figure 23. Aega stevelowei sp. nov.
- Page 53 and 54: Figure 25. Aega stevelowei sp. nov.
- Page 55 and 56: 4 teeth), lateral margin straight,
- Page 57 and 58: Figure 28. Aega urotoma Barnard, 9
- Page 59 and 60: Figure 30. Aega urotoma Barnard, 9
- Page 61 and 62: Aega whanui sp. nov. (Figs 3 -33) m
- Page 63 and 64: Figure 32. Aega whanui sp. nov. Hol
- Page 65 and 66: distribution: Lord Howe Rise to the
- Page 67 and 68: Figure 34. Aegapheles alazon (Bruce
- Page 69 and 70: Figure 35. Aegapheles birubi (Bruce
- Page 71 and 72: Figure 36. Aegapheles copidis sp. n
- Page 73 and 74: Variation: The left and right uropo
- Page 75 and 76: endopod. Uropod rami with apices br
- Page 77 and 78: Figure 40. Aegapheles mahana sp. no
- Page 79 and 80: teral margin not digitate; endopods
- Page 81 and 82: tenna peduncle article 2 inferior s
- Page 83 and 84: as ischium, .8 times as long as wid
- Page 85 and 86: Aegiochus beri (Bruce, 983), comb.
- Page 87 and 88: Figure 45. Aegiochus beri (Bruce, 1
- Page 89 and 90: Figure 46. Aegiochus bertrandi sp.
- Page 91 and 92: Figure 48. Aegiochus bertrandi sp.
- Page 93 and 94: Aegiochus coroo (Bruce, 983), comb.
- Page 95 and 96:
Figure 50. Aegiochus coroo (Bruce,
- Page 97 and 98:
size: Males 8.5- 3.0 mm (mean = .4,
- Page 99 and 100:
Figure 53. Aegiochus gordoni sp. no
- Page 101 and 102:
Figure 55. Aegiochus gordoni sp. no
- Page 103 and 104:
New figures of the holotypes of Aeg
- Page 105 and 106:
Figure 57. Aegiochus insomnis sp. n
- Page 107 and 108:
Figure 59. Aegiochus insomnis sp. n
- Page 109 and 110:
distribution: Known only from the P
- Page 111 and 112:
Figure 62. Aegiochus kakai sp. nov.
- Page 113 and 114:
Figure 64. Aegiochus kakai sp. nov.
- Page 115 and 116:
Figure 65. Aegiochus kanohi sp. nov
- Page 117 and 118:
Figure 67. Aegiochus kanohi sp. nov
- Page 119 and 120:
of ischium-carpus with short RS. Pe
- Page 121 and 122:
Figure 69. Aegiochus laevis (Studer
- Page 123 and 124:
southwestern or western Pacific; re
- Page 125 and 126:
Figure 72. Aegiochus nohinohi sp. n
- Page 127 and 128:
Figure 74. Aegiochus nohinohi sp. n
- Page 129 and 130:
33°47-44’S, 5 °59’- 52°0 ’
- Page 131 and 132:
Figure 76. Aegiochus piihuka sp. no
- Page 133 and 134:
Figure 78. Aegiochus piihuka sp. no
- Page 135 and 136:
Figure 79. Aegiochus pushkini (Kuss
- Page 137 and 138:
Figure 81. Aegiochus pushkini (Kuss
- Page 139 and 140:
Figure 83. Aegiochus pushkini (Kuss
- Page 141 and 142:
Aegiochus riwha sp. nov. (Figs 84-8
- Page 143 and 144:
Figure 85. Aegiochus riwha sp. nov.
- Page 145 and 146:
Figure 87. Aegiochus riwha sp. nov.
- Page 147 and 148:
Figure 88. Aegiochus tara sp. nov.
- Page 149 and 150:
Figure 90. Aegiochus tara sp. nov.
- Page 151 and 152:
uropods, and the elongate and flat
- Page 153 and 154:
Figure 92. Epulaega derkoma sp. nov
- Page 155 and 156:
Figure 94. Epulaega derkoma sp. nov
- Page 157 and 158:
Figure 96. Epulaega fracta (Hale, 9
- Page 159 and 160:
Figure 98. Epulaega fracta (Hale, 9
- Page 161 and 162:
as long as wide, inferior margin wi
- Page 163 and 164:
Rocinela bonita sp. nov. (Figs 00-
- Page 165 and 166:
Figure 101. Rocinela bonita sp. nov
- Page 167 and 168:
Figure 103. Rocinela bonita sp. nov
- Page 169 and 170:
3, more strongly produced uropod pe
- Page 171 and 172:
Figure 106. Rocinela garricki Hurle
- Page 173 and 174:
Figure 108. Rocinela garricki Hurle
- Page 175 and 176:
Figure 110. Rocinela leptopus sp. n
- Page 177 and 178:
Figure 112. Rocinela leptopus sp. n
- Page 179 and 180:
Figure 114. Rocinela pakari sp. nov
- Page 181 and 182:
Figure 116. Rocinela pakari sp. nov
- Page 183 and 184:
Figure 118. Rocinela pakari sp. nov
- Page 185 and 186:
Figure 119. Rocinela resima sp. nov
- Page 187 and 188:
Figure 121. Rocinela resima sp. nov
- Page 189 and 190:
Variation: Based on the four type s
- Page 191 and 192:
Figure 124. Rocinela runga sp. nov.
- Page 193 and 194:
3, and uropodal rami that are relat
- Page 195 and 196:
Figure 127. Rocinela satagia sp. no
- Page 197 and 198:
Figure 129. Rocinela satagia sp. no
- Page 199 and 200:
genus Aega (e.g. A. falklandica, A.
- Page 201 and 202:
Figure 131. Syscenus kapoo sp. nov.
- Page 203 and 204:
Figure 132. Syscenus latus Richards
- Page 205 and 206:
Figure 134. Syscenus latus Richards
- Page 207 and 208:
Figure 135. Syscenus moana Bruce, 2
- Page 209 and 210:
Figure 136. Syscenus springthorpei
- Page 211 and 212:
evision of the species based on goo
- Page 213 and 214:
a nomen nudum in a footnote, by Kr
- Page 215 and 216:
Rocinela insularis Schioedte & Mein
- Page 217 and 218:
Figure 137. Aegiochus antarctica (H
- Page 219 and 220:
flattened antennule peduncle articl
- Page 221 and 222:
0 acute RS (2 simple setae); carpus
- Page 223 and 224:
AMAR, R.; ROMAN, M.-L. 974: Invert
- Page 225 and 226:
DOLLFUS, A. 89 : Crustacés Isopode
- Page 227 and 228:
KUSSAKIN, O.G.; VASINA, G.S. 982: A
- Page 229 and 230:
ROHDE, K. 2005: Marine Parasitology
- Page 231 and 232:
AppENdIx 1. INvAlId AEgId NAmEs Inc
- Page 233 and 234:
Figure 139. Aega angustata Whiteleg
- Page 235 and 236:
of the uropodal exopod lacking serr
- Page 237 and 238:
present this record can only be con
- Page 239 and 240:
Figure 143. Aegiochus plebeia (Hans
- Page 241 and 242:
posterior margin (rather than conve
- Page 243 and 244:
H Figure 146. Syscenus intermedius
- Page 245 and 246:
Aegiochus aff. tara: ♀ (ovig. 27
- Page 247 and 248:
magnifica 1122112411 2213124211 131
- Page 249 and 250:
Aega, 7, 0, 2, 3, 4, 5, 27, 98 Aega
- Page 251 and 252:
Greenland, 2 2, 2 3, 2 5 Greenland