smallpox vaccine and vaccination in the intensified ... - libdoc.who.int
smallpox vaccine and vaccination in the intensified ... - libdoc.who.int
smallpox vaccine and vaccination in the intensified ... - libdoc.who.int
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
11. VACCINATION IN THE INTENSIFIED PROGRAMME<br />
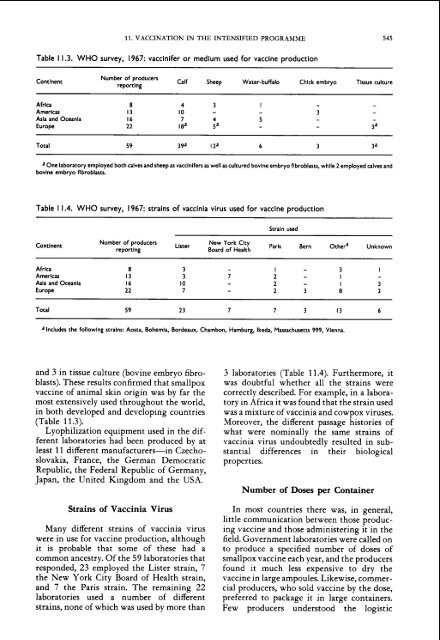
Table 1 1.3. WHO survey, 1967: vacc<strong>in</strong>ifer or medium used for <strong>vacc<strong>in</strong>e</strong> production<br />
Contlnent<br />
Number of producers<br />
report<strong>in</strong>g<br />
Calf Sheep Water-buffalo Chlck embryo Tissue culture<br />
Africa 8 4 3 I - -<br />
Americas 13 10 - - 3 -<br />
Asla <strong>and</strong> Oceanla 16 7 4 5 - -<br />
Europe 22 18' P - - 3a<br />
Total 59 39a IZa 6 3 V<br />
a One laboratory employed both calves <strong>and</strong> sheep as vacc<strong>in</strong>ifers as well as cultured bovlne embryo flbroblasts, whlle 2 employed calves <strong>and</strong><br />
bov<strong>in</strong>e embryo flbroblasts.<br />
Table 1 1.4. WHO survey, 1967: stra<strong>in</strong>s of vacc<strong>in</strong>ia virus used for <strong>vacc<strong>in</strong>e</strong> production<br />
Contlnent<br />
Africa<br />
Americas<br />
Asia <strong>and</strong> Oceania<br />
Europe<br />
Stra<strong>in</strong> used<br />
Number of producers Lister New York Clty<br />
Board of Health Paris Bern O<strong>the</strong>ra Unknown<br />
report<strong>in</strong>g<br />
Total 59 23 7 7 3 13 6<br />
a Includes <strong>the</strong> follow<strong>in</strong>g stra<strong>in</strong>s: Aorta, Bohemia, Bordeaux, Chambon, Hamburg, Ikeda, Massachusetts 999, Vienna.<br />
<strong>and</strong> 3 <strong>in</strong> tissue culture (bov<strong>in</strong>e embryo fibro-<br />
blasts). These results confirmed that <strong>smallpox</strong><br />
<strong>vacc<strong>in</strong>e</strong> of animal sk<strong>in</strong> orig<strong>in</strong> was by far <strong>the</strong><br />
most extensively used throughout <strong>the</strong> world,<br />
<strong>in</strong> both developed <strong>and</strong> develop<strong>in</strong>g countries<br />
(Table 1 1.3).<br />
Lyophilization equipment used <strong>in</strong> <strong>the</strong> dif-<br />
ferent laboratories had been produced by at<br />
least 11 different manufacturers-<strong>in</strong> Czecho-<br />
slovakia, France, <strong>the</strong> German Democratic<br />
Republic, <strong>the</strong> Federal Republic of Germany,<br />
Japan, <strong>the</strong> United K<strong>in</strong>gdom <strong>and</strong> <strong>the</strong> USA.<br />
Stra<strong>in</strong>s of Vacc<strong>in</strong>ia Virus<br />
Many different stra<strong>in</strong>s of vacc<strong>in</strong>ia virus<br />
were <strong>in</strong> use for <strong>vacc<strong>in</strong>e</strong> production, although<br />
it is probable that some of <strong>the</strong>se had a<br />
common ancestry. Of <strong>the</strong> 59 laboratories that<br />
responded, 23 employed <strong>the</strong> Lister stra<strong>in</strong>, 7<br />
<strong>the</strong> New York City Board of Health stra<strong>in</strong>,<br />
<strong>and</strong> 7 <strong>the</strong> Paris stra<strong>in</strong>. The rema<strong>in</strong><strong>in</strong>g 22<br />
laboratories used a number of different<br />
stra<strong>in</strong>s, none of which was used by more than<br />
3 laboratories (Table 11.4). Fur<strong>the</strong>rmore, it<br />
was doubtful whe<strong>the</strong>r all <strong>the</strong> stra<strong>in</strong>s were<br />
correctly described. For example, <strong>in</strong> a labora-<br />
tory <strong>in</strong> Africa it was found that <strong>the</strong> stra<strong>in</strong> used<br />
was a mixture of vacc<strong>in</strong>ia <strong>and</strong> cowpox viruses.<br />
Moreover, <strong>the</strong> different passage histories of<br />
what were nom<strong>in</strong>ally <strong>the</strong> same stra<strong>in</strong>s of<br />
vacc<strong>in</strong>ia virus undoubtedly resulted <strong>in</strong> sub-<br />
stantial differences <strong>in</strong> <strong>the</strong>ir biological<br />
properties.<br />
Number of Doses per Conta<strong>in</strong>er<br />
In most countries <strong>the</strong>re was, <strong>in</strong> general,<br />
little communication between those produc-<br />
<strong>in</strong>g <strong>vacc<strong>in</strong>e</strong> <strong>and</strong> those adm<strong>in</strong>ister<strong>in</strong>g it <strong>in</strong> <strong>the</strong><br />
field. Government laboratories were called on<br />
to produce a specified number of doses of<br />
<strong>smallpox</strong> <strong>vacc<strong>in</strong>e</strong> each year, <strong>and</strong> <strong>the</strong> producers<br />
found it much less expensive to dry <strong>the</strong><br />
<strong>vacc<strong>in</strong>e</strong> <strong>in</strong> large ampoules. Likewise, commer-<br />
cial producers, <strong>who</strong> sold <strong>vacc<strong>in</strong>e</strong> by <strong>the</strong> dose,<br />
preferred to package it <strong>in</strong> large conta<strong>in</strong>ers.<br />
Few producers understood <strong>the</strong> logistic