The primate cranial base: ontogeny, function and - Harvard University
The primate cranial base: ontogeny, function and - Harvard University
The primate cranial base: ontogeny, function and - Harvard University
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
130 YEARBOOK OF PHYSICAL ANTHROPOLOGY [Vol. 43, 2000<br />
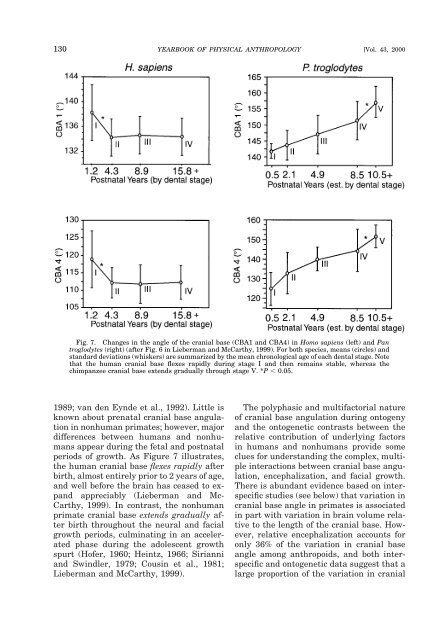
Fig. 7. Changes in the angle of the <strong>cranial</strong> <strong>base</strong> (CBA1 <strong>and</strong> CBA4) in Homo sapiens (left) <strong>and</strong> Pan<br />
troglodytes (right) (after Fig. 6 in Lieberman <strong>and</strong> McCarthy, 1999). For both species, means (circles) <strong>and</strong><br />
st<strong>and</strong>ard deviations (whiskers) are summarized by the mean chronological age of each dental stage. Note<br />
that the human <strong>cranial</strong> <strong>base</strong> flexes rapidly during stage I <strong>and</strong> then remains stable, whereas the<br />
chimpanzee <strong>cranial</strong> <strong>base</strong> extends gradually through stage V. *P 0.05.<br />
1989; van den Eynde et al., 1992). Little is<br />
known about prenatal <strong>cranial</strong> <strong>base</strong> angulation<br />
in nonhuman <strong>primate</strong>s; however, major<br />
differences between humans <strong>and</strong> nonhumans<br />
appear during the fetal <strong>and</strong> postnatal<br />
periods of growth. As Figure 7 illustrates,<br />
the human <strong>cranial</strong> <strong>base</strong> flexes rapidly after<br />
birth, almost entirely prior to 2 years of age,<br />
<strong>and</strong> well before the brain has ceased to exp<strong>and</strong><br />
appreciably (Lieberman <strong>and</strong> Mc-<br />
Carthy, 1999). In contrast, the nonhuman<br />
<strong>primate</strong> <strong>cranial</strong> <strong>base</strong> extends gradually after<br />
birth throughout the neural <strong>and</strong> facial<br />
growth periods, culminating in an accelerated<br />
phase during the adolescent growth<br />
spurt (Hofer, 1960; Heintz, 1966; Sirianni<br />
<strong>and</strong> Swindler, 1979; Cousin et al., 1981;<br />
Lieberman <strong>and</strong> McCarthy, 1999).<br />
<strong>The</strong> polyphasic <strong>and</strong> multifactorial nature<br />
of <strong>cranial</strong> <strong>base</strong> angulation during <strong>ontogeny</strong><br />
<strong>and</strong> the ontogenetic contrasts between the<br />
relative contribution of underlying factors<br />
in humans <strong>and</strong> nonhumans provide some<br />
clues for underst<strong>and</strong>ing the complex, multiple<br />
interactions between <strong>cranial</strong> <strong>base</strong> angulation,<br />
encephalization, <strong>and</strong> facial growth.<br />
<strong>The</strong>re is abundant evidence <strong>base</strong>d on interspecific<br />
studies (see below) that variation in<br />
<strong>cranial</strong> <strong>base</strong> angle in <strong>primate</strong>s is associated<br />
in part with variation in brain volume relative<br />
to the length of the <strong>cranial</strong> <strong>base</strong>. However,<br />
relative encephalization accounts for<br />
only 36% of the variation in <strong>cranial</strong> <strong>base</strong><br />
angle among anthropoids, <strong>and</strong> both interspecific<br />
<strong>and</strong> ontogenetic data suggest that a<br />
large proportion of the variation in <strong>cranial</strong>