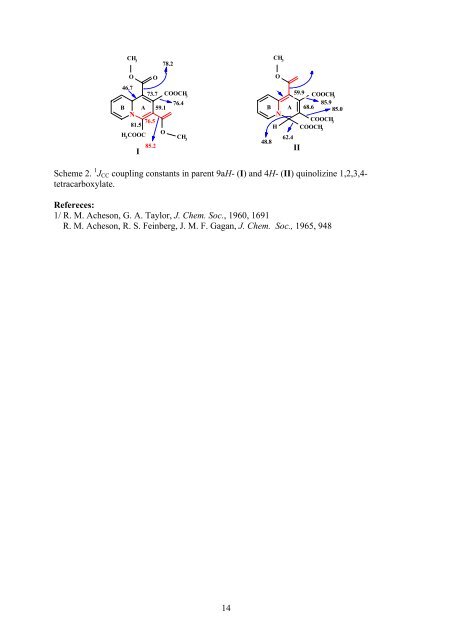
CH 3 78.2 O O 46.7 73.7 COOCH 3 B 76.4 A 59.1 N O 81.5 76.5 H 3 COOC O CH 3 85.2 I CH 3 O 70.1 B 48.8 N H O 88.7 59.9 COOCH 3 85.9 A 68.6 85.0 62.4 II COOCH 3 COOCH 3 Scheme 2. 1 J CC coupling constants in parent 9aH- (I) and 4H- (II) quinolizine 1,2,3,4- tetracarboxylate. Refereces: 1/ R. M. Acheson, G. A. Taylor, J. Chem. Soc., 1960, 1691 R. M. Acheson, R. S. Feinberg, J. M. F. Gagan, J. Chem. Soc., 1965, 948 14
NMR-BASED METHOD FOR THE LOCATION OF AMINO ACID MOIETIES IN BACTERIAL OLIGOSACCHARIDES Piotr Bernatowicz 1 , Andrzej Ejchart 1 , Gabriela Pastuch-Gawołek 2 , Wiesław Szeja 3 , Tomasz Lipiński 3 , and Andrzej Gamian 3 1 Laboratory of Biological NMR, IBB PAS, Warsaw, Poland 2 Department of Organic Chemistry, Biochemistry and Biotechnology, Silesian Technical University, Gliwice, Poland 3 Ludwik Hirszfeld Institute of Immunology and Experimental Therapy, PAS, Wroclaw, Poland Structural variations of the core oligosaccharides of several species of Gram-negative bacteria, comprising substitution of amino acid glycine to the molecular backbone, have been recently reported. The genetic control of this process is not yet understood and the rationale behind glycine incorporation into the core oligosaccharides remains unclear, but one could expect that it increases the ability of the bacteria to cause host diseases. Therefore, an analytical method to identify the type of amino acid and to locate the site of its substitution is of importance. The model monosaccharides, ester-linked with an amino acid, were selected for the elaboration of an NMR-based method: Gly→4GlcOMe, Gly→6GalOMe, and Ala→6GalOMe. 1 H/ 13 C chemical shift correlation via long range coupling constant, 3 J(C'-O- C-H), was exploited for the detection of the covalent linkage between the amino acid residue and the saccharide moiety. The corresponding cross-peak at 13 C chemical shift characteristic for ester carbonyl {δ(C')≈165-170 ppm}appears in a relatively empty spectral region. Its 1 H chemical shift allows one to identify the substitution site providing the spectral assignments of saccharide protons have been performed. At the C' chemical shift one can also observe crosspeak to H and H protons of the amino acid residue thus identifying its type. It was found that such the HMBC-based NMR method is robust, efficient and reliable. 15