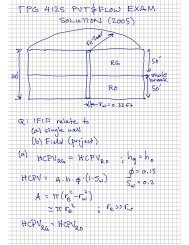
Water Production and Related Challenges - NTNU
Water Production and Related Challenges - NTNU
Water Production and Related Challenges - NTNU
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
<strong>Water</strong> <strong>Production</strong> <strong>and</strong><br />
<strong>Related</strong> <strong>Challenges</strong><br />
Flow Assurance, <strong>NTNU</strong> 4th March 2013<br />
Kari Ramstad, Specialist <strong>Production</strong> Technology<br />
Classification: Internal 2013-02-07
Content<br />
• <strong>Water</strong> production <strong>and</strong> management<br />
− Discharge vs. re-injection<br />
• Information from formation water<br />
− Sampling <strong>and</strong> analysis<br />
• <strong>Water</strong> related challenges<br />
− Scale, corrosion, naphtenates<br />
− Reservoir souring<br />
• Chemical managenent<br />
− Environment<br />
• Produced water (PW) monitoring<br />
2<br />
Classification: Internal 2013-02-07
<strong>Water</strong> production Norwegian Continental Shelf<br />
• High volumes of produced water<br />
− 83% discharge to sea<br />
− 17 % reinjection<br />
• Statoil’s 40 installations<br />
− 8-10 PWRI or disposal wells<br />
− <strong>Water</strong> management involves cost <strong>and</strong><br />
environmental considerations<br />
<strong>Water</strong><br />
Sources: Norwegian Petroleum Directorate, Klima og forurensningsdirektoratet (KLIF)<br />
3<br />
Classification: Internal 2013-02-07
Oil discharge in the North Sea<br />
• 5 % of total oil discharges in the North Sea<br />
come from petroleum industry<br />
• Produced water is the main source<br />
• OSPAR convention allows max 30 mg/l<br />
oil-in-water<br />
• Aim for zero harmful discharge <strong>and</strong><br />
continuous improvement of water cleaning<br />
technology<br />
Sources to oil discharges<br />
from petroleum activities<br />
Source: Norwegian Petroleum Directorate<br />
4<br />
Classification: Internal 2013-02-07
<strong>Water</strong> management options<br />
4<br />
3<br />
2<br />
Goal: Increased oil production combined with zero harmful discharge<br />
1<br />
5<br />
Classification: Internal 2013-02-07
Produced water treatment<br />
• <strong>Water</strong> treatment for injection,<br />
disposal or discharge<br />
− Reduce oil-in-water<br />
− Remove particles<br />
− Remove heavy metals<br />
− Reduce bacterial activity,<br />
toxic compounds, etc.<br />
• Reduce risk of<br />
− Injectivity losses<br />
− Mechanical failure<br />
− Environmental impact<br />
− Reservoir souring (H 2 S)<br />
Estimated injection rate [m3/d]<br />
Injection rate, m 3 /d<br />
2500<br />
o/w = 20 mg/l<br />
2000<br />
o/w = 50 mg/l<br />
o/w = 100 mg/l<br />
1500<br />
o/w = 200 mg/l<br />
1000<br />
500<br />
0<br />
0 5 10 15 20 25 30 35<br />
Particles, TSS [m g/l] mg/l<br />
Svorstøl, Tekna PW Management 2010<br />
6<br />
Classification: Internal 2013-02-07
Oil water separation<br />
• Reduce oil-in-water<br />
− Separator<br />
− Compact flotation unit<br />
• Gas <strong>and</strong> flocculant into PW<br />
− Electrostatic coalescer<br />
− Hydrocyclone<br />
− CTour<br />
• Condensate into PW<br />
− Filtration<br />
− Centrifuges<br />
7<br />
Classification: Internal 2013-02-07
Information from formation water<br />
• Source<br />
• Development<br />
A9<br />
• Migration<br />
• Reservoir underst<strong>and</strong>ing<br />
• Distance to non-discovered oil<br />
• Scaling potential<br />
• Corrosion potential<br />
A12<br />
A10<br />
A13<br />
A11<br />
A6AH<br />
A8<br />
A5AH<br />
A7<br />
A5<br />
A17<br />
A18<br />
A15<br />
A16<br />
A14<br />
• Hydrate potential<br />
• Souring<br />
=> Important input to reservoir management, field development <strong>and</strong> system design<br />
8<br />
Classification: Internal 2013-02-07
Chemical composition of formation rock<br />
Glaukonite - K 0.6<br />
Na 0.05<br />
Fe 3+ 1.3 Mg 0.4 Fe2+ 0.2 Al 0.3 Si 3.8 O 10 (OH) 2<br />
Siderite - FeCO 3<br />
Quartz - SiO 2<br />
9<br />
Classification: Internal 2013-02-07
<strong>Water</strong> compositions<br />
Ion (mg/l) Ormen Lange Oseberg Njord Gyda Utsira<br />
aquifer<br />
Seawater<br />
Na + 4428 12500 19000 65340 10100 11150<br />
K + 90 335 747 5640 262 420<br />
Ca 2+ 220 978 4050 30185 494 435<br />
Mg 2+ 31 135 392 2325 714 1410<br />
Ba 2+ 19 134 765 485 6.7 0.1<br />
Sr 2+ 12 157 891 1085 12.1 6.6<br />
Fe 0.6 0.1 23 - 5.7 0<br />
Cl - 6804 21900 41400 167400 18500 20310<br />
SO<br />
2-<br />
4 7.9 5 15 - 2 2800<br />
HCO<br />
-<br />
3 1008 633 230 76 1110 150<br />
Organic acids 640 120 360 - - -<br />
Salinity (TDS) 12650 36800 67513 272536 30100 36675<br />
10<br />
Classification: Internal 2013-02-07
<strong>Water</strong> sampling in exploration wells<br />
• Analyze the water <strong>and</strong> associated gas to<br />
reconstruct the formation water composition<br />
• DST – Drill Stem Test in the water zone<br />
− Large volume<br />
− Clean-up the well for low mud<br />
contamination<br />
• Wireline sampling<br />
− Pressurized 450 cc samples<br />
− Add tracers to the mud to determine<br />
degree of contamination<br />
11<br />
Classification: Internal 2013-02-07
Formation water analysis<br />
• Ions: Na + , K + , Ca 2+ , Mg 2+ , Ba 2+ , Sr 2+ , Fe, Cl - , SO 4<br />
2-<br />
,<br />
salinity, total alkalinity (HCO 3- ), trace elements<br />
• Organic acids, pH, particles<br />
• Density, resistivity (saturation log)<br />
• Isotopes δD, δ 18 O, 87 Sr/ 86 Sr (geochemistry)<br />
• Nutrients for Sulphate Reducing Bacteria (souring)<br />
• BTEX (Benzene, Toluene, Ethylbenzene, Xylenes)<br />
(environment, geochemistry)<br />
• Phenols (environment)<br />
• Heavy metals (environment)<br />
• Radioactive elements (environment)<br />
• Tracers (mud contamination)<br />
• Dissolved gas (recombine water composition)<br />
12<br />
Classification: Internal 2013-02-07
<strong>Water</strong> related challenges<br />
Mineral scale<br />
Typisk belegg i 21 systemet.<br />
Her i innløps heder på målestasjonen.<br />
Corrosion<br />
Korrosjon ved sveis nr. 7<br />
på ZPL210034 (innløp til<br />
målestasjon)<br />
Erosion<br />
Naphtenates<br />
S<strong>and</strong> & clay<br />
Emulsions<br />
13<br />
Classification: Internal 2013-02-07
Scale predictions<br />
Ca 2+ + CO 3<br />
2-<br />
= CaCO 3 (s)<br />
[Ca 2+ ][CO 3<br />
2-<br />
]<br />
Saturation Ratio SR CaCO3 = K sp (CaCO 3 )<br />
SR < 1<br />
CaCO 3 is undersaturated<br />
No precipitation<br />
SR = 1<br />
CaCO 3 is saturated<br />
Equilibrium (e.g. in the reservoir)<br />
SR > 1<br />
CaCO 3 is supersaturated<br />
CaCO 3 may precipitate depending<br />
on kinetics<br />
Thermodynamic equilibrium model<br />
14<br />
Classification: Internal 2013-02-07
CaCO 3 scaling potential along the well<br />
15<br />
Classification: Internal 2013-02-07
CaCO 3 scale in the tubing<br />
Caliper log<br />
• Compact scale from continuous pressure drop<br />
<strong>and</strong> CO 2 release<br />
m MD<br />
200<br />
400<br />
16<br />
Classification: Internal 2013-02-07
<strong>Water</strong> injection in heterogenous reservoirs<br />
<strong>Water</strong> injector<br />
Oil producer<br />
17<br />
Classification: Internal 2013-02-07
Mixing of formation water <strong>and</strong> seawater<br />
Ba 2+ + SO 4<br />
2-<br />
= BaSO 4 (s)<br />
• Low solubility gives sulphate scale<br />
• Solubility products<br />
− K sp (MgSO 4 ) = 10<br />
− K sp (CaSO 4 ) = 2.4 x 10 -5<br />
− K sp (SrSO 4 ) = 7.6 x 10 -7<br />
− K sp (BaSO 4 ) = 1.5 x 10 -9<br />
− K sp (RaSO 4 ) = 4 x 10 -11<br />
• Naturally Occurring Radioactive Materials<br />
18<br />
Classification: Internal 2013-02-07
Mixed scale<br />
CaCO 3 scale in upper tubing from<br />
pressure drop in produced water.<br />
Can be removed by acid (fast)<br />
m MD<br />
37.2<br />
500.0<br />
1000<br />
1000.0<br />
Caliper log<br />
5.0 nominal_OD 7.5<br />
5.0 drift_ID 7.5<br />
5.0 nominal_ID 7.5<br />
5.0 mean_diam 7.5<br />
5.0 min_diam 7.5<br />
5.0 max_diam 7.5<br />
No scale in the middle<br />
1500.0<br />
2000<br />
2000.0<br />
2500.0<br />
Radioactive BaSO 4 scale down hole<br />
from seawater breakthrough.<br />
Can be removed by EDTA/DTPA (slow)<br />
3000<br />
3000.0<br />
3641.1<br />
Cursor: 1053.082 m Av: 713<br />
19<br />
Classification: Internal 2013-02-07
Corrosion<br />
• Metal dissolution from reaction<br />
with surroundings<br />
• Anode oxydation, cathode reduction<br />
− Fe + 2 H + -> Fe 2+ + H 2 (g)<br />
• Corrosive environments<br />
− Condenced water (CO 2 )<br />
− Formation water/injection water (Cl - )<br />
− Drilling <strong>and</strong> completion fluids (Cl - )<br />
− CO 2<br />
Electrolyte<br />
C-steel<br />
Fe 2+ e -<br />
− H 2 S from reservoir souring<br />
− Organic <strong>and</strong> inorganic acids<br />
H 2<br />
H +<br />
e-<br />
− O 2 /Cl 2<br />
H +<br />
20<br />
Classification: Internal 2013-02-07
Naphtenates<br />
• Acids in the oil react with ions in the water<br />
− Occurs in separators<br />
− Na-naphtenate emulsion<br />
− Ca-naphtenate precipitate<br />
• Depending on<br />
− pH in the system<br />
− Molecular weight of the acids<br />
− Ion composition of the water<br />
<strong>Water</strong><br />
Ca<br />
COO 2+<br />
- -<br />
OOC<br />
COO -<br />
CO 2 H<br />
Na +<br />
Naphthoic acid<br />
CH 3 (CH 2 ) 9 CO 2 H<br />
Undecanoic acid<br />
Oil<br />
21<br />
Classification: Internal 2013-02-07
Reservoir souring<br />
• H 2 S forms in the reservoir by bacteria<br />
(SRB) <strong>and</strong> microorganisms<br />
• Difficult to simulate <strong>and</strong> predict<br />
− Bacteria, nutrients,<br />
sulphate (seawater injection),<br />
mineralogy, distance in reservoir<br />
• Corrosion, poor quality export oil/gas,<br />
HSE issues<br />
• H 2 S scavenger is costly <strong>and</strong> has<br />
environmental impact<br />
• PWRI (oil <strong>and</strong> organic acids) gives<br />
nutrients <strong>and</strong> more H 2 S<br />
− Use low sulphate injection water<br />
Oil organics<br />
Acetate + CO 2<br />
(SRB)<br />
SRP<br />
SO 4<br />
H 2 S<br />
Paul Evans, Chevron Energy Technology Company<br />
22<br />
Classification: Internal 2013-02-07
Chemical management<br />
23<br />
Classification: Internal 2013-02-07
Environmental classification of chemicals<br />
Category<br />
Black<br />
- not<br />
allowed<br />
Properties<br />
Prioritized list of White Paper No. 21 (2004-2005)<br />
OSPAR List of Chemicals for Priority Action (Strategy with regard to Hazardous Substances)<br />
Low biodegradability (BOD28
Chemical discharge<br />
• < 3 % of discharge is from petroleum industry<br />
• > 99 % are yellow or PLONOR chemicals<br />
• KLIF gives permission to use <strong>and</strong> discharge<br />
BaSO 4<br />
• 10 years’ research program<br />
− Improved models for monitoring biological responses<br />
− Long term environmental impacts by discharges to sea are moderate<br />
− Negative effects on fish etc. are limited by space <strong>and</strong> time<br />
− No indications that arctic species are more vulnerable than temperate<br />
− Variations depend on season, population <strong>and</strong> oceanographic conditions<br />
Sources: Klima og forurensningsdirektoratet (KLIF), European Journal of Mineralogy<br />
25<br />
Classification: Internal 2013-02-07
KLIF Recommendation 2010 (summary)<br />
• There should be no general requirement for re-injection of produced water on the major<br />
contributors Statfjord <strong>and</strong> Gullfaks. This is due to high cost, release of green house gases by<br />
injection, minor discharge of chemicals <strong>and</strong> no confirmed impact on the environment.<br />
• Troll B <strong>and</strong> C represent 40 % of the radioactive discharge offshore. Low increase in green<br />
house gas releases by re-injection. KLIF encourages the operator to consider re-injection of<br />
produced water, in co-operation with Statens Strålevern.<br />
• New fields should make transparent social-economical cost/benefit evaluations including<br />
holistic environmental evaluations to avoid discharge of produced water.<br />
• Lack of knowledge on long term environmental consequences <strong>and</strong> increasing produced water<br />
volumes in the years to come, require continuous focus on reduced discharges. KLIF will in<br />
dialogue with the operators consider:<br />
− Improvement of existing water cleaning equipment<br />
− New technologies for testing <strong>and</strong> implementation<br />
• Consider field specific requirements to reduce the discharge to sea<br />
Sources: Klima og forurensningsdirektoratet (KLIF)<br />
26<br />
Classification: Internal 2013-02-07
Produced water monitoring<br />
• Statoil: 4000 produced water samples per year<br />
• Monitor well performance <strong>and</strong> reservoir<br />
behaviour<br />
− Clean-up of completion fluids<br />
− Breakthrough of formation water<br />
− Breakthrough of injected (sea) water<br />
− Scale predictions<br />
− Corrosion control<br />
− Reservoir souring<br />
• Chemical management<br />
• Environment<br />
27<br />
Classification: Internal 2013-02-07
Ion tracking seawater break through<br />
500<br />
Produsert vann A-08<br />
100<br />
400<br />
80<br />
SW break through gives<br />
BaSO 4 scaling potential<br />
mg/l, bar<br />
300<br />
200<br />
100<br />
60<br />
40<br />
20<br />
%, ppm<br />
WC %<br />
Ba mg/l<br />
Sr mg/l<br />
SW %<br />
0<br />
12-nov-94<br />
11-apr-95<br />
08-sep-95<br />
05-feb-96<br />
04-jul-96<br />
01-des-96<br />
30-apr-97<br />
27-sep-97<br />
24-feb-98<br />
24-jul-98<br />
21-des-98<br />
20-mai-99<br />
17-okt-99<br />
15-mar-00<br />
12-aug-00<br />
09-jan-01<br />
08-jun-01<br />
05-nov-01<br />
Date<br />
0<br />
Ba, mg/l<br />
Sr, mg/l<br />
SW, % (Mg)<br />
H2S, ppm<br />
WC, %<br />
28<br />
Classification: Internal 2013-02-07
Ion ratios <strong>and</strong> pressure response<br />
• Break through of injection water monitored by water composition<br />
<strong>and</strong> bottom hole pressure<br />
Ba/Mg A-39<br />
A-39 og A-21(WI)<br />
1,20<br />
130.0<br />
5 000<br />
Ba/Mg<br />
1,00<br />
0,80<br />
0,60<br />
0,40<br />
0,20<br />
0,00<br />
9.1.2001<br />
19.4.2001<br />
28.7.2001<br />
5.11.2001<br />
13.2.2002<br />
24.5.2002<br />
Dato<br />
1.9.2002<br />
10.12.2002<br />
20.3.2003<br />
28.6.2003<br />
6.10.2003<br />
bar<br />
120.0<br />
110.0<br />
4 000<br />
100.0<br />
3 000<br />
90.0<br />
80.0<br />
2 000<br />
70.0<br />
1 000<br />
60.0<br />
50.0<br />
0<br />
01-okt-00<br />
09-jan-01<br />
19-apr-01<br />
28-jul-01<br />
05-nov-01<br />
13-feb-02<br />
24-mai-02<br />
01-sep-02<br />
10-des-02<br />
20-mar-03<br />
28-jun-03<br />
06-okt-03<br />
14-jan-04<br />
Dato<br />
Sm3/d<br />
BHP<br />
QWi<br />
29<br />
Classification: Internal 2013-02-07
Reservoir underst<strong>and</strong>ing<br />
• Well B-35 perforation<br />
− Oseberg/Rannoch/Etive 1993<br />
− Tarbert 1997<br />
• <strong>Water</strong> production 1998<br />
− Assumed Tarbert water<br />
• <strong>Water</strong> composition showed<br />
− B-35 produces mainly from<br />
Oseberg/Rannoch/Etive<br />
• Data implemented into the<br />
reservoir model<br />
Na/Sr<br />
105<br />
100<br />
95<br />
90<br />
85<br />
80<br />
Alfa Sør<br />
75<br />
70<br />
10 15 20 25<br />
Na/Ca<br />
B-14 (Tarbert AM)<br />
B-35 (ORENT AS)<br />
B-40 B (ORE AS)<br />
B-46 (ORE AS)<br />
30<br />
Classification: Internal 2013-02-07
<strong>Water</strong> production <strong>and</strong> related challenges<br />
• <strong>Water</strong> h<strong>and</strong>ling <strong>and</strong> treatment is essential<br />
− Cost <strong>and</strong> environment<br />
• Formation water gives input to<br />
− Reservoir underst<strong>and</strong>ing<br />
− Field development<br />
− System design<br />
• Produced water monitoring<br />
− Reservoir <strong>and</strong> well behaviour<br />
− Scale, corrosion <strong>and</strong> souring potentials<br />
− Chemical management<br />
− Environment<br />
Increased oil production combined with<br />
zero harmful discharge<br />
31<br />
Classification: Internal 2013-02-07
Abbreviations<br />
• AQW aquifer water<br />
• BaSO 4 barium sulphate<br />
• BOD28 percentage biodegradation after 28 days<br />
• BTEX benzene, toluene, ethylbenzene, xylenes<br />
• CaCO 3 calcium carbonate<br />
• CW connate water<br />
• δD delta deuterium vs. St<strong>and</strong>ard Mean Ocean <strong>Water</strong><br />
• δ 18 O delta oxygen-18 vs. St<strong>and</strong>ard Mean Ocean <strong>Water</strong><br />
• DST drill stem test<br />
• DTPA di-ethylene tri-amine penta-acetic acid<br />
• EC50 concentration affect growth rate of 50 % algae<br />
• EDTA ethylene di-amine tetra-acetic acid<br />
• HSE health safety environment<br />
• IW injection water<br />
• KLIF Klima og forurensningsdirektoratet<br />
• K sp solubility product<br />
• LC50 concentration where 50 % animals die<br />
• MEG mono-ethylene glycol<br />
• m MD meter measured depth<br />
• NORM naturally occurring radioactive materials<br />
• OSPAR Oslo-Paris convention<br />
• PLONOR pose little or no risk to the environment<br />
• Pow octanol–water partition coefficient<br />
• PPD pour point depressant<br />
• ppm parts per million<br />
• PVS polyvinyl sulphonate<br />
• PW produced water<br />
• PWRI produced water re-injection<br />
• SR saturation ratio<br />
• SRB sulphate reducing bacteria<br />
• SRP sulphate reducing prokaryotes<br />
• SW seawater<br />
• TDS total dissolved salts<br />
• WC water cut<br />
32<br />
2013-02-07
<strong>Water</strong> <strong>Production</strong> <strong>and</strong><br />
<strong>Related</strong> <strong>Challenges</strong><br />
<strong>NTNU</strong> 4th March 2013<br />
Kari Ramstad<br />
Specialist <strong>Production</strong> Technology<br />
karra@statoil.com<br />
Tel: +47 90249719<br />
www.statoil.com<br />
33<br />
Classification: Internal 2013-02-07