CHAPTER 4. THERMODYNAMICS: THE FIRST LAW
CHAPTER 4. THERMODYNAMICS: THE FIRST LAW
CHAPTER 4. THERMODYNAMICS: THE FIRST LAW
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
4-2<br />
how the change of state was carried out. Therefore, the changes in the values of the state functions<br />
are determined only by the initial and final states of the system. For example, consider the four states<br />
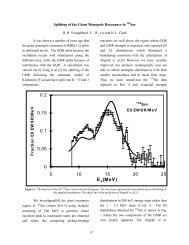
of a system composed of an ideal gas, as depicted in Fig. 4-1.<br />
path 1<br />
State 2<br />
P 2<br />
= 2 atm<br />
V 2<br />
= 22 L<br />
T 2<br />
= 546 K<br />
State 1<br />
P 1<br />
= 1 atm<br />
V 1<br />
= 22 L<br />
T 1<br />
= 273 K<br />
path 3<br />
State 4<br />
P 4<br />
= 2 atm<br />
V 4<br />
= 11 L<br />
T 4<br />
= 273 K<br />
path 2<br />
State 3<br />
P 3<br />
= 1 atm<br />
V 3<br />
= 11 L<br />
T 3<br />
= 136 K<br />
Figure 4-1. Transformation of a system from state 1 to state 4 via three different paths.<br />
Three different paths lead from state 1 to state <strong>4.</strong> However, the changes in all three of the state<br />
functions listed (P, V, and T) are the same for each path. Going from state 1 to state 4 via path 1,<br />
path 2, or path 3 results in P = 1 atm, V = 11 L, and T = 0 K. In other words, the final value<br />
of a state function does not depend on the path taken between the initial and final state. On<br />
the other hand, suppose that instead of different states of a system, the above (state) labels<br />
represented different cities on a map and the three paths represented different routes between them.<br />
It is obvious that distance is not a state function since the distance between city 1 and city 4 depends<br />
on the route taken. In the city analogy, is altitude a state function? Of course distance is not a<br />
thermodynamic variable, but two very important thermodynamic variables that are not state<br />
functions are work and heat.<br />
C. Work and heat