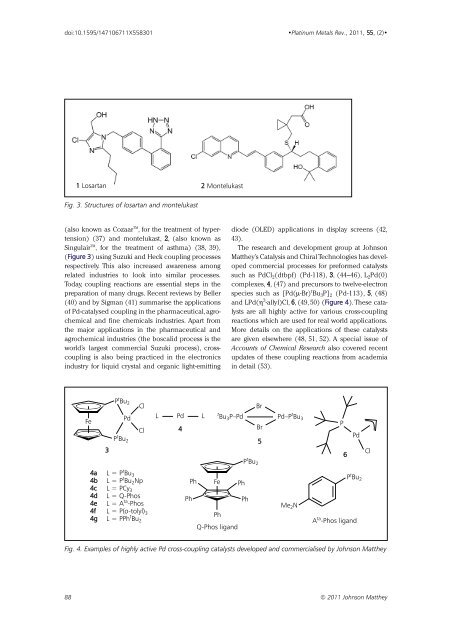
doi:10.1595/147106711X558301 •<strong>Platinum</strong> <strong>Metals</strong> Rev., 2011, 55, (2)• 1 Losartan 2 Montelukast Fig. 3. Structures of losartan and montelukast (also known as Cozaar TM , for the treatment of hypertension) (37) and montelukast, 2, (also known as Singulair TM , for the treatment of asthma) (38, 39), (Figure 3) using Suzuki and Heck coupling processes respectively. This also increased awareness among related industries to look into similar processes. Today, coupling reactions are essential steps in the preparation of many drugs. Recent reviews by Beller (40) and by Sigman (41) summarise the applications of Pd-catalysed coupling in the pharmaceutical,agrochemical and fine chemicals industries. Apart from the major applications in the pharmaceutical and agrochemical industries (the boscalid process is the world’s largest commercial Suzuki process), crosscoupling is also being practiced in the electronics industry for liquid crystal and organic light-emitting diode (OLED) applications in display screens (42, 43). The research and development group at Johnson Matthey’s Catalysis and Chiral Technologies has developed commercial processes for preformed catalysts such as PdCl 2 (dtbpf) (Pd-118), 3, (44–46), L 2 Pd(0) complexes, 4, (47) and precursors to twelve-electron species such as [Pd(µ-Br) t Bu 3 P] 2 (Pd-113), 5, (48) and LPd(η 3 -allyl)Cl, 6, (49, 50) (Figure 4). These catalysts are all highly active for various cross-coupling reactions which are used for real world applications. More details on the applications of these catalysts are given elsewhere (48, 51, 52). A special issue of Accounts of Chemical Research also covered recent updates of these coupling reactions from academia in detail (53). Fe 3 P t Bu 2 Pd P t Bu 2 Cl Cl L Pd 4 L Br t Bu 3 P–Pd Pd–P t Bu 3 Br 5 P t Bu 2 P 6 Pd Cl 4a L = P t Bu 3 4b L = P t Bu 2 Np Ph Fe Ph 4c L = PCy 3 4d L = Q-Phos 4e L = A ta Ph Ph -Phos 4f L = P(o-tolyl) 3 4g L = PPh t Ph Bu 2 Q-Phos ligand Me 2 N P t Bu 2 A ta -Phos ligand Fig. 4. Examples of highly active Pd cross-coupling catalysts developed and commercialised by Johnson Matthey 88 © 2011 Johnson Matthey
doi:10.1595/147106711X558301 •<strong>Platinum</strong> <strong>Metals</strong> Rev., 2011, 55, (2)• In order to address the issue of residual palladium in the final product, several solid-supported preformed palladium complexes have been developed and launched onto the catalyst market (54–56). Conclusions Palladium-catalysed cross-coupling is of great importance to real world applications in the pharmaceutical, agrochemicals, fine chemicals and electronics industries. The area has developed quite rapidly beyond the work of Heck, Negishi and Suzuki, though all three reactions are widely used. Academic groups such as those of Beller, Buchwald, Fu, Hartwig and Nolan as well as industrial groups such as that at Johnson Matthey, are now developing the field even further. Buchwald-Hartwig coupling has become particularly important for developing compounds containing carbon–nitrogen bonds for applications in industry, as well as α-arylation of carbonyl compounds such as ketones, esters, amides, aldehydes etc., and nitriles (57). The significant growth of crosscoupling reactions can be summarised in Professor K. C. Nicolaou’s words: “In the last quarter of the 20th century, a new paradigm for carbon–carbon bond formation has emerged that has enabled considerably the prowess of synthetic organic chemists to assemble complex molecular frameworks and has changed the way we think about synthesis”(58). More detailed articles summarising the history of cross-coupling in the context of the 2010 Nobel Prize in Chemistry with an outlook on the future of crosscoupling will be published elsewhere (59, 60). Glossary Ligand A ta -Phos Cy dppf dtbpf Np Ph Q-Phos t Bu Name p-dimethylaminophenyl(di-tert-butyl)phosphine cyclohexyl 1,1′-bis(diphenylphosphino)ferrocene 1,1′-bis(di-tert-butylphosphino)ferrocene neopentyl phenyl 1,2,3,4,5-pentaphenyl-1′-(di-tert-butylphosphino)ferrocene tert-butyl References 1 T. Colacot, <strong>Platinum</strong> <strong>Metals</strong> Rev., 2008, 52, (3), 172 2 “Metal-Catalyzed Cross-Coupling Reactions”, 2nd Edn., eds. A. de Meijere and F. Diederich, Wiley-VCH, Weinheim, Germany, 2004 3 C. Barnard, <strong>Platinum</strong> <strong>Metals</strong> Rev., 2008, 52, (1), 38 4 ‘Scientific Background on the Nobel Prize in Chemistry 2010: Palladium-Catalyzed Cross Couplings in Organic Synthesis’, The Royal Swedish Academy of Sciences, Stockholm, Sweden, 6th October, 2010: http:// nobelprize.org/nobel_prizes/chemistry/laureates/2010/sci. html (Accessed on 24 January 2011) 5 T. Mizoroki, K. Mori and A. Ozaki, Bull. Chem. Soc. Jpn., 1971, 44, (2), 581 6 K. Mori, T. Mizoroki and A. Ozaki, Bull. Chem. Soc. Jpn., 1973, 46, (5), 1505 7 R. F. Heck, J. Am. Chem. Soc., 1968, 90, (20), 5518 8 R. F. Heck and J. P. Nolley, J. Org. Chem., 1972, 37, (14), 2320 9 H. A. Dieck and R. F. Heck, J. Am. Chem. Soc., 1974, 96, (4), 1133 10 E. Negishi and S. Baba, J. Chem. Soc., Chem. Commun., 1976, (15), 596b 11 E. Negishi, A. O. King and N. Okukado, J. Org. Chem., 1977, 42, (10), 1821 12 A. O. King, N. Okukado and E. Negishi, J. Chem. Soc., Chem. Commun., 1977, (19), 683 13 J. F. Fauvarque and A. Jutand, J. Organomet. Chem., 1977, 132, (2), C17 14 K. Tamao, K. Sumitani, Y. Kiso, M. Zembayashi, A. Fujioka, S. Kodama, I. Nakajima, A. Minato and M. Kumada, Bull. Chem. Soc. Jpn., 1976, 49, (7), 1958 15 K. Tamao, Y. Kiso, K. Sumitani and M. Kumada, J. Am. Chem. Soc., 1972, 94, (26), 9268 16 K. Tamao, K. Sumitani and M. Kumada, J. Am. Chem. Soc., 1972, 94, (12), 4374 17 M. Kumada, in “Organotransition Metal Chemistry”, eds. Y. Ishii and M. Tsutsui, Plenum Press, New York, USA, 1975, p. 211 18 R. J. P. Corriu and J. P. Masse, J. Chem. Soc., Chem. Commun., 1972, (3), 144a 89 © 2011 Johnson Matthey