Nanoparticles for in-vitro and in-vivo biosensing and imaging
Nanoparticles for in-vitro and in-vivo biosensing and imaging
Nanoparticles for in-vitro and in-vivo biosensing and imaging
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
28 Metal nanoparticles<br />
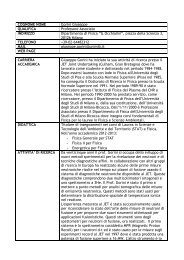
Figure 1.12: Simulation of the surface plasmon absorption <strong>for</strong> gold nanorods of different aspect ratio<br />
by L<strong>in</strong>k <strong>and</strong> coworkers. [52].<br />
calculated <strong>in</strong> Figure 1.12b. The dotted l<strong>in</strong>e represents a plot of an equation derived by<br />
L<strong>in</strong>k <strong>in</strong> order to predict the maximum of the longitud<strong>in</strong>al plasmon b<strong>and</strong> λ max [52]<br />
λ max = (33.34R − 46.31)ɛ m + 472.31 (1.24)<br />
It follows from Equation 1.24 that the maximum of the longitud<strong>in</strong>al plasmon resonance<br />
also l<strong>in</strong>early depends on the medium dielectric constant ɛ m . Hence, with <strong>in</strong>creas<strong>in</strong>g<br />
the medium dielectric constant, there is also a red-shift of λ max <strong>for</strong> a fixed aspect ratio<br />
(Figure 1.12 c with R=3.3).<br />
The fact that gold nanorods exhibit a longitud<strong>in</strong>al SPR <strong>in</strong> the near-<strong>in</strong>frared has<br />
drawn <strong>in</strong>terest of the biophysical community <strong>in</strong> the use of this particles <strong>in</strong> <strong>in</strong>-<strong>vivo</strong> study<br />
because of larger penetration depth of the laser light: <strong>in</strong> the region between 650-900 nm,<br />
≥ 300 µm depend<strong>in</strong>g on tissue types.<br />
Solid gold nanorods have several advantages over other contrast agents; their synthesis<br />
with various aspect ratios <strong>and</strong> tunable absorption wavelength <strong>in</strong> the near <strong>in</strong>frared region<br />
[58] [59] [8] is quite simple <strong>and</strong> well-established. The typical size of the nanorods (50-200<br />
nm) is potentially useful <strong>in</strong> applications such as drug delivery <strong>and</strong> gene therapy. In addition,<br />
the biosafety of gold is well known <strong>and</strong> it has been used <strong>in</strong> <strong>vivo</strong> s<strong>in</strong>ce the 1950’s [60]<br />
<strong>and</strong> recently the low cytotoxicity of gold nanoparticles <strong>in</strong> human cells has been studied<br />
<strong>in</strong> detail by Wyatt <strong>and</strong> coworkers [61].<br />
Moreover, gold nanorods can be used as photo-absorbers <strong>in</strong> NIR <strong>and</strong> realize photothermal<br />
therapy. Thus, upon laser irradiation at the surface plasmon absorption b<strong>and</strong>, the<br />
nanoparticles absorb energy <strong>and</strong> then immediately transfer it <strong>in</strong>to heat. If the nanoparticles<br />
are <strong>in</strong>corporated or <strong>in</strong>cubated with biomolecules, cells or tissues, heat causes the<br />
sharp <strong>in</strong>crease on the local temperature around the nanoparticles <strong>and</strong> thus damages to<br />
the surround<strong>in</strong>g materials. This process is called photothermal destruction <strong>and</strong> can be